Comparison of Acute Toxicity of Different Groups of Pesticides to Honey Bee Workers (Apis Mellifera L.)
Abstract
Honey bees (Apis mellifera) forage in agricultural areas, and are exposed to diverse pesticide poisoning. Toxic effects on Apis mellifera of different groups of pesticides were tested in the laboratory; fungicide (Metconazole), herbicide (Glyphosate), acaricide (Amitraz), organophosphate insecticide (Fenitrothion) and neonicotinoid insecticides (Thiacloprid, Thiamethoxam, Imidacloprid, Acetamiprid, Dinotefuran and Clothianidin). Commercial formulations were serially diluted from the recommended concentration (RC) to 10-6 times to carry out feeding and contact tests. Toxicity was transformed into lethal dose (LD50) and hazard question (HQ). The acute toxicity of pesticides showed similar patterns between feeding and contact tests. But feeding tests showed greater toxic to honey bee than contact test. The organophosphate and nitro-neonicotinoid insecticides were highly toxic with HQ values ranging greater than 1. However, cyano-neonicotinoids of Thiacloprid and Acetamiprid showed low toxicity. Even at the RC, 24 hr mortaliti es were 18 and 30%. The acaricide (Amitraz) showed intermediate level of toxicity at RC but negligible at the concentration lower than 10-1 times. A fungicide (Metconazole) and herbicide (Glyphosate) showed minimal impacts. The results imply that the selective use of pesticides could help conservation of pollinators in agricultural production systems.
Keywords:
Organophosphate, Nitro-neonicotinoid, Cyano-neonicotinoids, Metconazole, GlyphosateINTRODUCTION
Honey bees are efficient pollinators, worker honey bees collect pollen and nectar from as far as 10 km from the hive. During that time, they often encounter toxic materials of both natural and synthetic origin and may carry these xenobiotics back to the colony. Nectar and pollen may contain environmental pollutants or pesticides drawn from the soil and bees also collect water from environmental sources. Such surface water or guttation water may be contaminated with high concentrations of toxic metals and insecticides (Johnson, 2015). A variety of different pesticides which have the potential to harm honey bees are in use by agriculturists. In recent years, scientists have focused on honey bee toxicology and the effects of pesticides, particularly insecticides, given an exposure to bees.
An analysis of honey bees and their hive wax and pollen in the United States revealed that the majority of the samples was contaminated with at least one pesticide (Mullin et al., 2010). Similarly, 37 insecticide and fungicide chemicals were detected in honey bees and hive products sampled in France (Lambert et al., 2013). Annual agricultural use of pesticides has increased rapidly from the 1970s to 1990s in South Korea, but has declined since 2001. The quantity of pesticides used in 2011 was reported as 19.131 tons, and comprised 34.7% insecticides, 28.0% fungicides, and 27.1% herbicides. Organophosphates were the most used among the top pesticides (Cha et al., 2014).
Our tests involved oral and spray trials in order to assess the toxic effects of the pesticides often used in the agricultural field. Pesticides are toxic chemicals; they are designed to specifically control a target group of organisms. Insecticides are chemicals used to kill insects, so it is unsurprising that many insecticides have the potential to harm honey bees. For herbicides and fungicides, if the target of such chemicals is not the insects, they can be expected to be safe to the bees. However, extensive and prolonged uses of herbicides may affect the bees’ colonies (Goulson et al., 2015) and their productivity in various ways.
Neonicotinoid insecticides are one of the most effective classes of insecticides in the world. They are neurotoxins which act as nicotinic acetylcholine receptor agonists in the central nervous system of insects and cause overstimulation, paralysis, and death. But different neonicotinoid insecticides have slightly different chemical structures, some of which are more toxic to bees. The neonicotinoids are of 2 subgroups, those with a nitro functional group (-NO2) and those with a cyano functional group (-C=N) in their molecular structure. The nitro-group neonicotinoids (including Imidacloprid, Dinotefuran, Clothianidin and Thiamethoxam) are much more toxic to bees than the cyano-group neonicotinoid, which include Acetamiprid and Thiacloprid (Blacquiere et al., 2012; Brandt et al., 2016).
Fenitrothion is a phosphorothioate (organophosphate) insecticide that is widely used worldwide. Organophosphate are toxic to honeybees by their own and with specific actions on the insect nervous system. For herbicides and fungicides, if the target of such chemicals are not the insects, these chemicals are probably safe to bees. However, extensive and prolonged use of herbicides are likely to affect bees colony and bees productivity (Goulson et al., 2015).
We therefore tested some fungicide, herbicide and acaricides together with insecticides. Metconazole is a systemic triazole fungicide, acting as ergosterol biosynthesis inhibitors. These fungicides are generally of low toxicity to honey bees, but there is a synergistic effect of the fungicides when delivered together with insecticides, as evidenced in the literature. For example, the increased toxicity of pyrethroid insecticides to honeybees in the presence of ergosterol biosynthesis inhibitor fungicides has been reported (Pilling et al., 1995; Johnson et al., 2012). Moreover, a combination of some fungicides with insecticides has been revealed to be more deadly to the bees than either chemical alone (Iwasa et al., 2004; Johnson et al., 2013). The study of Thompson et al. (2014) has shown that the dose of fungicide that the bee receives is a key factor in determining the toxicity of the neonicotinoids. Glyphosate (N- (phosphonomethyl) glycine) is a broad-spectrum systemic herbicide and crop desiccant. Amitraz is a non-systemic acaricide and insecticide.
The objectives of the study are to determine and compare the acute toxicity of different pesticide formulation to honey bees, and to estimate the lethal dose (LD) values and Hazard Quotient (HQ) of each pesticide. To know which kind of exposure (oral and contact) and which of these pesticides would cause more serious effects on Apis mellifera, is of importance and may affect future issues of application. Thus we set the hypothesis as; nitro-neonicotinoids and Fenitrothion are more toxic, than acaricide, fungicide or herbicide, and feeding would pose more toxic than contact by spraying.
MATERIALS AND METHODS
Honey bees
We used worker honey bees (A. mellifera) of the Honey Bee Laboratory at Andong National University, Andong, Republic of Korea. Worker bees were collected from the hive by using a small amount of smoke, brushing them from the combs and transferring them into cylindrical iron cages. The cages with the bees were placed in the laboratory room (24℃) until needed for the test. Before treatment, we prepared and counted the worker bees in each cage, using anaesthetization with CO2 gas.
Pesticide
Commercial formulations available in Korea were used. To estimate the lethal concentration, we had exposed each pesticide with serial dilution from the producer’s recommended concentration to 10-6 times dilution by the factor of 10. Second grade distilled water was used for the dilution.
Tests were conducted using cylindrical cages (30 cm high, 25 cm diameter). The cage wall’s consisted of iron with a hole, and a cover and bottom made out of plastic. Into each cage 15~20 worker honey bees from the hive were placed.
Feeding test
Collected worker honeybees (30~40 individuals per cage) were starved for 2 hours before testing in the wire-mesh cage in the lab began. 5 mL pesticide contaminated 50% sugar solution with different concentration was provided in the plastic feeding dish on a cotton pad for 1 hour, so that the bees had free access to the pesticide contamination. After 1 hour, the contaminated feeding dish was removed and the weight-difference was measured to estimate the amount taken up by bees.
The feeding dish was then replaced with non-contaminated 50% sugar solution. Control bees were fed only 50% sugar solution. Each treatment had 3 replications. Mortality and other abnormal behaviors were observed at 1 h, 3 h, 6 h, 12 h, 24 h, 48 h after treatment. If no appendage movement was observed, we considered it as a dead bee.
Contact test
100 mL of the pesticide solution with different concentrations were prepared. After housing 30~40 individual honeybees in the mesh cage, we sprayed the pesticide solution with 600 mL hand sprayers (KOMAX G600, Sansoo Co., LTD, Korea) by standard methods (from 15 cm distance, 10 times). After 30 min, honey bees were measured and the weight-difference of bees to estimate the amount of pesticide taken up by them was recorded. Then these bees were transferred into a new cage and provided 50% sugar solution. Each treatment had 3 replications. Mortality and other abnormal behaviors were observed at 1 h, 3 h, 6 h, 12 h, 24 h, 48 h after treatment.
Statistical analysis
We determined the Lethal Dose (LD50) for both feeding and contact test, using LD50 or LD50 using Probit Analysis (Veterinary Pharamcology & Toxicology, SVVU, India) with Excel. We used the LC50, when calculating Hazard Quotients. Hazard Quotient (HQ)= Exposure Concentration/Reference Concentration (RfC) relative to the field application adopted for field concentration determination. If the Hazard Quotient is less than 1, no adverse effects, but if the Hazard Quotient is greater than 1, adverse health effects occur. Two-way ANOVA analysis, followed by Tukey multiple comparison tests (SPSS 16.0), was used to compare the effects of pesticides between feeding test and spray test.
RESULTS
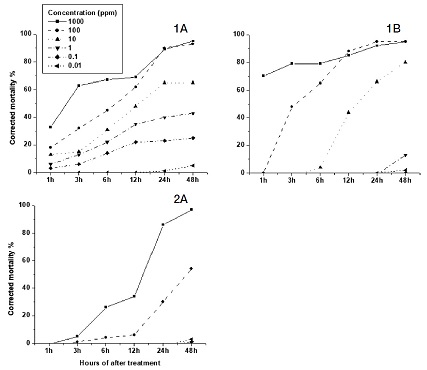
Neonicotinoid insecticides, with the exception of Thiacloprid and Acetamiprid showed the highest toxicity. Thiacloprid insecticide showed higher mortality only at high concentration of 500 ppm, with mortality rate of 18 and 50% for feeding and contact tests, respectively (Fig. 1, 1A, B). The feeding test resulted in a HQ value of 1 in only 48 hours (Table 2).
The feeding test (A) and contact test (B) of cyano - neonicotinoid insecticides compared for mortality percent different concentrations. Thiacloprid (1) and Acetamiprid (2).
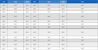
Comparison of LD50 at 24 hr and 48 hr after treatment, and HQ values in feeding and contact test involving honey bee
Acetamiprid showed mortalities lower than 40% for all concentration in the feeding test (Fig. 1, 2A), but in the contact test, maximum motality was less than 20% (Fig. 1, 2B). The comparative means were significantly different (p=0.006) between both tests (Table 2).
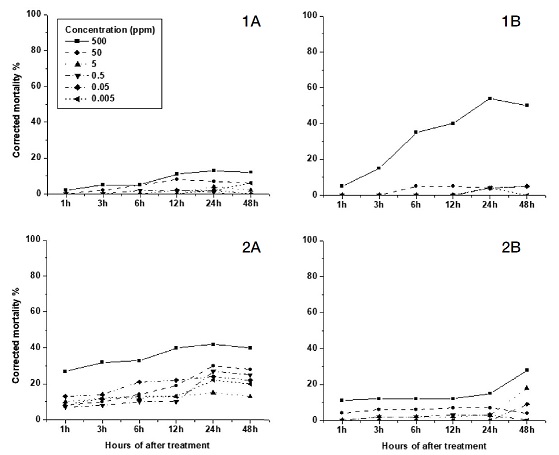
Imidacloprid showed high mortalities for all concentration during test hours in the feeding test. 24 hr mortality of 500 and 50 ppm were 90 and 70% respectively. For low concentration, mortality was greater than 50% in 48 hour (Fig. 2, 1A). Only high concentration showed more toxic effective in contact test (Fig. 2, 1B).
Feeding test (A) and contact test (B) of nitro - Neonicotinoid insecticides compared for mortality different concentrations. Imidaclprid (1), Clothianidin (2), Dinotefuran (3) and Thiamethoxam (4).
The difference between both tests is significant (p=0.018) (Table 2).
Clothianidin, Dinotefuran and Thiamethoxam gave almost the same results in both tests. Mortality of Clothianidin and Dinotefuran exposures were greater than 50% for high concentrations in the first 6 hours (Fig. 2, 2A, 3A). For Clothianidin a mortality rate of 500 ppm was greater than 50% in the first 1 hour for both tests (Fig. 1, 2A, B). In the spray trial, for low concentrations of Clothianidin, mortality was lower than feeding test (Fig. 2, 2B).
All concentrations of Dinotefuran resulted in greater toxic effectiveness (greater than 80% except 0.005 ppm) in the feeding test, than in the contact test; only the first two high concentrations resulted in higher mortality. The difference is significant between both test (p=0.001) (Table 2).
In the feeding of Thiamethoxam, the first three high concentrations showed greater than 80% mortality and the next three concentrations were less than 50% (Fig. 2, 4A). However, in the contact test, the last three concentractions produced no mortalities (Fig. 1, 4B).
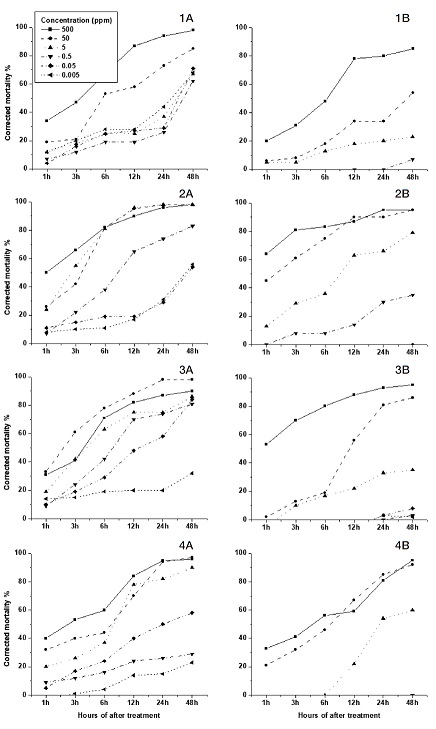
The Fenitrothion (Organophosphate insecticide) was highly toxic as was in the cases of nitro-group neonicotinoids. Feeding caused more toxic effects than the contact exposures (Fig. 3, 1B). Amitraz which is used as an acaricide and also insecticide showed noticible toxicity only at the highest concentration, recommended concentration in the feeding test (Fig. 3, 2A). But Hazard Quotient (HQ) was less than 1 (Table 2). Glyphosate (herbicide) and Metconazole (fungicide) showed mortalities less than 10% even at the highest concentration.
DISCUSSION
Honey bees are exposed to diverse groups of pesticides in agroecosystem. This study showed the toxicities from pesticide exposures are highly variable relative to the groups of pesticides. Organophosphate insecticide of Fenitrothion, and nitro-neonicotinoid insecticides are more toxic than cyano-neonicotinoids (Thiacloprid and Acetamiprid). The acaricide (Amitraz) showed an intermediate level of mortality while the tested herbicide and fungicide showed minimal impacts.
When comparing the results of feeding and contact test, feeding exposure was more toxic to honey bees than the contact exposure. The differences involving Imidacloprid and Dinotefuran were significantly different.
Cyano-neonicotinoid insecticides are relatively less toxic to honey bees than other insecticides. However, Thiacloprid affects A. mellifera’s behavior and immune system (Bant et al., 2016), and neonicotinoids affect the individual immunocompetence of honey bees, possibly leading to an impaired disease resistance capacity. Research results Acetamiprid lower toxic effects than other neonicotinoid insecticides on honey bees. Mortality caused by Acetamiprid was 50.85% at the field concentration of 100 ppm in feeding test (Laurino et al., 2011). Iwasa et al. (2004) suggested that P450s are an important mechanism for Acetamiprid and Thiacloprid detoxification and their low toxicity to honey bees. In this study, Acetamiprid showed mortality of less than 50% until 24 hr in the feeding test and less than 48% in the contact test (in 48 hr).
The LD50 value of nitro-neonicotinoid insecticides (Thiamethoxam, Imidacloprid, Dinotefuran and Clothianidin) were lower than other pesticides in both tests. And Hazard Quotients of high toxicity neonicotinoids showed values greater than one. The LC50 at 12 hr, Thiamethoxam and Clothianidin are resulted similar to result of Lee et al. (2015).
Thiamethoxam is highly toxic both in ingestion and indirect contact. However, the latter is somehow less dangerous at reduced concentrations. In the indirect contact test Thiamethoxam was lethal at a concentration 20 times lower than the field one, showing a degree of danger long after administration (Laurino et al., 2011). The European Commission (2013) voted to place a moratorium on the use of three neonicotinoid insecticides (imidacloprid, thiamethoxam and clothianidin) after the European Food Safety Authority (EFSA) determined that there were “high acute risks” to bees (EFSA, 2013). The order of toxicities for the insecticides was ranked as clothianidin>thiamethoxam>dinotefuran (Liu et al., 2017). In this study, they showed almost the same result.
The avoidance behavior to neonicotinoid insecticides showed bifurcated patterns (Value significantly lower than 0.5 means high avoidance); highly avoided (AI<1.2) to cyano-neonicotinoid insecticides; Thiacloprid, Acetamiprid while less avoided (AI>2.5) to nitro-neonicotinoids; Imidacloprid, Thiamethoxam and Dinotefuran (Kang and Jung, 2017).
The organophosphate insecticide (Fenitrothion) is extremely toxic (Oral: LD50 0.20 μg/bee, Contact: 0.16 μg/bee in 48 hr) to honeybees (Fenitrothion_specs_eval_ WHO_2010. page 29). In our study it was shown that, Fenitrothion was highly toxic as nitro-group neonicotinoids. In contact test, mortality rate was greater than 70% for 1000 ppm in 1 hour (Oral: LD50 0.116 μg/bee, Contact: 0.092 μg/bee in 48 hr).
Acaricide (Amitraz) is almost an insecticide, and more toxic to honey bees than fungicide and herbicide. The mortality rate of high concentration is greater than 60 %, similar to results of the study by Hillier et al. (2013). Herbicides (Glyphosate), and fungicide (Metconazole) showed low toxicities to honey bees. However, the combination of some fungicides with insecticides has been revealed to be more deadly to the bees than either chemical alone (Iwasa et al., 2004; Johnson et al., 2013). The results imply that the selective use of pesticides could help conservation of pollinators in agricultural production systems.
Acknowledgments
This research was funded by Climate Change and Insect monitoring Research Project, RDA, grant number PJ013463042018, and by the BSRP through the National Research Foundation of Korea (NRF), Ministry of Education, grant number NRF-2018R1A6A1A03024862.
References
-
Bailey, J., C. Scott-Dupree, R. Harris, J. Tolman and B. Harris. 2005. Contact and oral toxicity to honey bees (Apis mellifera) of agents registered for use sweet corn insect control in Ontario, Canada. Apidologie 36: 623-633.
[https://doi.org/10.1051/apido:2005048]

-
Blacquiere, T., G. Smagghe, C. M. Gestel and V. Mommaerts. 2012. Neonicotinoids in bees: a review on concentrations, side-effects and risk assessment. Ecotoxicology 21(4): 973-992.
[https://doi.org/10.1007/s10646-012-0863-x]

-
Brandt, A. and R. Buchler. 2016. The neonicotinoids thiacloprid, imidacloprid, and clothianidin affect the immunocompetence of honey bees (Apis mellifera L.). Journal of Insect Physiology 86: 40-47.
[https://doi.org/10.1016/j.jinsphys.2016.01.001]

-
Cha, E. S., M. Jeong and W. J. Lee. 2014. Agricultural pesticide usage and prioritization in South Korea. Journal of Agromedicine 19(3): 281-293.
[https://doi.org/10.1080/1059924X.2014.917349]

-
Chauzat, M. P., P. Carpentier, A. C. Martel, S. Bougeard, N. Cougoule, P. Porta, J. Lachaize, F. Madec, M. Aubert and J. P. Faucon. 2009. Influence of pesticide on honey bee (Hymenoptera: apidae) colony health in France. Environ. Entomol. 38(3): 514-523.
[https://doi.org/10.1603/022.038.0302]

-
David, A., C. Botias, A. Abdul-Sada, E. Nicholls, E. L. Rotheray, E. H. Hill and D. Goulson. 2016. Widespread contamination of wildflower and bee-collected pollen with complex mixtures of neonicotinoids and fungicides commonly applied to crops. Environment International 88:169-178.
[https://doi.org/10.1016/j.envint.2015.12.011]

- FAO Specifications and Evaluations for Agricultural. Pesticides: Thiamethoxam. 2000.
-
Fairbrother, A., J. Purdy, T. Anderson and R. Fell. 2014. Risks of Neonicotinoid insecticides to honey bees. Environmental Toxicology and Chemistry 33(4): 719-731
[https://doi.org/10.1002/etc.2527]

-
Faucon, J. P., C. Aurieres, P. Drajnudel, L. Mathieu, M. Ribiere, A. C. Martel, S. Zeggane, M. P. Chauzat and M. F. Aubert. 2005. Experimental study on the toxicity of imidacloprid given syrup to honey bee (Apis mellifera) colonies. Pest Management Science 61: 111-125.
[https://doi.org/10.1002/ps.957]

-
Goulson, D. 2013. An overview of the environmental risks posed by neonicotinoid insecticides. Journal of Applied Ecology 50: 977-987.
[https://doi.org/10.1111/1365-2664.12111]

-
Ghosh, S. and C. Jung. 2017. A short review on neonicotinoids; Use in crop protection and issue on honey bee and hive. Journal of Apiculture 32(4): 333-344.
[https://doi.org/10.17519/apiculture.2017.11.32.4.333]

-
Gill, R. J., O. Ramos-Rodriguez and N. E. Raine. 2012. Combined pesticide exposure severely affects individual - and colony - level traits in bees. Nature 491: 105-109.
[https://doi.org/10.1038/nature11585]

-
Hassani, A. K., M. Dacher, V. Gary, M. Lambin, M. Gauthier and C. Armengaud. 2007. Effects of sublethal dose of acetamiprid and thiamethoxam on the behavior of the honey bee (Apis mellifera). Springer 54(4): 653-663.
[https://doi.org/10.1007/s00244-007-9071-8]

-
Henry, M., M. Beguin, F. Requier, O. Rollin, J. Odoux, P. Aupinel, J. Aptel, S. Tchamitchian and A. Decourtye. 2012. A common pesticide decreases foraging success and survival in honey bees. Science 336: 347-350.
[https://doi.org/10.1126/science.1215039]

-
Hillier, N. K. and E. H. Fros. 2013. Fate of dermally applied miticides fluvalinate and amitraz within honey bee (Hymenoptera: Apidae) bodies. Journal of Economic Entomology.
[https://doi.org/10.1603/EC12300]

-
Jonson, R. M., M. D. Ellis, Ch. A. Mullin and M. Frazier. 2010. Pesticides and honey bee toxicity-USA. Apidologie 41: 312-331.
[https://doi.org/10.1051/apido/2010018]

-
Johnson, R. M., W. Mao, H. S. Pollock, G. Niu, M. A. Schuler and M. R. Berenbaum. 2012. Ecologically appropriate xenobiotics induce cytochrome P450 in Apis mellifera. PLoS One 7: e31051.
[https://doi.org/10.1371/journal.pone.0031051]

-
Johnson, R. M. 2015. Honey bee toxicology. Annu. Rev. Entomol. 60: 415-434.
[https://doi.org/10.1146/annurev-ento-011613-162005]

- Kang, M. S. and C. Jung. 2010. Ecotoxicology of several acaricides used in apple orchards to the honey bee, Apis mellifera: from the laboratory to the field study. Kor. J. Apic. 25: 155-161.
-
Kang, M. and C. Jung. 2017. Avoidance behavior of honey bee, Apis mellifera from commonly used fungicides, acaricides and insecticides in apple orchards. Journal of Apiculture 32(4): 295-302.
[https://doi.org/10.17519/apiculture.2017.11.32.4.295]

-
Kim, D. W., W. K. Yun and C. Jung. 2014. Residual toxicity of carbaryl and lime sulfur on the European honey bee, Apis mellifera (Hymenoptera: Apidae) and buff-tailed bumble bee, bombus terrestris (Hymenoptera: Apidae). Kor. J. Apic. 29(4): 341-348.
[https://doi.org/10.17519/apiculture.2014.11.29.4.341]

-
Krupke, Ch. H., G. J. Hunt, B. D. Eitzer, G. Andino and K. Given. 2012. Multiple routes of pesticide exposure for honey bees living near agricultural fields. PloS ONE 7(1): e29268.
[https://doi.org/10.1371/journal.pone.0029268]

- Laurino, D., A. Manino, A. Patetta, M. Ansaldi and M. Porporato. 2010. Acute oral toxicity of neonicotinoids on different honey bee strains. RADIA. XCIII: 99-102.
- Laurino, D., M. Porporato, A. Patetta and A. Manino. 2011. Toxicity of neonicotinoid insecticides to honey bees: laboratory tests. Bulletin of Insectology 64(1): 107-113.
- Laurino, D., A. Manino, A. Patetta and M. Porporato. 2013. Toxicity of neonicotinoid insecticides on different honey bee genotypes. Bulletin of Insectology 66(1): 119-126.
-
Lee, C. Y., S. M. Jeong, C. Jung and M. Burgett. 2016. Acute oral toxicity of neonicotinoid insecticides to four species of honey bee, Apis florea, A. cerena, and A. dorsata. Journal of Apiculture 31(1): 51-58.
[https://doi.org/10.17519/apiculture.2016.04.31.1.51]

-
Mogren, Ch. L. and J. G. Lundgren. 2016. Neonicotinoid-contaminated pollinator strips adjacent to cropland reduce honey bee nutritional status. Scientific reports 6: 29608.
[https://doi.org/10.1038/srep29608]

- Porrini, C., A. G. Sabatini, S. Girotti, F. Fini, L. Monaco, G. Celli, L. Bortolotti and S. Ghini. 2003. The death of honey bees and environmental pollution by pesticides: the honey bees as biological indicators. Bulletin of Insectology 56(1): 147-152.
-
Renaud, M., T. Akeju, T. Natal-da-Luz, S. Leston, J. Rosa, F. Ramos, J. P. Sousa and H. M. V. S. Azevedo-Pereira. 2018. Effects of the neonicotinoids acetamiprid and thiacloprid in their commercial formulations on soil fauna. Chemosphere 194: 85-93.
[https://doi.org/10.1016/j.chemosphere.2017.11.102]

-
Siede, R., L. Faust, M. D. Meixner, Ch. Maus, B. Grunewald and R. Buchler. 2017. Performance of honey bee colonies under a long-lasting dietary exposure to sublethal concentrations of the neonicotinoid insecticide thiacloprid. Pest Management Science 73: 1334-1344.
[https://doi.org/10.1002/ps.4547]

-
Thompson, H. M., S. L. Fryday, S. Harkin and S. Milner. 2013. Potential impacts of synergism in honey bees (Apis mellifera) of exposure to neonicotinoids and sprayed fungicides in crops. Apidologie 45:545-553.
[https://doi.org/10.1007/s13592-014-0273-6]

-
Tison, L., S. Holtz, A. Adeoye, O. Kalkan, N. S. Irmisch, N. Lehmann and R. Menzel. 2017. Effects of sublethal doses of thiacloprid and its formulation Calypso® on the learning and memory performance of honey bees. Journal of Experimental Biology 220: 3695-3705
[https://doi.org/10.1242/jeb.154518]

-
Zhu, W., D. R. Schmehl, Ch. A. Mullin and J. L. Frazier. 2014. Four common pesticides, their mixtures and a formulation solvent in the hive environment have high oral toxicity to honey bee larvae. PloS ONE 9(1): e77547.
[https://doi.org/10.1371/journal.pone.0077547]