A Comparative Study on the Two Different Methods IRMS and CRDS for Estimation of δ13C (‰) of Honey Samples
Abstract
We have analyzed the isotopic ratio of carbon (δ13C ‰) of honey samples (n=10) employing two different methods namely IRMS and CRDS. In general, the honey samples satisfied the specification suggested by codex. The δ13C values for the tested honey samples ranged from -12.0 to -26.6‰. A lower value indicates the possible adulteration of honey with sugar syrup or honey from the beehive in which bees were fed sugar syrup coming from C4 plants. Results obtained from two methods showed that there is no significant difference in the values for respective honey samples. However, higher accuracy was observed in the results obtained from CRDS system. Comparable and consistent results, lower maintenance cost and ease of handling make CRDS system a better alternative to traditional state-of-the-art IRMS method for carrying out routine analysis of honey samples.
Keywords:
Adulteration, Carbon, High precision, Economic, C3, C4 pathwaysINTRODUCTION
Isotopic analysis is enormously useful in wide range of disciplines engaged in environmental and climate science, agriculture, food science and medical or biochemical research. It also has remarkable applications in the industrial scale particularly in the food industry (edible oil, wine, fruit juice and product industry etc. to cite few examples) often to understand the source of the desired product which could lead to the food safety and ensures human health. Similarly, the application of isotopic analysis is useful to find out the source of honey which in turn helps in the assessment of honey quality. Honeybees collect nectar from the flowering plants. The nectar undergoes a series of chemical change through physical and chemical (enzymatic viz. invertase, diastase and glucose oxidase) processes and ultimately transforms into honey (Nicolson and Human, 2008; Doner, 1977 cited from Brodschneider and Crailsheim 2010). Besides higher nutritional value honey is regarded more often because of its several medicinal properties (Khan et al., 2007; Mandal and Mandal, 2011; Cortes et al., 2011; Hadagali and Chua, 2014). Thus it is utmost important to ensure the quality of honey. Realizing the importance several countries frame regulation for honey specification including moisture content, sugar profile, HMF level etc. in order to avoid adulteration (Bogdanov et al., 1999). First identified and later establishment of the fact that the isotopic composition of plants differs widely in the 1950s and 1970s respectively opened the scope of isotopic analysis to distinguish plants based on their photosynthetic pathways i.e. C3 and C4 pathways (Ehleringer, 1991). Stable carbon isotope ratio analysis, generally expressed as 13C/12C=δ13C (‰) has been used as a tool to detect the difference. This could help in identifying the honey thus nectar source from which nectar has been collected. Generally honeybees collect nectar from dicotyledonous C3 plants characterized with higher δ13C (ranging within 23‰ to 28‰) in comparison to δ13C values of monocotyledonous C4 plants (9‰ to 15 ‰) (Anklam, 1998; Simsek et al., 2012; Guler et al., 2014 cited from Soares et al., 2017). Cane and corn, both C4 plants, are generally used as source of industrial sugar and sugar syrup. Thus authentic honey samples are expected to have characteristic properties of C3 plants i.e. higher δ13C values while honey adulterated with sugar syrup should have characteristic properties of C4 plants i.e. lower δ13C values. However, the traditional method used for isotope ratio measurement is Isotopic Ratio Mass Spectrometry (IRMS) which has been included in the official method of analysis for several regulations (AOAC official method 998.12). However, because of bulkiness, expensive and time consumption IRMS is difficult to operate for quick and regular use in medium scale laboratory particularly situated in remote place. On the other hand, recently developed Cavity Ring-Down Spectroscopy (CRDS) system has the advantages with respect to the said characters over IRMS methods. Thus the current research has been undertaken to compare these two methods namely IRMS and CRDS for the measurement of the isotopic ratio of honey samples.
MATERIALS AND METHODS
Honey samples collection: Honey samples were collected from Andong National University collaborative beekeepers in Gyeongsangbuk province and Jeju. Representatives of floral origins are chestnut (Castanea crenata Siebold and Zucc., Fagaceae), false acacia (Robinia pseudoacacia L., Fabaceae) and rapeseed (Brassica napus L., Brassicaceae). Honey samples were collected in airtight sterile glass bottles, brought to the laboratory and stored in condition at room temperature (25°C) and relative humidity approximately 55% until further analysis.
Physicochemical analyses of the honey sample: All the chemical analyses including moisture content, electric conductivity (EC), sugar analysis, polyphenol estimation were conducted following the standard method by Codex (2001), Jung and Chon (2016) and Jung et al. (2017).
Isotopic ratio analytical methods
IRMS: We used Delta V Plus with characterized Ion source 3KV and monitored masses m/z 1~8 (Thermo Fisher Scientific) Continuous Flow Isotope Ratio Mass Spectrometer in order to determine δ13C in the honey samples. The isotopic ratio was measured following the standard method of Craig (1957) adopted in the AOAC methodology (AOAC 998.12). Honey sample in a ceramic boat has been placed in the combustion system admitted to 600mm mercury (Hg), oxygen over purified CuO at 700°C followed by liquid nitrogen trap. The sample was heated at ≥850°C and CO2 was condensed in liquid nitrogen trap. Further, the condensed CO2 underwent in the purification system and identified with mass spectrometer especially designed for isotope ratio measurement. Mass spectrometer separates ionic forms of molecules according to their specific mass-to-charge ratio (m/z). The value of δ13C (‰) is calculated as follows:
CRDS: A trace of the honey sample was placed in the combustion module (Picarro G2121-I Cavity Ring-Down Spectroscopy isotopic CO2 gas analyzer, Picarro Inc. California, USA). The CO2 resulting from combustion chamber was collected via Picarro’s Liaison High throughput interface. Nitrogen was used as carrier gas with a flow rate of 90mL/min and oxygen was used as combustion gas. After an appropriate mixing time to ensure the isotopic equilibrium, the collected CO2 was automatically passed into CRDS sampling chamber for δ13C analysis. Wavelength-scanned CRDS system operates in the infra-red. The basic working principle lies in the theory stating that different isotopologues have different absorption spectra in infra-red. Molecules rotate or vibrate at infra-red measurable frequencies. The relation between wavelength and reduced mass of a molecule is as follows where k is the spring constant and μ is the reduced mass (mass of two atoms) of A-B system.
In time of mass of either atom changes as a consequence of results reduced mass changes and thus wavelength changes. In this CRDS technique, there is a laser beam in a cavity with two high reflectivity mirrors with non- zero transmission. The decay of the light pulse is monitored by a detector which measures the intensity of the light transmitted through one of the mirrors. The attenuation of light travelling through the sample is measured (O’keefe and Deacon, 1988; Picarro CRDS information).
Statistical analysis: In order to enhance the reliability and reproducibility of the results, each honey sample was analyzed in triplicate by each method. To examine the difference between the results obtained from two different methods, results were subjected to the t-test. For the comparison of the accuracy of the analytical values from two methods, regression analysis was conducted along with calculation of coefficients of variation (CV, %). Statistical analyses were carried out using SPSS software package (SPSS ver. 24.0, IBM).
RESULTS AND DISCUSSION
Physicochemical properties of tested honey samples
Table 1 represents the physicochemical properties of the honey samples analyzed. Moisture content was found within a range of 16.9 to 21.3%. The permissible value for moisture is 20% according to most of the regulations including Codex standard (Bogdanov et al., 1999). Bees regurgitate nectar over 20 minutes of time and deposit it in the honeycomb once its moisture content is about 20%. Further in order to condense it more bees fan it to speed up evaporation. The less moisture content minimizes the scope of microbial contamination and honey can be preserved well. Moisture content over 25% increases the scope of fermentation of honey. Electrical conductivity (EC) is an indication of ionisable acids and compounds in aqueous solution. The values obtained for the tested honey samples were found within the range specified by honey Codex (0.8 mS/cM i.e. 80 mS/M) except the value we got for sample no.8. The pH range of the tested honey samples falls within 3.1 to 5.7 which are in agreement with the other published reports (Azeredo et al., 2003; Solayman et al., 2016, National Honey Board). The acidity of honey is caused by organic acids like citric, oxalic, acetic etc. either from nectar or secreted from bees. Carbohydrate composition of the honey samples followed the general trend i.e. fructose was found to be predominate followed by glucose. In order to compliance to Codex standard for honey the tested honey samples contained together fructose and glucose more than 60g, however, for sample no. 2 together fructose and glucose amount together stood just on margin. The total polyphenol content of the tested honey samples ranged from 194.9 to 266.5mg GAE per 100g of honey. Results of present study showed that tested honey samples contained higher polyphenols than that reported in reports of Otilia et al. (2005), Wilczy´nska (2010) and, however within the range of several scientific reports (Vit et al., 2008; Ciappini and Stoppani, 2014; Sime et al., 2015). Although phosphotungtic acid and phosphomolibdic acid often reacts with non-phenolic reducing compounds like ascorbic acid, some sugars and amino acid leading to overestimation, this Folin-Ciacalteu method remains useful and commonly used to evaluate the relative content of polyphenolic contents.
δ13C values for the tested honey samples and comparison of two methods
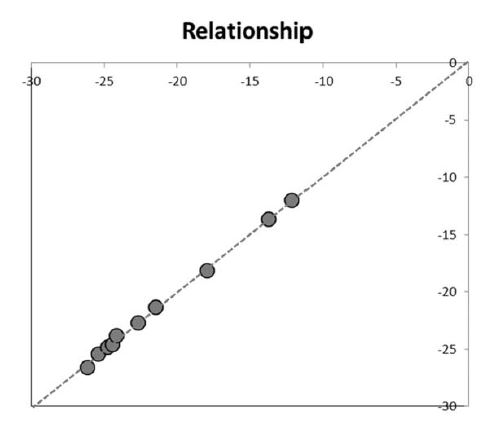
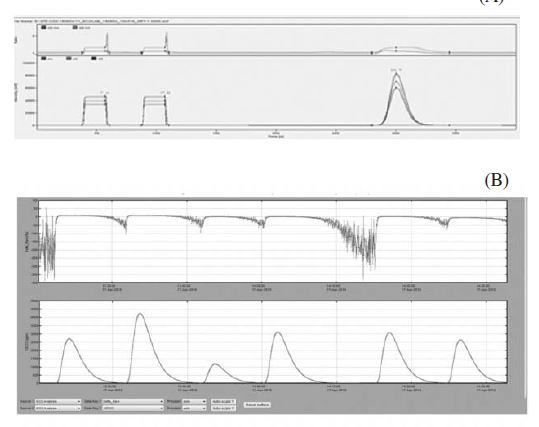
The δ13C values for the tested honey samples ranged from -12.0 to -26.6‰ (Table 2). Results obtained from two methods showed that there is no significant difference (t-0.025, dF=18, p>0.05). The observation is in agreement with other published reports in which the results obtained from both methodologies were found comparable (Balslev-Clausen et al., 2013; Jung and Chon, 2016; Mantha et al., 2018). The similar observation also found true for 2H/1H and 18O/16O isotopic ratio analysis (Santos et al., 2013; Maruyama and Tada, 2014). However, except for sample no. 2, higher accuracy has been accounted for CRDS method. Regression analysis with regression coefficient (R) 0.998 (y=x) demonstrated the comparability and accuracy of the newly employed method of CRDS with the state-of-the-art IRMS method (Fig. 1). Moreover, less coefficient of variation (CV%) value obtained in CRDS method in comparison to IRMS reflects the higher precision and accuracy of the CRDS method (Table 2). Further, in the spectra in CRDS, there was lack of interference from other molecular species indicating the non-requirement of the sample processing in order to dry water or other gaseous molecules (Fig. 2).
The lower values were obtained for sample no. 3, 4 and 6 clearly because these honey samples were collected from beehive in which bees were fed with sugar syrup coming from C4 plant source. Honey adulteration is one example of economically driven adulteration and a quick and easy process is always desired to ensure the quality of the product. Of course, an element analyzer, liquid chromatography coupled with isotope ratio mass spectrometry is a reliable method in the context but it requires much maintenance cost and high technical knowledge to handle the instrument. Thus it often underserves the scientific society as well as industry. On the other hand, CRDS method is more integrated, easy to handle and moreover the maintenance cost is also comparatively lower. Conversion of organic samples including honey to CO2 for isotopic analysis is performed by dry combustion with excess oxygen. Prior to combustion, the sample should be dried completely in order to achieve greater accuracy. Otherwise, it could lead to the explosion of tubes during combustion due to a higher volume of water vapor, although a little improvement exists. Moreover, sometimes different gaseous species interferes in the spectra like carbonate. Thus the sample preparation is crucial for mass spectrometry because most of the error in this method comes from the sample preparation (Boutton, 1991). However, in the combustion module of CRDS combustion product is carried onward from the combustion furnace through a water trap made up of Magnesium Perchlorate to the Liaison High throughput interface. Also in the traditional IRMS method there is a chance of interfering with undesired molecules. To cite an example samples containing carbonate carbon should be treated with dilute hydrochloric acid or phosphoric acid in order to avoid interfere of carbonate carbon which is often enriched with 13C and might influence the result (Boutton, 1991; Showers and Angle, 1986; Balslev-Clausen et al., 2013).However, there is a limitation of detecting adulteration with sugar from C3 plant origin like sugar from sugar beet through this method.
Based on comparability and consistency of results obtained from CRDS method, lower maintenance cost, less time consumption and easy handling we conclude that high-performance CRDS system a better alternative to traditional state-of-the-art IRMS method for carrying out routine analysis of honey samples.
Acknowledgments
The authors acknowledge Central Public laboratory facilities for carrying out the experiments. This research was partly supported from Andong National University Research Fund in 2017-2018.
References
-
Anklam, E., (1998), A review of the analytical method to determine the geographical and botanical origin of honey, Food Chem, 63, p549-562.
[https://doi.org/10.1016/s0308-8146(98)00057-0]

- AOAC, Association of Official Analytical Chemists, (2005), Official methods of analysis (998.12). C-4 plant sugars in honey. Internal standard stable carbon isotope ratio method, Official Meth. Anal. 44, p33.
-
Azeredo, L. da C., M.A.A. Azeredo, S.R. de Souza, V.M.L. Dutra, (2003), Protein contents and physicochemical properties in honey samples of Apis mellifera of different floral origins, Food Chem, 80, p249-254.
[https://doi.org/10.1016/s0308-8146(02)00261-3]

- Balslev-Clausen, D., T.W. Dahl, N. Saad, and M.T. Rosing, (2013), Precise and accurate δ13C analysis of rock samples using flashcombustion-cavity ring down laser, J. Anal At. Spectrom, 28, p516-523.
-
Bogdanov, S., C. Lüllmann, P. Martin, W. von der Ohe, H. Russmann, G. Vorwohl, L.P. Oddo, A-G. Sabatini, G.L. Marcazzan, R. Piro, C. Flamini, M. Morlet, J. Lhertier, R. Borneck, P. Marioleas, A. Tsigouri, J. Kerkvilet, A. Ortiz, T. Ivanov, B. D’Arcy, B. Mossel, and P. Vit, (1999), Honey quality and international regulatory standards: review by the International Honey Commission, Bee World, 80, p61-69.
[https://doi.org/10.1080/0005772x.1999.11099428]

- Boutton, T.W., (1991), Stable carbon isotope ratios of natural materials: I. Sample preparation and mass spectrometric analysis, p155-171, In Coleman, D.C., and B., Fry eds, Carbon Isotope Techniques, Academic Press, p286.
- Brodschneider, R., and K. Crailsheim, (2010), Nutrition and health in honeybees, Apidologie, 41, p278-294.
-
Ciappini, M.C., and F.S. Stoppani, (2014), Determination of antioxidant capacity, flavonoids, and total phenolic content in Eucalyptus and clover honeys, J.Apic. Sci, 58, p103-111.
[https://doi.org/10.2478/jas-2014-0010]

- CODEX, (2001), Revised CODEX Standard for honey, CODEX Stan.12-1981, Rev. 1 (1987). Rev. 2 (2001).
- Cortes, M.E., and P. Vigil, (2011), The medicinal value of honey: a review on its benefits to human health, with a special focus on its effects on glycemic regulation, Cien. Inv. Agr, 38, p303-317.
-
Craig, H., (1957), Isotopic standards for carbon and oxygen and correction factors for mass spectrometric analysis of carbon dioxide, Geochimicaet Cosmochimica Acta, 12, p133-149.
[https://doi.org/10.1016/0016-7037(57)90024-8]

-
Doner, L.W., (1977), The sugars of honey- a review, J. Sci. Food Agric, 28, p443-456.
[https://doi.org/10.1002/jsfa.2740280508]

- Ehleringer, J.R., (1991), 13C/12C fractionation and its utility in terrestrial Plant studies, p187-200, In Coleman, D.C., and B., Fry eds, Carbon Isotope Techniques, Academic Press, p286.
- Guler, A.H., H. Kocaokutgen, A.V. Garipoglu, H. Onder, D. Ekinci, and S. Biyik, (2014), Detection of adulterated honey produced by honeybee (Apis mellifera L.) colonies fed with different levels of commercial industrial sugar (C(3) and C(4) plants) syrups by the carbon isotope ratio analysis, Food Chem, 155, p155-160.
-
Hadagali, M.D., and L.S. Chua, (2014), The anti-inflammatory and wound healing properties of honey, European Food Res. Technol, 239, p1003-1014.
[https://doi.org/10.1007/s00217-014-2297-6]

-
Jung, C., E. Cho, S. Lee, and J-W. Chon, (2017), Quality characteristics of honey on the market: case study from Daegu-GyeongBuk province, J. Apic, 32, p51-58.
[https://doi.org/10.17519/apiculture.2017.04.32.1.51]

-
Jung, C., and J-W. Chon, (2016), Quality assessment of honey from different floral origin in Korea, J. Apic, 31, p103-111.
[https://doi.org/10.17519/apiculture.2016.06.31.2.103]

-
Khan, F.R., Z.U. Abadin, and N. Rauf, (2007), Honey: nutritionaland medicinal value, Int. J. Clin. Prac, 61, p1705-1707.
[https://doi.org/10.1111/j.1742-1241.2007.01417.x]

-
Mandal, M.D., and S. Mandal, (2011), Honey: its medicinal property and antibacterial activity, Asian Pac. J. Trop. Biomed, 1, p154-160.
[https://doi.org/10.1016/s2221-1691(11)60016-6]

-
Mantha, M., J.R. Urban, W.A. Mark, A. Chernushev, and K.M. Kubachka, (2018), Direct comparison of cavity ring down-spectrometry and isotope ratio mass spectrometry for detection of sugar adulteration in honey samples, J. AOAC Int.
[https://doi.org/10.5740/jaoacint.17-0491]

- Maruyama, S., and Y. Tada, (2014), Comparison of water analysis between cavity ring-down spectroscopy and isotope ratio mass spectrometry, Geochemical Journal, 48, p105-109.
- National Honeybee Board, pH and acids in honey [www.nhb.org], Web source:https://www.bjcp.org/mead/ph_acid.pdf. Accessed 23rd June, 2018.
-
Nicolson, S.W., and H. Human, (2008), Bees get a head start on honey production, Biol. Lett, 4, p299-301.
[https://doi.org/10.1098/rsbl.2008.0034]

- O’ Keefe, O., and D.A.G. Deacon, (1988), Cavity ring-down optical spectrometer for absorption measurements using pulsed laser sources, Reviews of Scientific Instruments, 59, p2544-2551.
- Otilia, B., C. Socaciu, L. Marghitas, and D. Dezmirean, (2005), Correlation between total phenols, flavonoids, coulour intensity and botanical origin of some honeys from Transilvania region, Bull. Univ. Agric. Sci. Vet. Med, 61, p349-353.
- Picarro, CRDS information, Web source: https://www.picarro.com/technology/cavity_ring_down_spectroscopy. Accessed 22nd June, 2018.
-
Santos, T.H.R., M.R. Zucchi, T. Lemaire, and A.E.G. Azevedo, (2013), Comparison between IRMS and CRDS methods in the determination of isotopic ratios 2H/1H and 18O/16O in water, AIP Conf. Proc, 1529, p107-109.
[https://doi.org/10.1063/1.4804097]

-
Showers, W.J., and D.G. Angle, (1986), Stable isotopic characterization of organic carbon accumulation on the Amazon continental shelf., Cont. Shelf. Res, 6, p227-244.
[https://doi.org/10.1016/0278-4343(86)90062-2]

-
Sime, D., M. Atlabachew, M. Redi-Abshiro, and T. Zewde, (2015), Total phenols and antioxidant activities of natural honeys and propolis collected from different geographical regions of Ethiopia, Bull. Chem. Soc. Ethiop, 29, p163-172.
[https://doi.org/10.4314/bcse.v29i2.1]

- Sismek, A., M. Bilsel, and A.C. Goren, (2012), 13C/12C pattern of honey from Turkey and determination of adulteration in commercially available honey sample using EAIRMS, Food Chem, 130, p1115-1121.
-
Soares, S., J.S. Amaral, M.B.P.P. Oliveira, and I. Mafra, (2017), A comprehensive review on the main honey authentication issues: production and origin, Comprehensive Reviews in Food Science and Food Safety, 16, p1072-1100.
[https://doi.org/10.1111/1541-4337.12278]

-
Solayman, M., M.A. Islam, S. Paul, Y. Ali, M.I. Khalil, N. Alam, and S.H. Gan, (2016), Physicochemical properties, minerals, trace elements, and heavy metals in honey of different origins: a comprehensive review, Comprehensive Reviews in Food Science and Food Safety, 15, p219-233.
[https://doi.org/10.1111/1541-4337.12182]

- SPSS statistics ver. 24.0. IBM.
- Vit, P., M.G. Gutierrez, D. Titera, M. Vendar, and A.J.R. Malaver, (2008), Mieleschecascategorizadassegunsu activated antioxidante, Acta de Bioquimica Clinical Latino-americana, 42, p237-244.
- Wilczy´nska, A., (2010), Phenolic content and antioxidant activity of different types of polish honey- a short report, Polish J. Food Nutr. Sci, 60, p309-313.