Effects of Weather Conditions on Nectar Secretion and Honeybee Foraging Activity on Tilia cordata, and an Assessment of Its Melliferous Potential
Abstract
The expansion of pollinator populations, particularly honeybees, is closely tied to the consistent availability of nectar resources. Yet, agricultural intensification has led to a decline in both the diversity and quality of nectar-producing flora. To promote biodiversity and ensure the long-term viability of beekeeping, it is essential to identify nectar tree species and assess their value through analyses of nectar secretion and other relevant characteristics. This study focused on Tilia cordata Mill. in Uzbekistan, analyzing the diurnal dynamics of nectar secretion alongside honeybee foraging behavior. These patterns were evaluated in the context of key environmental variables such as temperature, relative humidity, and light intensity. In addition, the research explored the degree of pollination dependency of T. cordata for seed development and estimated its potential honey production per unit area to assess its effectiveness as a melliferous species. On average, individual flowers secreted 0.52 μL of nectar per day, with a mean sugar concentration of 60.43%. The nectar secretion measured at 2-hour intervals from 10:00 to 16:00 showed that nectar output reached its peak at 10:00 AM and gradually decreased as the day progressed. The highest level of honeybee visitation occurred at noon, with an average of 80.93 bees, while the lowest was recorded at 4:00 PM with 62.74 bees. Nectar volume exhibited a negative correlation with temperature, but positive correlations with humidity and light intensity. In contrast, bee visitation did not show a significant relationship with these weather parameters. The pollination dependency rate for T. cordata was calculated at 81%, reflecting a strong reliance on insect-mediated pollination. The estimated honey yield was approximately 276.4±42.5 L per hectare. These results indicate that T. cordata holds significant promise for use in nectar forestry, offering a reliable nectar source and supporting high-quality honey production.
Keywords:
Tilia cordata, Apis mellifera, Nectar, Climate, PollinatorINTRODUCTION
Pollinators have co-evolved with flowering plants for approximately 120 million years, forming mutualistic relationships where pollination services are exchanged for floral rewards such as nectar and pollen (Engel, 2000; Poinar and Danforth, 2006; Irwin et al., 2010; Bascompte, 2019). Today, these interactions remain fundamental to terrestrial ecosystems. Pollinators are responsible for the reproduction of nearly 87% of all flowering plant species and contribute to the pollination of about 75% of the world’s most significant food crops (Klein et al., 2007; Ollerton et al., 2011; Potts et al., 2016). Their role is thus vital for sustaining biodiversity, maintaining ecosystem resilience, and ensuring food security. Among pollinators, honeybees (Apis mellifera) are the most economically important in monoculture crop systems, where their absence can result in yield losses exceeding 90% for certain crops, such as fruits and nuts (McGregor, 1976; Southwick and Southwick, 1992; Watanabe, 1994).
The decline of pollinating insects has far-reaching ecological and socio-economic consequences. A reduction in pollinator populations threatens the stability of ecosystems, compromises food production systems, and may even contribute to local poverty in rural regions dependent on agriculture (Memmott et al., 2007; Christmann, 2019; Rahimi and Jung, 2024). One of the principal drivers of pollinator decline is the loss of floral diversity and abundance, largely due to agricultural intensification (Tilman et al., 2001). A decrease in the diversity of melliferous plant species may lead to a decline in the diversity and abundance of pollinators (Baude et al., 2016; Reilly et al., 2020). In Uzbekistan, recent land use changes-such as the expansion of cotton, alfalfa, forage legumes, melons, and fruit trees-have significantly narrowed the diversity and temporal distribution of nectar-producing plants (Ylejbees.com, n.d.). At the same time, the Uzbek beekeeping industry has shown a growing trend, with honey production increasing from 3,172 tons in 2011 to 13,737 tons in 2019 (Djurabaev and Rashidov, 2021). This growing demand for apicultural resources underscores the importance of ensuring stable and diverse nectar sources.
Given these trends, it is critical to identify nectar-producing tree species that are not only adapted to local environmental conditions but also capable of supporting pollinator populations with stable nectar supplies throughout the flowering season. Integrating such species into the landscape can promote pollinator biodiversity and enhance crop yields, as several studies have demonstrated the positive effects of nectar-rich plantings near agricultural fields (Garibaldi et al., 2014; Son and Jung, 2021). The introduction of well-adapted nectar source trees offers a sustainable pathway for bolstering both ecological health and beekeeping productivity.
One promising candidate is the small-leaved linden (Tilia cordata Mill.), a member of the Malvaceae family, widely distributed across temperate regions of Europe, Western Asia, and parts of North America and Asia (GBIF, n.d.). Typically growing between 20 and 40 meters in height, T. cordata is commonly cultivated as an ornamental tree in parks and along urban streets. Its leaves are predominantly glabrous and alternate in arrangement, while its flowers bloom in clusters of 5-11 cream-yellow blossoms, each measuring approximately 13-15 mm in diameter. Flowering occurs from late May to early June, typically lasting about two weeks (Pawlikowski, 2010; Sultanova et al., 2022). The flowers produce a strong fragrance that attracts numerous pollinators, especially honeybees, which account for an estimated 89-91% of floral visitors, underscoring their pivotal role in the species̓ reproductive ecology (Pawlikowski, 2010).
Beyond its ecological significance, T. cordata also holds a substantial economic value. It is used for timber and bark, but most notably, it serves as a major nectar source for honeybees, thereby supporting high honey yields (Kim et al., 2022; Sultanova et al., 2022). As a forest nectar species, T. cordata has strong potential to contribute to the sustainable development of apiculture, particularly due to its resilience. The species exhibits high tolerance to drought and low temperatures and thrives under a variety of soil and climatic conditions. This makes it well-suited for inclusion in mixed-species forests and reforestation programs, particularly in the context of climate adaptation (De Jaegere et al., 2016; Falk et al., 2016; Rumiantsev et al., 2021). In addition, T. cordata has demonstrated consistent flowering behavior under the warm climatic conditions of Uzbekistan, suggesting its viability as a landscape or urban greening species in the region (Jumayev et al., 2021).
Despite some information on the blooming periods of key nectar-producing plants in Uzbekistan (Jumayev et al., 2021), there remains a significant lack of data on important floral traits. Specifically, detailed information on nectar volume per flower, floral density, diurnal nectar secretion patterns, pollination dependency, and correlations with meteorological variables is limited. Without this information, it is difficult to reliably assess the potential of these species to function as consistent nectar sources for pollinators throughout the season.
To address these knowledge gaps, the present study evaluates the potential of T. cordata as a nectar source species under the environmental conditions of Uzbekistan. The specific objectives are to: (1) investigate the diurnal pattern of nectar secretion during peak flowering seasons; (2) analyze how major weather variables temperature, humidity, and light intensity-influence nectar production; and (3) estimate the species̓ potential honey yield per hectare and assess its dependency on insect pollination. By fulfilling these goals, the study aims to provide foundational data to support the strategic introduction of T. cordata in nectar forestry efforts, with the broader objective of enhancing pollinator resources and advancing sustainable apiculture in the region.
MATERIALS AND METHODS
1. Study site and plant materials
The study was carried out in Tashkent, Uzbekistan (14°21ʹ42ʺN, 69°20ʹ34ʺE), at the experimental apiary of Tashkent State Agrarian University. The focal trees were Tilia cordata specimens planted as ornamental landscape trees in the vicinity of the apiary. These trees, approximately 10 years old, had an average height of 5.91 meters, an average diameter at breast height (DBH) of 32.67 centimeters, and an average crown width of 3.72 meters, with all measurements taken using a measuring tape.
To estimate the total number of flowers per tree, measurements were taken for three components: the average number of flowers per inflorescence, the number of inflorescences per branch, and the total number of branches per tree (Table 1). Three well-maintained T. cordata individuals were selected as representative specimens for detailed observation (Fig. 1). All measurements related to nectar secretion, honeybee visitation rates, and pollination dependency were conducted on these same trees during their synchronized flowering period.
Growth characteristics of Tilia cordata trees used for field observations, including tree age, height, diameter at breast height (DBH), and crown width
2. Nectar secretion pattern
The investigation of nectar secretion patterns in Tilia cordata was conducted during the species’ peak flowering period, from June 3 to June 8, 2024, following the same daily schedule as the honeybee visitation survey. The observations were conducted during the same period and at the same time points as the environmental data collection. Nectar was sampled from a total of 30 flowers, with 10 flowers selected per tree from three individual trees.
To prevent nectar removal by honeybees or other floral visitors before sampling, unopened flower buds were enclosed using pollinator-exclusion mesh bags (13 cm×18 cm) before anthesis. Each mesh bag contained at least five buds. Once anthesis occurred, the mesh was removed and nectar was extracted directly from the nectary using 1 μL and 2 μL capillary tubes. Following each sampling session, the exclusion bags were reattached to prevent further insect visitation.
Given the delicate floral morphology of T. cordata, individual flowers are frequently abscised after a full day of sampling. As some flowers open at different times within the same day, resulting in variations in nectar accumulation, newly opened flowers at similar opening times were selected each day to minimize such variation and ensure accurate nectar measurements. Total nectar volume was calculated based on the length of nectar-filled within the capillary tubes, using a method consistent with that of Son et al. (2023). For dry nectar volume, samples were placed in a drying oven at 40°C until constant weight was achieved, allowing estimation of the dry nectar content from the residual volume.
3. Honeybee foraging pattern
Observations of honeybee (Apis mellifera) foraging behavior on Tilia cordata were conducted over six days during during the same period and at the same time points as the environmental data collection. During each observation session, the number of honeybees visiting flowers was recorded by visually counting individuals as they flew to the tree.
Visual observations were used to monitor honeybee foraging activity, but this method may underestimate visitation due to observer bias and the fast movement of individuals (Westphal et al., 2008). To minimize such bias, the tree canopy was divided into four sections corresponding to the cardinal directions (north, south, east, and west). Each section was observed for 15 seconds, resulting in a total of one minute of observation time per tree during each time point. Following data collection, the relationship between honeybee visitation rates and environmental variables was analyzed to assess the influence of weather conditions on foraging activity.
4. Environmental data collection
To monitor environmental conditions, HOBO data loggers were installed in proximity to the three study trees to continuously record air temperature (°C) and relative humidity (%). These climatic parameters were subsequently analyzed for nectar secretion patterns. In addition, light intensity (illuminance) was measured manually at 10:00, 12:00, 14:00, and 16:00 each day in an open, unobstructed flat area adjacent to each tree. Care was taken to ensure that measurements were not affected by shading from nearby vegetation or structures.
During the observation period from June 3 to June 8, the average air temperature was 25.7°C, ranging from a minimum of 10.8°C to a maximum of 39.1°C. The lowest temperatures were typically recorded around 5:00 a.m., while the highest temperatures occurred near 1:00 p.m. Relative humidity averaged 42.8%, with a minimum of 19.8% and a maximum of 63.8%. Humidity levels generally peaked around 6:00 a.m. and reached their lowest values around 2:00 p.m. Regarding light intensity, the lowest readings were recorded at 16:00, whereas the highest was observed at noon. No rainfall was recorded during the study period. Weather conditions remained mostly clear, with minimal cloud cover except for slight overcast at 12:00 and 14:00 on June 3 (Table 2).
5. Estimation of honey yield and pollination dependency
The potential honey yield was estimated based on the average daily dry nectar volume per flower. This value was used to calculate the expected honey production per hectare by incorporating several factors: the average number of flowers per tree, the mean duration of the flowering period, the assumed moisture content of mature honey, and a planting density of 625 trees per hectare (Kim et al., 2021; Kim et al., 2022; Park et al., 2025). The calculation was carried out using Equation 1.
| (Eq. 1) |
Where N is the average daily dry nectar volume secreted per flower (L), F is the estimated total in this calculation, F represents the number of flowers per tree, D is the average duration of the flowering period (in days), T is the tree density per hectare (trees/ha), and M denotes the moisture content of honey, expressed as a proportion (e.g., 0.20 for 20%).
The standard deviation of the estimated honey yield (ΔH) was determined using error propagation principles (Equation 2), taking into account the relative uncertainties of each contributing variable. In this context, ΔN, ΔF, ΔD, and ΔT correspond to the standard deviations of daily nectar volume per flower (N), number of flowers per tree (F), flowering duration (D), and tree density per hectare (T), respectively.
| (Eq. 2) |
To assess pollination dependency (PD), mesh exclusion bags (40 cm×60 cm) were installed over flower clusters before anthesis to prevent insect-mediated pollination, including visits by honeybees. Each bag enclosed approximately 50-80 flowers. The experiment was conducted on a single tree, with five bagged treatments (exclusion plots) and five unbagged controls (open plots). Following the completion of flowering, the exclusion bags were removed, and fruit set rates were recorded for both treatments.
Pollination dependency was calculated using Equation 3, following the method described by Jung (2008) and Jung and Shin (2022).
| (Eq. 3) |
where YO is the yield (i.e., fruit set) from open-pollinated flowers, and YC is the yield from pollinator-excluded flowers.
6. Statistical analysis
All statistical analyses were performed using R version 4.3.2. To compare nectar volume across different time points, the Shapiro-Wilk test was initially applied to assess the normality of the data. As normality assumptions were not met for nectar volume at any time point, the non-parametric Kruskal-Wallis test was employed. Upon detecting significant differences, Dunn’s post hoc test with Bonferroni correction was used to conduct pairwise comparisons between time points.
Differences in honeybee visitation frequency among periods were analyzed using a one-way analysis of variance (ANOVA). When significant effects were observed, Tukey’s Honest Significant Difference (HSD) test was applied for multiple pairwise comparisons.
To evaluate the influence of environmental variables on nectar secretion and honeybee visitation, normality was again tested with the Shapiro-Wilk test. Given that normality was violated, Spearman’s rank correlation, a non-parametric method, was used to examine relationships.
For the pollination dependency analysis, fruit set rates between the pollinator exclusion treatment and control groups were compared. The normality of the data was verified with the Shapiro-Wilk test and the homogeneity of variances was assessed using Bartlett’s test. Based on these results, an independent samples t-test was conducted to evaluate significant differences in mean fruit set rates between groups.
RESULTS
1. Nectar secretion pattern
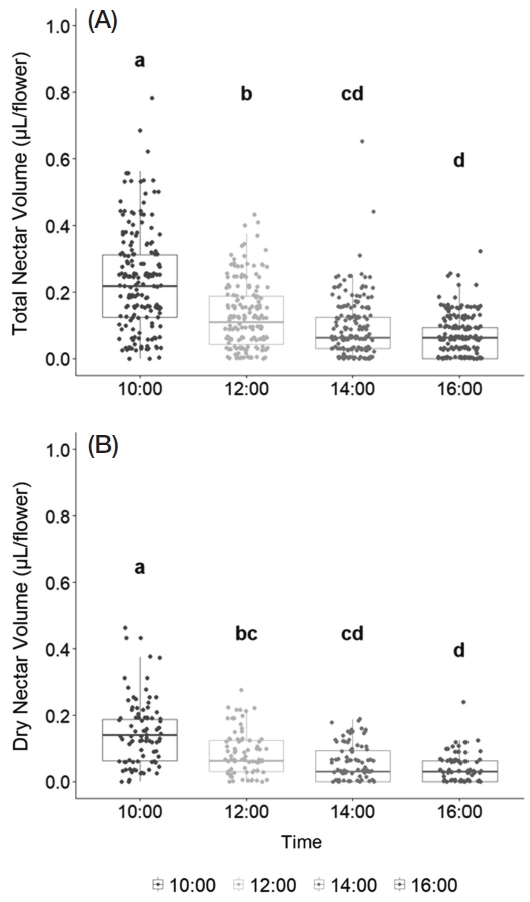
Over the six days of observation, the average total nectar volume secreted per flower was 0.52 μL, while the average dry nectar volume measured 0.31 μL. From these values, the moisture content of nectar per flower was estimated to be approximately 39.6% on average (Table 3).
On average, the highest nectar volume per flower was recorded at 10:00 AM (0.24±0.15 μL), while the lowest volume was observed at 4:00 PM (0.06±0.06 μL). A similar pattern was noted for dry nectar volume, which also peaked at 10:00 AM (0.15±0.09 μL). Nectar secretion per flower exhibited statistically significant variation depending on the time of day (Fig. 2).
2. Honeybee foraging density
The frequency of honeybee visitation did not differ significantly across all observation times overall; however, a statistically significant difference was detected specifically between 12:00 and 16:00 (p<0.05). The highest visitation rate occurred at noon, with an average of 80.93 bees, which significantly declined to 62.74 bees by 16:00 (Fig. 3).
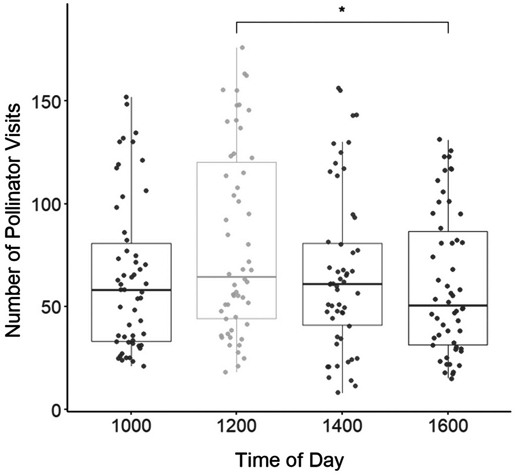
Honey bee visitation frequency per tree at different times of the day. Boxplots represent the distribution of the number of visits recorded at 1000, 1200, 1400, and 1600 hours. Each dot indicates an individual tree. One-way ANOVA revealed a significant difference among periods (p=0.048), and Tukey̓s HSD post-hoc test showed a statistically significant difference between 1200 and 1600 hours (p=0.043).
3. Correlation with climatic factors
Spearman’s rank correlation analysis between climatic factors and nectar secretion revealed a weak negative correlation with temperature, which was not statistically significant. Relative humidity showed a significant positive correlation with nectar volume (p<0.05), while light intensity demonstrated a moderate but non-significant positive correlation (Fig. 4). Similarly, correlations between honeybee visitation frequency and climatic variables-temperature, humidity, and light intensity-were all non-significant (p>0.05) (Table 4).
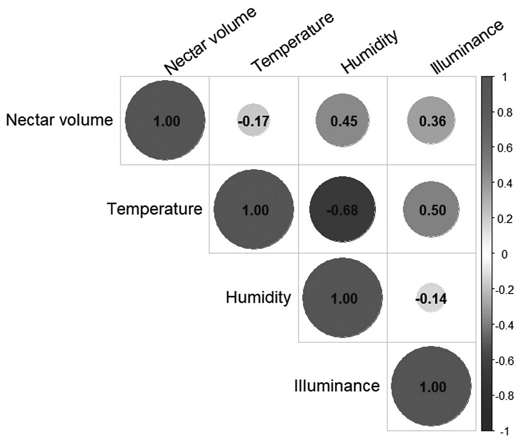
Spearman̓s rank correlation matrix among nectar volume and climatic variables. Circle size and color indicate the strength and direction of correlation. Red and blue represent positive and negative correlations, respectively. Values indicate correlation coefficients.
4. Estimation of honey yield and pollination dependency
The average dry nectar volume produced per flower in Tilia cordata was 0.32±0.01 μL. The total number of flowers per 10-year-old tree was estimated at approximately 78,984±3,718. Considering an average flowering period of 14±2 days, the total dry nectar production per tree was calculated to be about 353.8±54.4 mL. Assuming a moisture content of 20% in honey, the corresponding honey yield per tree was estimated at 442.3±67.9 mL. Using a planting density of 625 trees per hectare-based on an average diameter at breast height (DBH) of 32.67 cm, crown width of 3.72 m, and 4×4 m spacing (FAO, 1995; USDA NRCS, 2010; Kim et al., 2021)-the potential honey yield per hectare was estimated at approximately 276.4±42.5 L/ha.
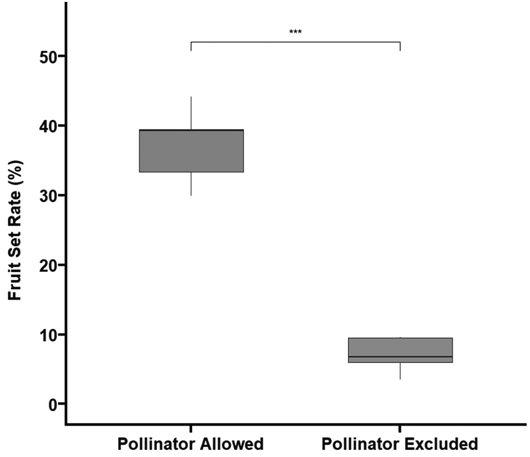
Comparison of fruit set rates between pollinator-access and pollinator-exclusion treatments revealed that open (pollinator-access) trees had a mean fruit set of 37.22%, while exclusion treatments showed a significantly lower fruit set of 7.08% (t-test, p<0.001) (Fig. 5). Based on these results, the pollination dependency (PD) of T. cordata was calculated as 80.98%.
DISCUSSION
This study investigated the variation in nectar secretion of Tilia cordata in relation to key environmental factors including temperature, relative humidity, and light intensity. The analysis indicated a negative correlation between temperature and nectar production, whereas both relative humidity and light intensity showed positive associations with increased nectar secretion. These findings align with patterns reported in earlier research.
For instance, McCombs et al. (2022) observed in two native species from the U.S. montane grasslands-Balsamorhiza sagittata and Eriogonum umbellatum-that nectar volume decreased while sugar concentration increased as temperatures rose. Additionally, a negative relationship between temperature and nectar secretion has been consistently documented across diverse geographic regions and plant species (Mu et al., 2015; Eremia et al., 2017; Takkis et al., 2018).
Regarding humidity, elevated relative humidity can reduce nectar evaporation, thereby enhancing nectar volume. Conversely, low humidity conditions may accelerate evaporation, resulting in higher sugar concentrations (Bertsch, 1983; Pacini et al., 2003). Similar to our results, many studies have reported an increase in nectar secretion with rising relative humidity levels (Chabert et al., 2020; Bareke et al., 2021).
The influence of light intensity on nectar secretion is largely mediated by photosynthetic activity. Photosynthesis generates sucrose, which is transported through the phloem to nectaries for secretion as nectar. Consequently, photosynthetic capacity directly affects nectar production (Pacini et al., 2003; Cawoy et al., 2008). The positive correlation observed between light intensity and nectar secretion in this study is consistent with these physiological mechanisms.
The average daily nectar volume per flower of Tilia cordata was measured at 0.52 μL, markedly lower than values reported from Poland (5.81 μL; Dmitruk et al., 2024) and Belgium (1.5 μL; Jacquemart et al., 2018). These discrepancies likely reflect the hotter and drier climate conditions of Uzbekistan, where this study was conducted.
Based on estimated honey yield, T. cordata nectar exhibited a relatively high average sugar concentration of 60.4%. Taking into account the daily nectar volume per flower and the full flowering period, the potential honey production per hectare was estimated at approximately 276.4±42.5 L/ha. These results indicate that T. cordata possesses strong melliferous properties even under the relatively arid and warm environmental conditions of Uzbekistan.
Previous research has reported a wide range of honey production potential among major nectar plants, from 14 to 829 kg/ha (Adgaba et al., 2017; Na et al., 2024). The estimate for T. cordata in this study falls near the median of this range, underscoring its potential as a reliable and stable nectar source. In particular, the combination of high sugar content and abundant nectar secretion suggests that T. cordata can provide a consistent and valuable food resource for honeybees and other essential pollinators.
Honeybee visitation was consistently observed across all time periods and intervals. Moreover, the pollination dependency rate was approximately 81%, demonstrating that successful seed production in T. cordata strongly depends on pollinators, especially honeybees. This underscores a close ecological interdependence between T. cordata and its pollinators.
In conclusion, T. cordata exhibits sufficient nectar secretion capacity in hot and arid climates and shows moderate to high honey production potential combined with strong reliance on pollinators. These characteristics affirm the species’ role in a mutually dependent ecological relationship with honeybees. The findings highlight the significant potential of T. cordata as a melliferous species in Uzbekistan. This study provides a foundational reference for future afforestation efforts involving nectar plants and is expected to contribute to broader objectives including ecosystem stability, pollinator conservation, improved crop productivity, and the sustainable development of the beekeeping industry.
CONCLUSION
This study aimed to assess the melliferous potential of Tilia cordata and its ecological reliance on pollinators by examining nectar secretion patterns under varying climatic conditions, estimating honey yield, and evaluating pollination dependence. The results indicated that nectar secretion tended to decline with increasing temperature, although this relationship was not statistically significant. Conversely, both relative humidity and light intensity were positively correlated with nectar volume, with higher values promoting increased secretion.
The average daily nectar volume per flower was measured at 0.52 μL, which is lower than previously reported figures from Poland and Belgium. This reduction is likely a physiological adaptation to the hot and arid climate characteristic of Uzbekistan. The estimated nectar production per hectare was approximately 276.4±42.5 L, suggesting that T. cordata can serve as a reliable nectar source despite its relatively brief flowering period.
Pollination dependence analysis demonstrated that T. cordata relies substantially on pollinators rather than self-pollination. This highlights the species’ dual role as a critical food resource for pollinators-especially honeybees-and as a plant species dependent on their activity for successful reproduction. In summary, T. cordata exhibits physiological adaptability to climatic stressors and possesses considerable potential as a melliferous species. It holds promise as a valuable botanical resource for pollinator conservation and for promoting ecological stability in arid environments.
Acknowledgments
This work was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF-2018R1A6A1A03024862) and Uzbekistan-Collaboration Research Smart AI UZ (2022K1A3A9A05036394). We gratefully acknowledge the research team in Uzbekistan for their valuable support during the fieldwork, and Dr. Ehsan Rahimi for his helpful contributions to the manuscript preparation.
References
-
Adgaba, N., A. Al-Ghamdi, Y. Tadesse, A. Getachew, A. M. Awad, M. J. Ansari, A. A. Owayss, S. E. A. Mohammed and A. S. Alqarni. 2017. Nectar secretion dynamics and honey production potentials of some major honey plants in Saudi Arabia. Saudi J. Biol. Sci. 24(1): 180-191.
[https://doi.org/10.1016/j.sjbs.2016.05.002]

- Bareke, T., A. Addi, K. Wakjira and T. Kumsa. 2021. Dynamics of nectar secretion, honey production potential and colony carrying capacity of Coffea arabica L., Rubiaceae. J. Agric. Environ. Int. Dev. 115(1): 125-138.
-
Bascompte, J. 2019. Mutualism and biodiversity. Curr. Biol. 29(11): R467-R470.
[https://doi.org/10.1016/j.cub.2019.03.062]

-
Baude, M., W. E. Kunin, N. D. Boatman, S. Conyers, N. Davies, M. A. K. Gillespie, R. D. Morton, S. M. Smart and J. Memmott. 2016. Historical nectar assessment reveals the fall and rise of floral resources in Britain. Nature 530: 85-88.
[https://doi.org/10.1038/nature16532]

-
Bertsch, A. 1983. Nectar production of Epilobium angustifolium L. at different air humidities; nectar sugar in individual flowers and the optimal foraging theory. Oecologia 59: 40-48.
[https://doi.org/10.1007/bf00388069]

-
Cawoy, V., J. M. Kinet and A. L. Jacquemart. 2008. Morphology of nectaries and biology of nectar production in the distylous species Fagopyrum esculentum. Ann. Bot. 102(5): 675-684.
[https://doi.org/10.1093/aob/mcn150]

-
Chabert, S., C. Sénéchal, A. Fougeroux, J. Pousse, F. Richard, E. Nozières, O. Geist, V. Guillemard, S. Leylavergne, C. Malard, A. Benoist, G. Carré, É. Caumes, C. Cenier, A. Treil, S. Danflous and B. E. Vaissière. 2020. Effect of environmental conditions and genotype on nectar secretion in sunflower (Helianthus annuus L.). OCL 27: 51.
[https://doi.org/10.1051/ocl/2020040]

-
Christmann, S. 2019. Do we realize the full impact of pollinator loss on other ecosystem services and the challenges for any restoration in terrestrial areas? Restor. Ecol. 27(4): 720-725.
[https://doi.org/10.1111/rec.12950]

-
De Jaegere, T., S. Hein and H. Claessens. 2016. A review of the characteristics of small-leaved lime (Tilia cordata Mill.) and their implications for silviculture in a changing climate. Forests 7(3): 56.
[https://doi.org/10.3390/f7030056]

-
Djurabaev, O. D. and J. K. Rashidov. 2021. The main directions of effective management and development of the beekeeping industry. E3S Web Conf. 282: 02002.
[https://doi.org/10.1051/e3sconf/202128202002]

-
Dmitruk, M., B. Denisow, E. Chrzanowska, A. Dąbrowska and M. Bożek. 2024. Comparison of nectar and pollen resources in various Tilia species: A case study from southern Poland. Trees 38(4): 953-967.
[https://doi.org/10.1007/s00468-024-02527-4]

-
Engel, M. S. 2000. A new interpretation of the oldest fossil bee (Hymenoptera: Apidae). Am. Mus. Novit. 2000(3296): 1-11.
[https://doi.org/10.1206/0003-0082(2000)3296<0001:anioto>2.0.co;2]

- Eremia, N., A. Chiriac, E. Scripnic and S. Modvala. 2017. Influence of temperature on nectar collection and storage in the hive during honey harvest. Sci. Pap. Anim. Sci. Ser. Zootehnie 68: 40-44.
- Falk, W., H. J. Klemmt, F. Binder and B. Reger. 2016. Die Winterlinde-Standort, Wachstum und waldbauliche Behandlung in Bayern. LWF Wissen 78: 20-29.
- FAO. 1995. Planning and Design of Forest Plantations. FAO For. Pap. No. 123. Retrieved from https://www.fao.org/4/t0122e/t0122e08.htm
-
Garibaldi, L. A., L. G. Carvalheiro, S. D. Leonhardt, M. A. Aizen, B. R. Blaauw, R. Isaacs, M. Kuhlmann, D. Kleijn, A. M. Klein, C. Kremen and L. Morandin. 2014. From research to action: enhancing crop yield through wild pollinators. Front. Ecol. Environ. 12(8): 439-447.
[https://doi.org/10.1890/130330]

-
Irwin, R. E., J. L. Bronstein, J. S. Manson and L. Richardson. 2010. Nectar robbing: ecological and evolutionary perspectives. Annu. Rev. Ecol. Evol. Syst. 41(1): 271-292.
[https://doi.org/10.1146/annurev.ecolsys.110308.120330]

-
Jacquemart, A. L., L. Moquet, P. Ouvrard, J. Quetin-Leclercq, M. F. Hérent and M. Quinet. 2018. Tilia trees: toxic or valuable resources for pollinators? Apidologie 49: 538-550.
[https://doi.org/10.1007/s13592-018-0581-3]

- Jung, C. 2008. Economic value of honeybee pollination on major fruit and vegetable crop in Korea. Korean J. Apic. 23: 147-152.
- Jung, C. and J. H. Shin. 2022. Evaluation of crop production increase through insect pollination service in Korean agriculture. Korean J. Appl. Entomol. 61(1): 229-238.
-
Jumayev, J. M., M. Z. Kholmurodov and K. A. Khalilova. 2021. Phenology and growth indicators of honey trees and bushes in Uzbekistan. E3S Web Conf. 244: 02050.
[https://doi.org/10.1051/e3sconf/202124402050]

- Kim, M. J., M. Son, J. Lee and C. Jung. 2022. Blooming time of Tilia amurensis Rupr. in mountainous area and prediction of its blooming progress using growing degree day model. Korean J. Agric. For. Meteorol. 24(1): 1-12.
-
Kim, Y. K., H. W. Yoo, H. Y. Kwon and S. J. Na. 2021. Estimation of nectar secretion, sugar content and honey production of Prunus mume (Siebold) Siebold and Zucc. J. Apic. 36(3): 141-147.
[https://doi.org/10.17519/apiculture.2021.09.36.3.141]

-
Klein, A. M., B. E. Vaissière, J. H. Cane, I. Steffan-Dewenter, S. A. Cunningham, C. Kremen and T. Tscharntke. 2007. Importance of pollinators in changing landscapes for world crops. Proc. R. Soc. B 274: 303-313.
[https://doi.org/10.1098/rspb.2006.3721]

-
McCombs, A. L., D. Debinski, K. Reinhardt, M. J. Germino and P. Caragea. 2022. Warming temperatures affect meadow-wide nectar resources, with implications for plant-pollinator communities. Ecosphere 13(7): e4162.
[https://doi.org/10.1002/ecs2.4162]

- McGregor, S. E. 1976. Insect pollination of cultivated crop plants (No. 496). Agric. Res. Serv. USDA.
-
Memmott, J., P. G. Craze, N. M. Waser and M. V. Price. 2007. Global warming and the disruption of plant-pollinator interactions. Ecol. Lett. 10(8): 710-717.
[https://doi.org/10.1111/j.1461-0248.2007.01061.x]

-
Mu, J., Y. Peng, X. Xi, X. Wu, G. Li, K. J. Niklas and S. Sun. 2015. Artificial asymmetric warming reduces nectar yield in a Tibetan alpine species of Asteraceae. Ann. Bot. 116(6): 899-906.
[https://doi.org/10.1093/aob/mcv042]

-
Na, S. J., Y. K. Kim and J. M. Park. 2024. Nectar characteristics and honey production potential of five rapeseed cultivars and two wildflower species in South Korea. Plants 13(3): 419.
[https://doi.org/10.3390/plants13030419]

-
Ollerton, J., R. Winfree and S. Tarrant. 2011. How many flowering plants are pollinated by animals? Oikos 120: 321-326.
[https://doi.org/10.1111/j.1600-0706.2010.18644.x]

-
Pacini, E., M. Nepi and J. L. Vesprini. 2003. Nectar biodiversity: a short review. Plant Syst. Evol. 238(1): 7-21.
[https://doi.org/10.1007/s00606-002-0277-y]

- Park, J. M., S. J. Na, H. Y. Kwon and Y. K. Kim. 2025. Evaluation of Nectar Characteristics and Potential Honey Production of Robinia pseudoacacia. J. Korean Soc. For. Sci. 114(1): 84-93.
- Pawlikowski, T. 2010. Pollination activity of bees (Apoidea: Apiformes) visiting the flowers of Tilia cordata Mill. and Tilia tomentosa Moench in an urban environment. J. Apic. Sci. 54(2): 73-79.
-
Poinar, G. O. Jr and B. N. Danforth. 2006. A fossil bee from Early Cretaceous Burmese amber. Science 314(5799): 614-614.
[https://doi.org/10.1126/science.1134103]

-
Potts, S. G., V. Imperatriz-Fonseca, H. T. Ngo, M. A. Aizen, J. C. Biesmeijer, T. D. Breeze, L. V. Dicks, L. A. Garibaldi, R. Hill, J. Settele and A. J. Vanbergen. 2016. Safeguarding pollinators and their values to human well-being. Nature 540(7632): 220-229.
[https://doi.org/10.1038/nature20588]

-
Rahimi, E. and C. Jung. 2024. A global estimation of potential climate change effects on pollinator-dependent crops. Agric. Res.: 1-11.
[https://doi.org/10.1007/s40003-024-00802-x]

-
Razanov, S. F., H. S. Khaietskyi, O. O. Alieksieiev and H. V. Hytsol. 2019. Evaluation of the forest nectar-polliniferous trees and efficiency of their usage in the nectar-bearing line of bees in the conditions of Vinnytsia Region. Agric. For. 12: 214-224.
[https://doi.org/10.37128/2707-5826-2019-1-17]

-
Reilly, J. R., D. R. Artz, D. Biddinger, K. Bobiwash, N. K. Boyle, C. Brittain, J. Brokaw, J. W. Campbell, J. Daniels, E. Elle, J. D. Ellis, S. J. Fleisher, J. Gibbs, R. L. Gillespie, K. B. Gundersen, L. Gut, G. Hoffman, N. Joshi, O. Lundin, K. Mason, C. M. McGrady, S. S. Peterson, T. L. Pitts-Singer, S. Rao, N. Rothwell, L. Rowe, K. L. Ward, N. M. Williams, J. K. Wilson, R. lsaacs. and R. Winfree. 2020. Crop production in the USA is frequently limited by a lack of pollinators. Philos. Trans. R. Soc. B-Biol. Sci. 287(1931): 20200922.
[https://doi.org/10.1098/rspb.2020.0922]

-
Rumiantsev, M., V. Luk’yanets, O. Kobets, I. Obolonyk, O. Tarnopilska, S. Pozniakova, S. Musienko and O. Tupchii. 2021. Distribution and natural regeneration of Tilia cordata Mill. in Ukrainian plain forests in a changing climate. Folia Forest. Pol., Ser. A - For. 63(2): 93-102.
[https://doi.org/10.2478/ffp-2021-0014]

-
Son, M. W., G. H. Lee and C. Jung. 2023. Analysis of floral nectar secretion and plant-pollinator interactions based on flower characteristics of 18 honey plant species. J. Apic. 38(3): 255-266.
[https://doi.org/10.17519/apiculture.2023.09.38.3.255]

- Son, M. W. and C. Jung. 2021. Effects of blooming in ground cover on the pollinator network and fruit production in apple orchards. Korean J. Appl. Entomol. 60(1): 115-122.
-
Southwick, E. E. and L. Southwick Jr. 1992. Estimating the economic value of honey bees (Hymenoptera: Apidae) as agricultural pollinators in the United States. J. Econ. Entomol. 85(3): 621-633.
[https://doi.org/10.1093/jee/85.3.621]

-
Sultanova, R., M. Martynova and R. Sazgutdinova. 2022. Honey-bearing potential of Tilia cordata Mill. forests in the Southern Urals. Front. Ecol. Evol. 10: 832442.
[https://doi.org/10.3389/fevo.2022.832442]

-
Takkis, K., T. Tscheulin and T. Petanidou. 2018. Differential effects of climate warming on the nectar secretion of early- and late-flowering Mediterranean plants. Front. Plant Sci. 9: 874.
[https://doi.org/10.3389/fpls.2018.00874]

-
Tilman, D., J. Fargione, B. Wolff, C. D̓antonio, A. Dobson, R. Howarth and D. Swackhamer. 2001. Forecasting agriculturally driven global environmental change. Science 292(5515): 281-284.
[https://doi.org/10.1126/science.1057544]

- USDA NRCS. 2010. Tree Density Table. United States Department of Agriculture Natural Resources Conservation Service. Retrieved from https://efotg.sc.egov.usda.gov/references/public/WI/CRP-627-Tree-Density-Table.pdf
-
Watanabe, M. E. 1994. Pollination worries rise as honey bees decline. Science 265(5176): 1170-1170.
[https://doi.org/10.1126/science.265.5176.1170]

-
Westphal, C., R. Bommarco, G. Carré, E. Lamborn, N. Morison, T. Petanidou, S. G. Potts, S. P. M. Roberts, H. Szentgyörgyi, T. Tscheulin, B. E. Vaissière, M. Woyciechowski, J. C. Biesmeijer, W. E. Kunin, J. Settele and I. Steffan-Dewenter. 2008. Measuring bee diversity in different European habitats and biogeographical regions. Ecol. Monogr. 78(4): 653-671.
[https://doi.org/10.1890/07-1292.1]

- Ylejbees.com. n.d. Beekeeping in Uzbekistan. Retrieved May 21, 2025, from https://pcelovodstvodoskaobavlenij.ylejbees.com/beekeeping-in-the-world/beekeeping-in-uzbekistan