Volatiles Analysis of Honey by Gas Chromatography-Mass Spectrometry (GC-MS): Comparison of SPME Volatiles Extraction Methods
Abstract
Volatile organic compounds (VOCs) are the fingerprint of a specific honey and could be used to differentiate honey from different botanical and geographic floral origins. This work was conducted to compare different methods SPME volatile extraction from honey samples of acacia, chestnut, and jujube originated from South Korea. The volatile compositions were analyzed based on dynamic headspace solid-phase microextraction (HS-SPME) using divinylbenzene-carboxen polydimethylsiloxane (DVB/CAR/PDMS) fiber followed by thermal desorption gas chromatography-mass spectrometry (GC-MS). Chestnut honey yielded most diverse volatiles and acacia least. Two methods resulted in distinctively differences in the chemical composition of major volatiles and semi-volatiles components when extracted with and without addition of NaCl showed Jaccard similarities of 0.1~0.2 on three different honey samples. Additionally, volatile profiles were highly separated by the extraction methods used. We found volatile organic compounds (VOCs) analysis of honey without salt addition as a preferred faster method and 2~4g neat sampling would yield consistent volatile profiles from SPME. Such studies of identification of VOCs along with floral origins are important and contribute in the efforts to standardize honey quality and avoid fraudulent labeling of the product.
Keywords:
Acacia, Chestnut, Jujube, SPME, VOCs, 4-Chlorooctane, Floral originINTRODUCTION
Honey is an important product of Apis bees from the nectar of plants and has for long been an excellent nutritional option for many generations due to its therapeutic values (Cuevas-Glory et al., 2007). The chemical composition of honey is mainly dependent on the floral origin of the nectar foraged by Apis mellifera bees (Roberts et al., 2002). Because of high price of certain honey types increment of a quantity of the product by adulteration with low cost and nutritional value substances (Arvanitoyannis et al., 2005; Cotte et al., 2007) or mislabeling regarding the botanical origin sometimes occurs (Alissandrakis et al., 2007). In order to determine the botanical origin of honey, analysis of pollen (melissopalynology), and organoleptic or physicochemical properties are traditionally employed (Radovic et al., 2001). The chemical compounds present in honey can undergo modifications through time and storage conditions, reducing the dependability of those methods based on quantification of physicochemical parameters (Iglesias et al., 2004). Thus, the development of reliable, rapid and cost-effective methodologies for their discrimination and classification based on their botanical origin is of high importance. GC/MS fingerprinting has been proved to be a powerful method for the discrimination between honey samples (Baroni et al., 2006; Senyuva et al., 2009). Monofloral honey is produced from nectar that is derived either wholly or mainly from a single species or plant and therefore is comprised of specific VOCs (Jerković and Marijanović, 2010; Piasenzotto et al., 2003). Accordingly, VOCs could be used to discriminate between monofloral honeys from different floral origins given that the high number of VOCs gives a distinct profile that could represent the fingerprint of each honey type. Several researchers reported method on the honey volatiles fractionated by employing a simultaneous steam distillation-extraction method (Bicchi et al., 1983; Bouseta and Collin, 1995; D’Arcy et al., 1997; Guyot et al., 1998; Guyot et al., 1999; Nickerson and Likens, 1966). Bicchi et al. (1983), with the intention of avoiding sugar interference, proposed a two-step protocol which included preliminary acetone extraction followed by simultaneous Likens-Nikerson steam distillation and solvent extraction. Since heat treatments can lead to artifacts or Millard reaction products, a modified version of the Likens-Nikerson method was adapted by using the vacuum at room temperature (Maignial et al., 1992; Wilkins et al., 1993). Among them, dichloromethane extraction under an inert atmosphere followed by simultaneous steam distillation Dichloromthane extraction appeared to be a useful method for honey aroma characterization (Bouseta and Collin, 1995; Bicchi et al.,1983).
Static headspace (SHS) analysis has not been widely applied to analyze honey volatiles due to low concentrations of volatiles in honey and the low recoveries obtained for semi-volatile compounds (Rowland et al., 1995). Dynamic HS purge-and-trap techniques are more applicable for identification and quantification of a wide range of volatiles and semi-volatiles of honey of different floral and geographical origin, with better sensitivity than that of static headspace (Bouseta et al., 1992).
On the other hand, the SPME-GC/MS method has proved to be rapid, solvent-free, a very reliable technique since high reproducibility and sensitivity for extraction and identification of honey volatile compounds compared with traditional methods (Cuevas-Glory et al., 2007). Moreover, SPME has the advantage of being a simple and a relatively economic extraction technique. SPME fiber coatings, such as CAR/PDMS fiber (75μm), PDMS/DVB (65μm) and DVD/CAR/PDMS (50/30μm), have shown good results with honey volatile extraction. Among five types of fibers such as CAR/DVB, CW/PDMS, PDMS, PA, and DVB/CAR/PDMS compared the semi-polar DVB/CAR/PDMS fiber allowed the best efficiency of extraction for volatile compounds under the same conditions tested (Pontes et al., 2007). The objectives of the present study were to compare honey volatile extraction using SPME with NaCl addition and neat samples methods at different amounts and to determine the optimum method for the extraction. Additionally, we aimed to analysis of volatile compounds of unifloral honey samples in Korean apiculture such as chestnut, jujube and acacia honey using these methods.
MATERIALS AND METHODS
Honey samples collections
Honey samples were collected from collaborative beekeepers associated to Andong National University, Gyoungbuk, Korea. For the study, honey samples were collected from 3 plant origins such as chestnut (Castaneas crenata Siebold & Zucc; family Fagaceae), acacia (Robinia pseudoacacia L.; family Fabaceae), and jujube (Ziziphus jujuba Mill; family Rhamnaceae).
Honey quality factors (Table 1) were analyzed based on the methodologies reported by Jung and Chon (2016) and Jung et al. (2017).
Extraction of volatiles from samples
For sample with NaCl, 3g honey was dissolved in 10ml of saturated sodium chloride (NaCl) solution using a shaker for 10 min, while for the samples without NaCl neat 1~5g samples were used without salt addition. Extraction of honey volatiles with and without salt addition was performed on divinylbenzene-carboxen polydimethylsiloxane (DVB/CAR/PDMS, 50/30μm) on a 1cm StableFlex fibre, recommended for flavors (volatiles and semi-volatiles) based on the method reported by Pontes et al. (2007) and Wolski et al. (2006), respectively with some modification. Before analysis, the fiber was preconditioned in the injection port of a GC. The following SPME condition was used: Honey samples were heated and agitated at agitator temp of 40°C with a pre-incubation time and revolution per minute (rpm) of 1 h and 500 rpm, respectively. Pre-incubation agitator on and off for 3 s each, extraction rpm: 500 rpm, fiber depth from bottom is 10mm, extraction time: 30 min, using a front injector with a desorption time of 5 min. GC cycle time (for preparation a head) was 57 min and bake out time and temperature were 5 min and 250°C, respectively.
Analysis of volatiles
After headspace extraction was completed, the SPME fiber was withdrawn from the vial and promptly introduced into the GC injector under the following conditions. GC/MS analyses were performed using Bruker Scion 455-GC coupled to quadrupole mass Ms TQ detector with combPAL autosampler. The SPME fiber was desorbed in GC injector at 220°C for 5 min in splitless mode and chromatographic separation was carried out on a 30m×0.25mm×0.25μm film thickness BR-5MS (selectivity similar to 5% diphenyl/95% dimethyl polysiloxane) capillary column. Analysis of volatile compounds was carried out in the electron impact ionization mode at 70 eV, and the mass range was m/z 29~550. The GC oven temperature was programmed from 40°C (held for 5 min.) to 250°C at a rate of 5°C. Helium was used as a carrier gas at a constant flow of 0.9ml/min. The interface and source temperatures were 150 and 230°C, respectively. The identi cation of the volatile compounds was performed with mass spectra (NIST libraries and literature data).
Statistical analysis
Volatiles compound profiles were further statistically analyzed for the similarity and consequently subjected to cluster analysis to determine the interrelationship among the extraction methods. For the similarity estimation, Jaccard similarity index was used because of its simplicity and wide acceptance. However, since Jaccard index calculates qualitative aspect only, so we further performed Pearson correlation matrix and presented dendrogram of each extraction method using SPSS program (IBM SPSS statistics, ver 24).
RESULTS AND DISCUSSION
From the quality analysis of experimented honey samples (Table 1), all honey samples showed high-quality standards including moisture content, sugar composition, and carbon isotope ratio values (Jung and Chon, 2016; Yucela et al., 2013; Jung et al., 2017).
Comparison of VOC profiles from two methods
Overall qualitative chemical profiles from different extraction methods by floral sources are represented in Table 2. From chestnut honey, 17 VOCs were extracted from NaCl added sample while 21.8±4.43 VOCs were extracted from neat samples. There were only 4~5 VOCs common from both methods. Jaccard similarity of two methods was very low with a value of 0.1. From acacia honey, 11 VOCs were extracted from NaCl added sample while 9.68±2.19 VOCs were extracted from neat samples. There were only 3~5 common compounds from both methods. Jaccard similarity of two methods was very low with the value of 0.1~0.2. From jujube honey, 16 VOCs were extracted from NaCl added sample while 10.7±4.5 VOCs were extracted from neat samples. There were only 2~4 common compounds from both methods. Jaccard similarity of two methods was very low with a value of 0.1. Also from jujube honey, 1g sampling for SPME would not enough to produce volatiles for the analysis.

Summary of volatile compound analyzed from each honey samples by extraction methods, and number of shared VOCs with NaCl methods (C) followed by Jaccard similarity (Sj)
Pearson correlation matrix showed the quantitative relationships of chemical profiles relative to the extraction methods (Table 3). For all 3 honey samples analyzed, the profiles from NaCl methods were negatively correlated with neat extraction methods regardless of amount subjected (1~5g) (Fig. 1). However, there was a higher correlation among neat samples of 3 and 4g for chestnut and high correlation among all neat samples for acacia, and a slight correlation was found between 2 and 3g samples, and 3 and 4g samples for jujube honey samples.

Pearson correlation matrix of volatile compound profiles from each honey samples by extraction methods
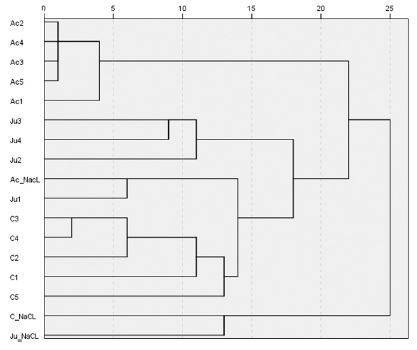
Cluster dendrogram of volatile compound profiles of chestnut (C), acacia (Ac) and jujube (Ju) honey samples by extraction methods of NaCl added (C_NaCl, Ac_NaCl, Ju_NaCl) or neat samples of 1-5g.
From the cluster analysis of the chemical profiles of chestnut, acacia and jujube honey samples relative to the extraction methods, one cluster from 2~5g neat samples from acacia was highly aggregated with similar patterns to the same amount neat samples extracted from chestnut and jujube honey samples. However chemical profiles from NaCl added samples showed a quite different clustering from those of neat samples (Fig. 1). This implies that amount of 2~4g neat sampling would yield consistent volatile profiles from SPME headspace sampling.
Honey volatiles extracted after salt addition
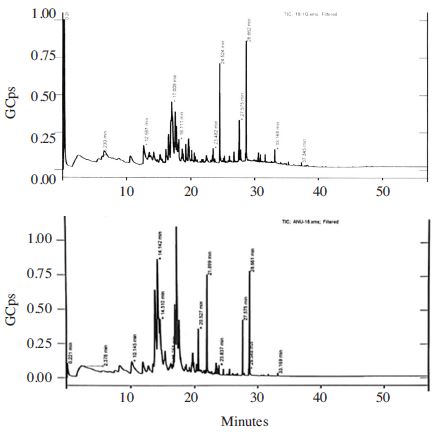
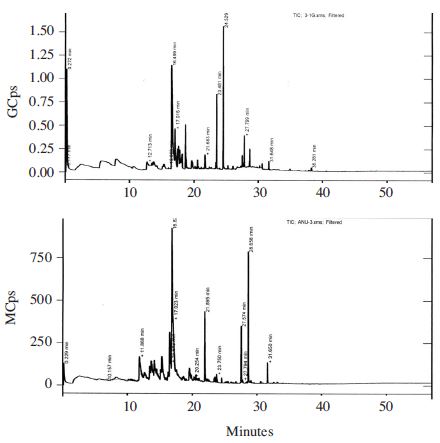
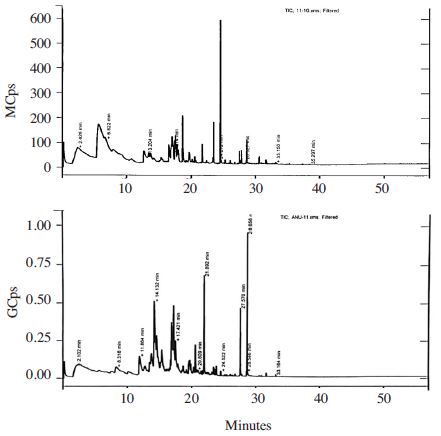
It was reported the addition of salt to a sample greatly increases the extraction efficiency for many analytes, particularly polar, semi-volatiles and volatile compounds (Pawliszyn, 1999). In this study, we used DVB/CAR/PDMS fiber for extraction of volatiles from honey samples. It was reported that semi-polar divinylbenzene/carboxen on polydimethylsiloxane, (DVB/CAR/PDMS) fiber allowed the best efficiency of extraction for volatile compounds compared with other fibers tested such as polydimethylsiloxane (PDMS), polyacrylate (PA), carboxen/Polydimethylsiloxane (CAR/PDMS), and carbowax/divinylbenzene (CW/DVB) (Pontes et al., 2007). The pro les of volatile honey samples we analyzed were complex. The number of GC peaks of volatiles in 3g of chestnut honey, acacia and jujube dissolved in saturated NaCl were 82, 111 and 120, respectively. The identi ed components in honey samples involve different classes of chemical compounds including alcohols, ketones, aldehydes, acids, linear and branched hydrocarbons, terpenes, chloroalkane, benzene, and furan derivatives (Tables 4~6 and Fig. 2~4). Some other compounds introduced by the SPME method were not considered, such as siloxanes (contaminants probably from fiber, septum, and column) (Ceballos et al., 2010).
The major compounds of chestnut honey sample with NaCl added constitute a hydrocarbon decane (11.0%), ketones 2-nonanone (10.9%) and 2-dodecanone (10.0%) as principal compounds (Table 4, Fig. 2). 2-Nonanone and 2-dodecanone were absent in 1-5g neat chestnut honey samples. Guyot et al. (1998) reported acetophenone, phenylethyl alcohol, and 2-aminoacetophenone as major compounds of chestnut honey. Here we found phenylethyl alcohol (4~5.1%) and 4-aminoacetophenone (3.5%) in neat chestnut honey samples. The difference in major compounds of chestnut with those reported might be due to the differences in extraction methods used and geographical origin of the honey.
Acacia honey extracted with SPME after addition of salt was composed of 4-methylbenzaldehyde (22.3%) (Table 5) as a major component which was absent in neat acacia honey samples. Radovic et al. (2001) reported cis-linalool oxide and heptanal as a volatile marker of acacia honey using dynamic headspace method using adsorbed on a Tenax TA trap. However, both compounds were absent in acacia honey we analyzed.
Jujube honey samples dissolved in saturated NaCl exhibited undecane (8.5%), 2-nonanone (7.2%), and m-toluic acid, 6-ethyl-3-octyl ester (5.9%) as major compounds which were absent in the same honey sample without salt addition (Table 6).
Honey volatiles extracted without salt addition
After standing the samples analyzed by addition of NaCl for some days, we observed the formation of some fungus on the samples. The fungus development increases with the number of days of sample standing. Without the addition of salt, volatile and less polar compounds can be extracted with SPME from honey samples. In SPME, the amount of sample used during extraction is very important to provide the highest signal intensity while avoiding saturation effects occurring on the fiber (Robotti et al., 2017). An average number of peaks representing volatiles profiles of without salt addition of chestnut honey, acacia and jujube extracted by the SPME followed by GC-MS analysis were 91, 66 and 124, respectively. The GC-MS analysis result of 1g neat chestnut honey showed 4-chlorooctane (18.1%) and 4-methylbenzaldehyde (13.2%) as major compounds (Table 4, Fig. 2). The percentage composition of 4-methylbenzaldehyde in 5g sample was 4.9% indicating the decrease in the percentage composition of this compound with an increase in the amount of honey. 2,6-Dimethyl-2,7-octadiene-1,6-diol was the third major compound in the chestnut honey. The percentage composition of 2,6-dimethyl-2,7-octadiene-1,6-diol in 1, 2, 3, 4 and 5g honey samples was 6.37, 4.50, 7.64, 7.41 and 6.87%, respectively. This result showed almost constant composition with an increase of the amount of the sample analyzed. Similarly, phenylethyl alcohol showed comparable composition from 1~4g (4.0~5.1%) in the neat chestnut honey sample and its composition was found low (1.5%) in 3g same honey dissolved in saturated NaCl. 4-chlorooctane and 4-methylbenzaldehyde which are major compounds in the neat chestnut honey were absent from 3g sample dissolved in saturated NaCl.
Acacia honey samples 1~5g without salt addition constitutes 22.3~54.63% of α-methyl-α-[4-methyl-3-pentenyl]oxiranemethanol] as a major compound (Table 5). α-Methyl-α-[4-methyl-3-pentenyl]oxiranemethanol] was absent in chestnut honey samples analyzed (Table 4, Fig. 2). Other predominant components of neat 1~4g acacia honey samples were 4-chlorooctane (15.3%), 2,2,6,7-tetramethyl-10-oxatricyclo[4.3.0.1(1,7)]decan-5-one (6.3-14.2%) and 2-furanmethanol-5-ethenyltetrahydro-.al (8.3-12.2%). 2-Furanmethanol-5-ethenyltetrahydro-.al was also found commonly in both chestnut and jujube samples. 2,2,6,7-tetramethyl-10-oxatricyclo[4.3.0.1(1,7)] decan-5-one was also found in jujube honey samples without salt addition and absent form chestnut honey even at a trace level.
Solvent fractions of the methanol extract of acacia honey is composed of major compounds such as trichloromethane, acetoxyethane, hexanaphthene, and acetidin when the sample was dissolved in solvent and analyzed by GC-MS (Paik et al., 2018a). On the other hand, when SPME method was used the main aromatic compounds in the solvent fractions were hydrazomethane, azulene, cyclotrisioxane and hydrazine etc. (Paik et al., 2018a). Based on the published work by Ceballos et al. (2010) we did not consider similar siloxane compounds reported by (Paik et al., 2018a) as constituents of acacia honey we analyzed which might be contaminants from fiber, septum and column. Similarly, solvent fractions of methanol extracts of mandarin honey of Korean origin yielded major organic compounds such as trichloromethane, methylolpropane, cyclopentane and acetoxyethane (Paik et al., 2018b). On other hand, the principal aromatic compounds were acrolein, acetic acid, chloracetonitrile, acrylaldehyde, silanamine, cycloheptatriene and others (Paik et al., 2018b). Paik et al. (2018b) reported the presence of different major VOCs of the same honey sample using different methods consistent with our results.
Neat jujube honey samples (2~5g) contains dimethyl sulfide (5.9~6.67%), 4-methylbenzaldehyde (5.5~8.1%) as principal components (Table 6, Fig. 4). However, dimethyl sulfide was absent and 4-methylbenzaldehyde was detected in a low concentration (1.1%) in 3g jujube honey sample dissolved in saturated NaCl. Dimethyl sulphide was also previously reported as constituent of 14 honey samples (Kaskoniene et al., 2008). From Korean jujube honey, Paik et al. (2018a) reported benzaldeheyde and other aldehydes as major compounds similar to our findings.
Differences and similarly of chemicals from different methods
VOCs common from honey samples of 3 different botanical origins were (E)-decenal, 4-methyl-2-heptanone, benzaldehyde, 2-furanmethanol-5-ethenyltetrahydro-.al, 4-methylbenzaldehyde, 3,7-dimethyl-1,6-octadien-3-ol, phenylethyl alcohol, 2-ethylhexanoic acid, 1,3-bis(1,1-dimethylethyl)benzene, 2,6-bis-1,2,5-cyclohexadiene-1,4-dione and 2,4-bis(1.1-dimethylethyl)phenol.
From chestnut honey, VOC profiles were listed in Table 4. Five most abundant volatiles were 2,6-dimethyl-2,7-octadiene-1,6-diol, benzaldehyde, 4-chlorooctane, phenylethyl alcohol and 2-furanmethanol-5-ethenyltetrahydro-al. Volatiles commonly analyzed from both methods were 2-furanmethanol-5-ethenyltetrahydro-al, 2,6-bis-1,2,5-cyclohexadiene-1,4-dione, 3,7-dimethyl-1,6-octadien-3-ol and (E)-decenal. Unique chemicals found only from chestnut honey were 2,3-butanediol, 3-methylhexanoic acid, hexanoic acid, 2-dodecanone, Oacetyloxim-3-pyridinecarboxaldehyde, 4-pyridinecarbonitrile, benzyl alcohol, (S)-α-methylbenzenemethanol, benzoin, 2-methyl-2-undecanethiol, 2-nonanol, tetrahydro-5,6-dimethyl-2H-pyran-2-one, cyclopentaneundecanoic acid, 1-(ethenyloxy)jcyclohexane, 1-hexyl-3-methylcyclopenatne, tetradecane, dodecane, nonanoic acid and 4-aminoacetophenone.
From acacia honey, VOC profiles were listed in Table 5. Three most abundant volatiles were α-methyl-α-(4-methyl-3-penteneyl) oxiranemethanol, 2-furanmethanol-5-ethenyltetrahydro-al and 2,2,6,7-[tetramethyl-10-oxatricyclo(4.3.0.1.(1,7)]decane-5-one. VOCs common from both methods were α-methyl-α-(4-methyl-3-penteneyl) oxiranemethanol, 2-furanmethanol-5-ethenyltetrahydro-.al, 2,2,6,7-tetramethyl-10-oxatricyclo (4.3.0.1.(1,7)]decane-5-one and 5-methyl-2-heptene and 2,4-bis(1.1-dimethylethyl)phenol. Unique chemicals only from acacia honey were 2,3-dioxodioxime- O,O’-butanenitrile, 2,3-dioxo-, dioxime, O,O’diacetylbutanenitrile, 3-furaldehyde, 3-methylbenzaldehyde, 2-methyl-1-pentadecane, 3,4-diethyl-3-hexanol and exofenchol.
From jujube honey, VOC profiles were listed in Table 6. Five most abundant chemicals were dimethyl sulfide, 4-methylbenzaldehyde, 3,7-dimethyl-1,6-octadien-3-ol, benzaldehyde and toluene. VOCs common from both methods were 4-methylbenzaldehyde, 3,7-dimethyl-1,6-octadien-3-ol, 3.5-dimethyl-1-hexene, decane, Odecylhydoxylamine and 2,6-bis-1,2,5-cyclohexadiene-1,4-dione. Unique chemicals only from jujube honey were (Z)-octene, 3.5-dimethyl-1-hexene, dimethyl sulfide, 3-trifluoroacetoxypentadecane, 4,5-dimethyl-2-heptanone, 3,5-dimethyloctane, m-toluic acid, 6-ethyl-3-octyl ester, 2-O-benzyl-D-arabinose, 2,7-dimethyl-2,6-octadien-1-ol and O-decylhydoxylamine.
CONCLUSIONS
Honey is a very complex product and has various properties and composition depending not only on the nectar-providing plant species but also on bee species, geographic area, season and storage system. However, honey from different floral sources may have different aromas and tastes based on its volatile composition which in turn is dependent on the extraction methods, botanical and geographical sources. Here we used SPME fiber coatings, DVB/CAR/PDMS, 50/30μmwhich was reported best for honey volatile extraction (Pontes et al., 2007). We found there were differences in the composition of volatile major constituents with and without the addition of salt. Even though the number of samples is small, the results of analysis of HS-SPME honey samples (2~4g) without salt addition showed a good profile for further work. It is hard to suggest the floral marker of each sample at this point, as this study was based on a small number of honey samples. We believe the floral markers can be identified and improved by increasing sample numbers from each botanical origin.
Acknowledgments
This work was supported by Priority Research Centers Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science, and Technology (NRF-2018R1A6A1A03024862). Aman Dekebo thanks Adama Science and Technology University for allowing extension of his sabbatical leave to conduct this research.
References
-
Alissandrakis, E., P. A. Tarantilis, P. C. Harizanis, and M. Polissiou, (2007), Comparison of the volatile composition in thyme honeys from several origins in Greece, J. Agric. Food Chem, 55, p8152-8157.
[https://doi.org/10.1021/jf071442y]

-
Arvanitoyannis, I. S., C. Chalhoub, P. Gotsiou, N. Lydakis-Simantiris, and P. Kefalas, (2005), Novel quality control methods in conjunction with chemometrics (multivariate analysis) for detecting honey authenticity, Crit. Rev. Food Sci. Nutr, 45, p193-203.
[https://doi.org/10.1080/10408690590956369]

-
Baroni, M. V., M. L. Nores, M. P. Diaz, G. A. Chiabrando, J. P. Fassano, C. Costa, and D. A. Wunderlin, (2006), Determination of volatile organic compound patterns characteristic of five unifloral honey by solid-phase microextraction-gas chromatography-mass spectrometry coupled to chemometrics, J Agric. Food Chem, 54, p7235-7241.
[https://doi.org/10.1021/jf061080e]

-
Bicchi, C., F. Belliardo, and C. Fratinni, (1983), Identification of the volatile components of some piedmontese honeys, J. Apic. Res, 22(2), p130-136.
[https://doi.org/10.1080/00218839.1983.11100574]

-
Bouseta, A., S. Collin, and J-.P. Doufour, (1992), Characteristic aroma profiles of unifloral honeys obtained with a dynamic headspace GC-MS system, J. Apic. Res, 31(2), p96-109.
[https://doi.org/10.1080/00218839.1992.11101268]

-
Bouseta, A., and S. Collin, (1995), Optimized Likens-Nickerson methodology for quantifying honey flavors, J. Agric. Food Chem, 43(7), p1890-1897.
[https://doi.org/10.1021/jf00055a025]

-
Ceballos, L. A., J. A. Pino, E. Clara, C. E. Quijano-Celis, and A. Dago, (2010), Optimization of a HS-SPME/GC-MS method for determination of volatile compounds in some Cuban unifloral honeys, J. Food Qual, 33, p507-528.
[https://doi.org/10.1111/j.1745-4557.2010.00330.x]

-
Cotte, J. F., H. Casabianca, J. Lheritier, C. Perrucchietti, C. Sanglar, H. Waton, and M. F. Grenier-Loustalot, (2007), Study and validity of 13C stable carbon isotopic ratio analysis by mass spectrometry and 2H site-specific natural isotopic fractionation by nuclear magnetic resonance isotopic measurements to characterize and control the authenticity of honey, Analytica Chimica Acta, 582, p125-136.
[https://doi.org/10.1016/j.aca.2006.08.039]

-
Cuevas-Glory, L. F., J. A. Pino, L. S. Santiago, and E. Sauri-Duch, (2007), A review of volatile analytical methods for determining the botanical origin of honey, Food Chem, 103, p1032-1043.
[https://doi.org/10.1016/j.foodchem.2006.07.068]

- D’Arcy, B. R., G. B. Rintoul, C. Y. Rowland, and A. J. Blackman, (1997), Composition of Australian honey extractives. 1. Norisoprenoids, monoterpenes, and other natural volatiles from blue gum (Eucalyptus leucoxylon) and yellow box (Eucalyptus melliodora) honeys, J Agric. Food Chem, 45(5), p1834-1843.
-
Guyot, C., V. Scheirman, and S. Collin, (1999), Floral origin markers of heather honeys: Calluna vulgaris and Erica arborea, Food Chem, 64(1), p3-11.
[https://doi.org/10.1016/s0308-8146(98)00122-8]

-
Guyot, C., A. Bouseta, V. Scheirman, and S. Collin, (1998), Floral origin markers of chestnut and lime tree honeys, J. Agric. Food Chem, 46(2), p625-633.
[https://doi.org/10.1021/jf970510l]

-
Iglesias, M. T., C. De Lorenzo, M. Polo, P. J. Martln-Alvarez, and E. Pueyo, (2004), Usefulness of amino acid composition to discriminate between honeydew and floral honeys. Application to honeys from a small geographic area, J. Agric. Food Chem, 52(1), p84-89.
[https://doi.org/10.1021/jf030454q]

- Jerković, I., and Z. Marijanović, (2010), Volatile composition screening of Salix spp. Nectar honey. Benzenecarboxylic acids, norisoprenoids, terpenes and others, Chem. Biodivers, 7, p2309-2325.
-
Jung, C., and J.-W. Chon, (2016), Quality assessment of honey from different floral origin in Korea, J. Apic, 31(2), p103-111.
[https://doi.org/10.17519/apiculture.2016.06.31.2.103]

-
Jung, C., E. Cho, S. Lee, and J.-W. Chon, (2017), Quality characteristics of honey on the market: Case study from Daegu-GyenongBuk provinces, J. Apic, 32(1), p51-58.
[https://doi.org/10.17519/apiculture.2017.04.32.1.51]

- Kaskoniene, V., P. R. Venskutonis, and V. Cksteryte, (2008), Composition of volatile compounds of honey of various floral origin and beebread collected in Lithuania, Food Chem, 111, p988-997.
-
Maignial, L., P. Pibarot, G. Bonetti, A. Chaintreau, and J. P. Marion, (1992), Simultaneous distillation-extraction under static vacuum: isolation of volatile compounds at room temperature, J. Chromatogr, 606, p87-94.
[https://doi.org/10.1016/0021-9673(92)85260-z]

- Nickerson, G. B., and S. T. Likens, (1966), Gas chromatographic evidence for the occurrence oil components in beer, J. Chromatogr, 21, p1-5.
-
Paik, W. K., A. K. Kwak, K. H. Kim, M. L. Lee, Y. S. Choi, and H. K. Kim, (2018a), Studies on the organic compounds and mineral constituents of Acanthopanax (Acanthopanax senticosus) and Acacia (Robinia pseudo-acacia) honey produced in Korea, J. Apic, 33(1), p43-53.
[https://doi.org/10.17519/apiculture.2018.04.33.1.43]

-
Paik, W.K., A. K. Kwak, K. H. Kim, M. L. Lee, Y. S. Choi, and H. K. Kim, (2018b), Studies on the proximate components, organic compounds, mineral constituents and vitamin C of Mandarin (Citrus unshiu) honey produced in Korea, J. Apic, 33(1), p33-42.
[https://doi.org/10.17519/apiculture.2018.04.33.1.33]

- Pawliszyn, J., (1999), Applications of solid-phase microextraction, Royal Society of Chemistry, Cambridge, UK.
- Piasenzotto, L., L. Gracco, and L. Conte, (2003), Conte, Solid phase micro extraction (SPME) applied to honey quality control, J. Sci. Food Agric, 83, p1037-1044.
-
Pontes, M., J.C. Marques, and J.S. Camara, (2007), Screening of volatile composition from Portuguese multifloral honeys using headspace solid-phase microextraction-gas chromatography-quadrupole mass spectrometry, Talanta, 74, p91-103.
[https://doi.org/10.1016/j.talanta.2007.05.037]

-
Radovic, B. S., M. Careri, A. Mangia, M. Musci, M. Gerboles, and E. Anklam, (2001), Contribution of dynamic headspace GC-MS analysis of aroma compounds to authenticity testing of honey, Food Chem, 72(4), p511-520.
[https://doi.org/10.1016/s0308-8146(00)00263-6]

- Roberts, T. A., P. Aureli, C. Flamini, and M. Yndestad, (2002), Honey and microbiological hazards, In Proceedings of the scientific committee on veterinary measures relating to public health.
-
Robotti, E., F. Campo, M. Riviello, M. Bobba, M. Manfredi, E. Mazzucco, F. Gosetti, G. Calabrese, E. Sangiorgi, and E. Marengo, (2017), Optimization of the extraction of the volatile fraction from honey samples by SPME-GC-MS, Experimental design, and multivariate target functions, J. Chem, p1-14.
[https://doi.org/10.1155/2017/6437857]

-
Rowland, C. Y., A. J. Blackman, B. R. D’Arcy, and G. B. Rintoul, (1995), Comparison of organic extractives found in leatherwoos (Eucryphia lucida) honey and leatherwood flowers and leaves, J. Agric. Food Chem, 43(3), p753-763.
[https://doi.org/10.1021/jf00051a036]

-
Senyuva, H. Z., J. Gilbert, S. Silici, A. Charlton, C. Dal, N. Curel, and D. Cimen, (2009), Profiling Turkish honeys to determine authenticity using physical and chemical characteristics, J. Agric. Food Chem, 57, p3911-3919.
[https://doi.org/10.1021/jf900039s]

- Wilkins, A. L., Y. Lu, and S. T. Tan, (1993), Extractives from New Zealand honeys. 4. Linalool derivatives and others components from nodding thistle (Cardans nutans) honey, J. Agric. Food Chem, 41(6), p873-878.
- Wolski, T., K. Tambor, H. Rybak-Chmielewska, B. Kedzia, (2006), Identification of honey volatile components by solid phase microextraction (SPME) and Gas Chromatography/Mass Spectrometry (GC/MS), J. Apic. Sci, 50(2), p115-125.
- Yucela, P. K., H. Guclua, Y. Merta, O. Oktara, T. Koseoglua, and B. Yllmaz, (2013), Measurement of carbon isotope ratios of honey samples from turkey by EA-IRMS, https://www.apimondia.com/congresses/2013/Economy/Plenary-Session/Measurement%20Of%20Carbon%20Isotope%20Ratios%20Of%20Honey%20Samples%20From%20Turkey%20-%20Hulya%20Gu%C3%A7lu.pdf (Accessed on June 11, 2018).