Acaricidal Activities against Varroa destructor and Chemical Composition of Korean Propolis Essential Oils
Abstract
Varroa destructor is an ectoparasitic mite and one of the major contributors of honey bee population decline over the world. Propolis is a resinous gum material collected by bees from surrounding plants used for sealing and protecting hives, and was reported to have acaricidal effects on Varroa mites. In this study, we compared the chemical composition of propolis essential oils (PEOs) from three sites, Andong, Gongju field and mountain, and tested their acaricidal effects against Varroa mites in the laboratory. Propolis volatiles of Korean origin were composed predominantly of sesquiterpenes, aromatic compounds, and fatty acids. The major constituents in Andong PEO were the sesquiterpenes, β-eudesmol (31.97%) and γ-eudesmol (9.55%), and benzoic acid(10.21%). Gongju field PEO had the aromatic principal constituents bis(2-ethylhexyl) phthalate (14.73%). On the other hand, the predominant compounds in Gongju mountain PEO were 2-methylbenzyl cyanide(17.93%) and 2-propen-1-one, 1-(2,6-dihydroxy-4-methoxyphenyl)-3-phenyl-, (E)-(17.15%). Our GC-MS analysis showed variation in chemical composition at different sites, which might be due to differences in vegetation. Gongju mountain PEO exhibited the strongest activity against V. destructor (LD50=294.86 µg/mL) among the evaluated propolis essential oils. However, the acaricidal activities of Gongju mountain was lower than that of the positive control, tau-fluvalinate(LD50=142.85µg/mL). Further studies on propolis samples collected from different sites may reveal more potent acaricidal PEOs, potentially contributing to the development of safer environmental controls for mites.
Keywords:
Korean propolis, Essential oil, Sesquiterpenes, Fatty acids, Varroa destructorINTRODUCTION
Propolis is one of the beehive products which is collected by honey bees from different parts of plants, such as buds and exudates (Greenaway et al., 1990). Honey bees, such as Apis mellifera, use propolis to seal hive walls and its entrances, strengthen the combs, and embalm dead invaders (Ghisalberti, 1979). Various chemical investigations on propolis solvent extracts and essential oils have shown that its composition is complex and depends on the local plant flora, region, and climatic conditions of the collection site (Jihene et al., 2018). Propolis also contains volatile organic compounds, which, despite their low proportions, have important biological effects(Bankova et al., 1994; Borčić et al., 1996; Torres et al., 2008; Jihene et al., 2018). Various terpenes (monoterpenes and sesquiterpenes), alcohols (mainly aromatic alcohols), phenols, aldehydes, ketones, acids (from acetic to stearic acid), esters, many series of alkanes, alkylated benzenes, and naphthalene have been reported as volatile organic compounds in propolis from different origins (Bankova et al., 1992; Marcucci et al., 2001).
Varroa destructor (Anderson and Trueman, 2000) plays a critical role in the decline of honey bee populations. This parasite feeds on the bee’s fat body and hemolymph (Ramsey et al., 2019), leading to various infections at the individual and colony levels (Annoscia et al., 2012). Additionally, the mites serve as vectors for several bee viruses (Grozinger and Flenniken, 2019). Infestation by Varroa mites can result in colony diseases, death, and substantial economic losses to the beekeeping industry (Orantes-Bermejo et al., 2010; Nazzi et al., 2012).
Various approaches, including physical, biological, and chemical methods, have been employed to control the mites. In recent years, synthetic acaricides such as tau-fluvalinate, amitraz, and coumaphos have been used as the primary means of control. However, these chemicals result in the accumulation of residues in hive products can be harmful to bees (Bogdanov et al., 1998; Rosenkranz et al., 2010). To address these concerns, natural alternatives such as organic acids (formic acid, oxalic acid, and lactic acid), essential oils (thymol, carvacrol, and menthol) (Akyol and Yeninar, 2009; Smodiš Škerl et al., 2011; Tutun et al., 2018; Pusceddu et al., 2021), and propolis (Drescher et al., 2017; Pusceddu et al., 2018; Hussein and Ayoub, 2019; Pusceddu et al., 2021; Bragança Castagnino et al., 2023; Ayad et al., 2024) have been reported to be environmentally friendly options with strong acaricidal activities against the mites (Vilarem et al., 2021).
There have been several reports on the chemical composition and biological activities of propolis essential oils (PEO) originated from European (Janes and Bumba, 1974), Asian (Greenaway et al., 1989; Kaškonienė et al., 2014), Latin American (Salatino et al., 2005) and a few African countries (Bankova et al., 1998; Haile et al., 2012; Jihene et al., 2018; Ayari et al., 2020).
However, to the best of our knowledge, the chemical composition and acaricidal activity of Korean PEOs have not yet been investigated. Therefore, the present study aimed to analyze the chemical composition of essential oils from Korean propolis collected from three localities and to evaluate their acaricidal effects against V. destructor.
MATERIALS AND METHODS
1. Materials and chemicals
Sugar, a deep freezer, a Clevenger apparatus, a condenser, a plastic cylinder (6.5 cm in height and 3.5 cm inner diameter), and cryogenic 20 mL glass vials (4.3 cm in height and 3.0 cm inner diameter), Whatman No. 1 filter paper, acetone, and tau-fluvalinate were used. Chloroform and anhydrous sodium sulfate were purchased (Daejung chemicals and metals Co., LTD, Siheung-si, Gyeonggi-do Province, and Duksan pure chemical Co. LTD, Ansan-si, Gyeonggi-do) in Korea, respectively. All other chemicals were purchased from Sigma-Aldrich St. (Louis, Missouri, USA), unless specified.
2. Sample collection
Korean propolis, produced by Apis mellifera was collected from the Beelab at GyeongKuk National University (formerly Andong University), Andong, as well as from Gongju mountain and Gongju field in spring 2021.
3. Extraction
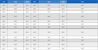
Fresh propolis was collected from cloths by putting the cloths in a deep freezer at -80℃ (Ilshin BioBase Co., Ltd., Dongducheon-si, Korea). The samples were then ground and subjected to hydro-distillation in a Clevenger-type apparatus for 3 h. The oils were dried with anhydrous sodium sulfate, and the yielded oils are presented in Table 1. Essential oil yield was calculated as % (w/w).
4. GC-MS condition
GC-MS analysis was conducted using a GC (7890B, Agilent Technologies, Santa Clara, California, USA) coupled with an MS (5977A Network, Agilent Technologies, Santa Clara, California, USA). An HP 5MS column (non-polar column, Agilent Technologies) with 30 m × 250 μm internal diameter (i.d.) and 0.25 μm film thickness was equipped in GC. The carrier gas was helium with flow rate of 1 mL/min. The injector temperature was 230℃, and injection mode was performed in split mode with split ratio 10 : 1. The initial oven temperature was at 40℃ and held for 5 min. It was then increased to 250℃ at a rate of 6℃/min and held at this temperature for 20 min. The total run-time was 60 min. Mass spectra were recorded in electron ionization (EI) mode at 70 eV, scanning the 50-650 m/z range. The compound identification was done by comparing the mass spectra (MS) of the compounds with those in the database of NIST11 (National Institute of Standards and Technology, Gaithersburg, USA). Relative amounts of the detected compounds were calculated based on the peak areas of the total ion chromatograms (TIC) and literature search.
5. Collection of Varroa mites
In this study, a total of 200 adult female Varroa mites were collected from adult bees of colonies maintained at the Experimental Apiaries of Gyeongkuk National University. Colonies that had not been treated with acaricides for at least one month were used in all trials. Mites were separated from the bees by placing them in a large jar with a mesh lid containing 20 g of powdered sugar. The jar was then inverted and shaken. The dislodged Varroa fell through the mesh lid and were collected in a sieve to remove the powdered sugar. The Varroa mites were tipped onto a clean sheet of paper. Five active females were selected under the microscope, and later transferred into 20 mL glass vials treated with test samples.
6. Contact toxicity assay of propolis essential oil to Varroa destructor
Tau-fluvalinate was dissolved in 96% ethanol to prepare concentrations equivalent to the standard positive control for assay. Propolis essential oils were tested for their acaricidal activity against Varroa mites using the complete exposure test. After preliminary tests, five serial concentrations of each test sample were prepared (0.5 mg/mL, 0.125 mg/mL, 0.03 mg/mL, and 0.008 mg/mL) in acetone from a stock solution of 2 mg/mL. Then, 1 mL of each solution was placed into a 20 mL glass scintillation vial using micropipette based on the method described by Sabahi et al. (2018). Acetone (purity 99.5%; CAS No. 67-64-1; Daejung, Siheung-si, Gyeonggi-do Province, Korea) was used as the negative controls. The vials were rolled out to distribute the solution on their inner walls. After the acetone evaporated (ap. 5 min) under a steady stream of nitrogen gas, the vials were capped. Five adult female mites were introduced into each vial using a fine paintbrush and left in the dark at room temperature (26℃ and 65% RH). A total of 165 mites were used for each test samples in triplicate. To determine the toxicity of the oils, the number of dead mites was recorded at 4 h post-treatment (hpt). Mites were determined as dead if they did not move when observed under a microscope after being touched with a fine-tipped brush.
7. Fumigant toxicity of propolis essential oil to honey bees
The test was carried out using a plastic cylinder (8 cm in height and 8.5 cm inner diameter). Whatman No. 1 filter paper was placed in the plastic cylinder and 1 mL of solution propolis oil (2 mg/mL, 0.5 mg/mL, 0.125 mg/mL, 0.03 mg/mL acetone) was uniformly distributed on the filter paper. The solvent was allowed to evaporate for 3 min and 12 workers honey bees were placed in the plastic cylinder treated with the propolis oil. The same procedure was applied for the negative control, using only acetone. The number of dead honey bees were counted at 4, 8, 12, 24, 48 and 72 h post-treatmnt.
8. Statistical analysis
All the results are expressed as mean ± SD (standard deviation). The one-way ANOVA followed by Dunnett’s test was used for statistical analysis. Differences with P values < 0.05 were considered statistically significant. For the acaricidal activity test, individual test samples, regression lines, 4h-LC50 values, X2 and 95% confidence limits were calculated for the toxicity test responses of propolis oils to adult females of Varroa mites (V. destructor) using the Probit analysis in SPSS version 16 (Chicago, SPSS Inc., 2007). Since the toxicity results were binomial response (alive or dead mites), we used probit analysis. Statistical significance of differences in mortality between the oils collected from different sites were examined using the Chi-Square test of independence.
RESULTS
1. Propolis oil yield
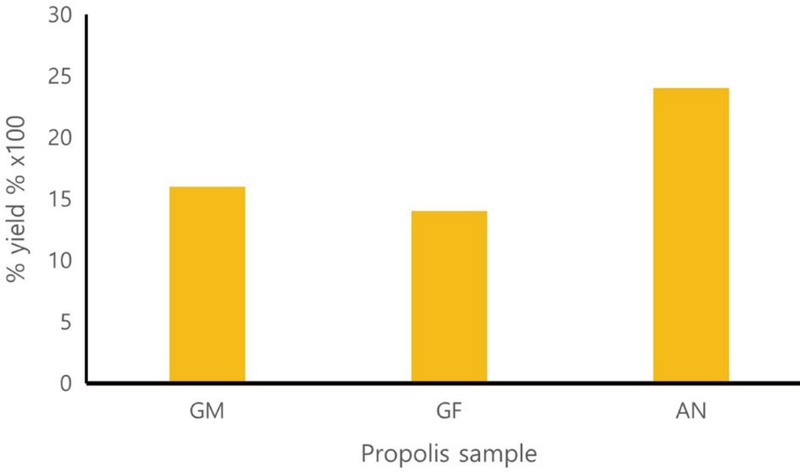
It has been reported that the optimal percentage propolis oil yield is obtained at 3 h (Ayari et al., 2020). We used this optimized method to extract essential oils uisng the hydro-distillation technique. The perecntage yield of the oils from propolis collected from Gongju mountain and Gongju field hives were 0.16% and 0.14%, respectively (Table 1 and Fig. 1). However, samples collected from Andong yielded a higher oil yield (0.24%). In general the precentagy oil yield of Korean propolis collected from three sites ranged from 0.14% to 0.24%.
2. Chemical composition
The composition of Korean propolis essential oils is shown in Table 2 and Fig. 2-4. A total of 133 volatile organic compounds were identified from Korean propolis samples. 22, 91 and 93 compounds were detected in the volatile oils of propolis collected from Andong, Gongju field, and Gongju mountain sites, respectively (Table 2). The major constituents of propolis oil from Andong were the sesquiterpenes β-eudesmol (31.97%) and γ-eudesmol (9.55%), as well as benzoic acid (10.21%) (Fig. 2). On the other hand, propolis samples from other sites, such as Gongju mountain and Gongju field, contained lower concentrations of β-eudesmol, 6.14% and 2.51%, respectively.
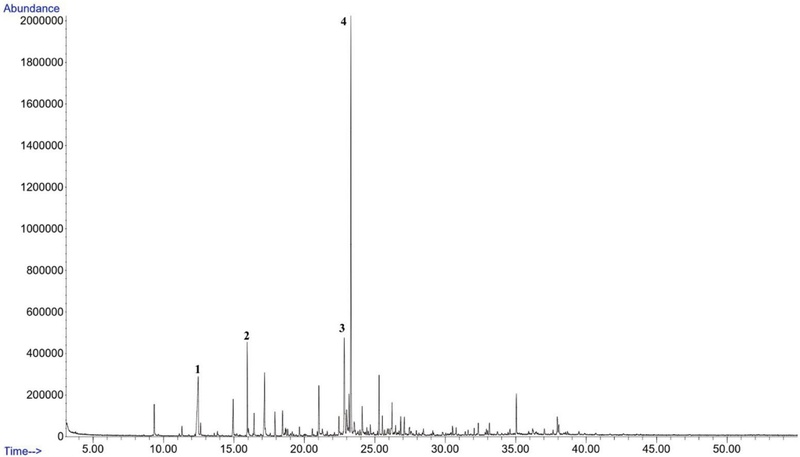
GC of Korean propolis collected from Andong (AN). Major compounds 1. Benzoic acid (10.21%); 2. 2-Propen-1-ol, 3-phenyl(5.78%); 3. γ-eudesmol(9.55%); 4. β-Eudesmol(31.97%).
The propolis oil collected form Gongju mountain contained aromatic principal constituents such as 2-methylbenzyl cyanide (17.93%) and 2-Propen-1-one, 1-(2,6-dihydroxy-4-methoxyphenyl)-3-phenyl-, (E)-, (17.15%) (Table 2 and Fig. 3). The propolis from Gongju field had bis(2-ethylhexyl) phthalate (14.73%) as the major compound (Table 2 and Fig. 4).
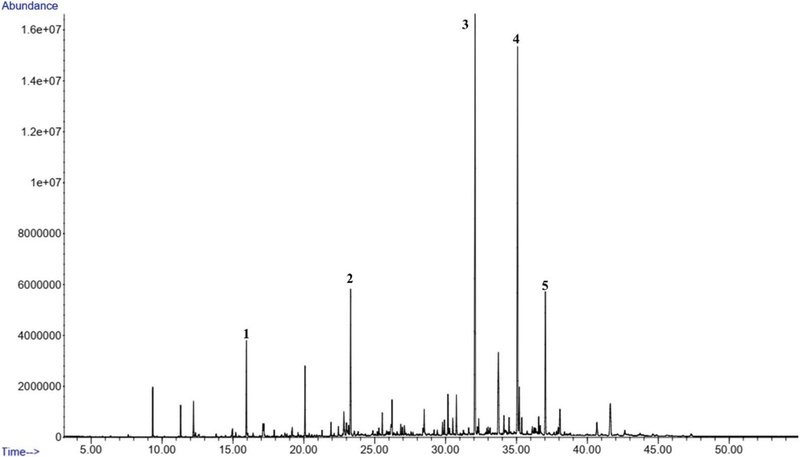
GC of Korean propolis collected from Gongju mountain (GM). Major compounds 1. 2-Propen-1-ol, 3-phenyl(3.21%); 2. β-Eudesmol (6.14%); 3. 2-Methylbenzyl cyanide (17.93%); 4. 2-Propen-1-one, 1-(2,6-dihydroxy-4-methoxyphenyl)-3-phenyl-, (E)- (17.15%); 5. Bis(2-ethylhexyl) phthalate (6.36%).
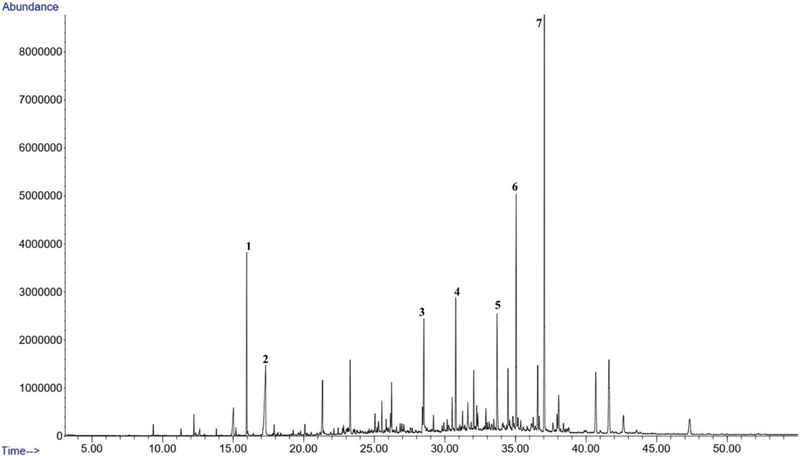
GC of Korean propolis collected from Gongju field (GF). Major compounds 1. 2-Propen-1-ol, 3-phenyl (4.66%); 2. n-Decanoic acid (5.06%); 3. n-Hexadecanoic acid (3.67%); 4. Benzyl cinnamate (4.10%); 5. Nonadecane, 9-methyl- (4.24%); 6. 2-Propen-1-one, 1-(2,6-dihydroxy-4-methoxyphenyl)-3-phenyl-, (E)(7.43%); 7. Bis(2-ethylhexyl) phthalate (14.73%).
3. Toxicity to V. destructor and honey bees
Varroa destructor is an ectoparasitic mite that affects colonies of honey bee, Apis mellifera, in several countries in the world. Synthetic acaricides have been used for the traditional way of control during the last years but resistant mites and residues in honey bee products have increased. Substances of botanical origin have emerged as natural alternative acaricides to diminish the population levels of the mite. In the present work, the bioactivity of propolis oils from different locations in Korean against V. destructor was evaluated. The propolis essential oil of Gongju mountain significantly showed the strongest activity (LC50=294.86 μg/mL) against V. destructor compared to the remaining samples. Acaricidal activity of Gongju mountain propolis oil is lower than those of the positive control, tau-fluvalinate (LC50=142.85) (Table 3). On the other hand, propolis oil from the Gongju field propolis oils showed the lowest LC50 value (1200.69 μg/mL) against V. destructor. The standard positive control (tau-fluvalinate) was significantly more toxic to V. destructor than all propolis oil samples examined. On the other hand, no honey bee mortality was recorded in the study period (4, 8, 12, 24, 48, and 72 h).
DISCUSSION
Our Korean PEO percentage yield results are comparable to those reported for Tunisian propolis (0.095-0.32%). A higher propolis essential oil yield was reported for Ethiopian propolis by Haile et al. (2012) (0.92-1.2%), and lower oil yields were reported for Algerian propolis (Segueni et al., 2010) (0.03-0.11%) and Greek propolis (0.03-0.10%) (Melliou et al., 2007). The differences in the reported percentage oil yields might be due to different factors, such as environmental conditions, botanic sources, and bee species that collected the samples.
β-Eudesmol was also reported as a major component of one sample from Tunisia (12.43%) (Ayari et al., 2020), Morocco (11-30%) (El-Guendouz et al., 2019), and Taiwan (13.9%) (Trusheva et al., 2017) propolis essential oils in lower concentration compared to Andong but higher than those in Gongju propolis EOs. French propolis was reported to have β-eudesmol as a principal component (30%) (Bankova et al., 2014) comparable to Andong propolis EOs. The north Italian propolis contained β-eudesmol as a major compound (2.33-12.8%) (Pellati et al., 2013). Compounds such as benzoic acid (10.21%) and 2-propen-1-ol, 3-phenyl (5.78%) were also found as predominant compounds in the propolis oil collected from Andong. Benzoic acid (3.1-30.1%) and benzyl benzoate (0.2-13.1%) have also been reported as predominant compounds in North Italian propolis oil obtained by hydro-distillation method (Pellati et al., 2013; Bankova et al., 2014). Propolis volatile oils collected from Eastern Turkey were also reported to have major aromatic alcohol compounds, such as phenylethyl alcohol (7.7%) and benzyl alcohol (7.4%) (Bankova et al., 2014). Bis(2-ethylhexyl) phthalate identified in the Gongju field EO was also reported in lower concentration (1.79%) from the ethanol extract of Indonesian propolis(Kalsum et al., 2016), compared with our results.
Algerian propolis ethanol extracts (10%) were reported to be effective in killing Varroa mites while being harmless to honey bees (Ayad et al., 2024). Therefore, propolis oil can be used as safely to control V. destructor without side effects on honey bees.
According to Meyer’s toxicity index, extracts with LC50<1000 μg/mL are considered toxic, while extracts with LC50>1000 μg/mL are considered non-toxic (Meyer et al., 1982). Clarkson’s toxicity criterion for the toxicity assessment of plant extracts classifies extracts in the following order: extracts with LC50 above 1000 μg/mL are non-toxic, LC50 of 500-1000 μg/mL are low toxic, extracts with LC50 of 100-500 μg/mL are medium toxic, while extracts with LC50 of 0-100 μg/mL are highly toxic (Clarkson et al., 2004). Based on the above toxicity levels, propolis oil from Gongju mountain and Andong showed medium toxicity to V. destructor. It is worth mentioning that propolis oil from Gongju field was non-toxic to the mites. The reason for the difference in the activity of propolis in different sites surrounding the Gongju city is due to difference in their volatile organic compounds.
The presence of the major compound bis(2-ethylhexyl) phthalate, or the synergistic effects of the mixture of volatile organic compounds in Gongju mountain propolis oil, might be responsible for its enhanced activity, which needs further investigation. Further work on collection of propolis from different sites may lead to identification of acaricidal volatile organic compounds (VOC) which enables to control V. destructor in environmentally friendly manner.
CONCLUSION
The three Korean propolis essential oils (PEO) investigated exhibited varying chemical compositions and acaricidal properties. They primarily consist of sesquiterpenes, aromatic compounds, and fatty acids. Propolis oils from Gongju Mountain and Andong demonstrated significant acaricidal activity against V. destructor while remaining safe for honey bees. Further exploration involving a broader range of propolis essential oils collected from diverse Korean regions could be crucial for identifying more potent EOs against Varroa mites. Additionally, future research should focus on investigating the mode of action underlying the high acaricidal activity of propolis EOs.
Acknowledgments
This work was supported by a Research Grant of GyeongKuk National University (2025-2026).
We thank Hong Seokmin for his assistances in the collection of Gongju propolis samples.
References
-
Akyol, E. and H. Yeninar. 2009. Use of oxalic acid to control Varroa destructor in honey bee (Apis mellifera L.) colonies. Turk. J. Vet. Anim. Sci. 33: 285-288.
[https://doi.org/10.3906/vet-0712-16]

-
Anderson, D. and J. Trueman. 2000. Varroa jacobsoni (Acari: Varroidae) is more than one species. Exp. Appl. Acarol. 24: 165-189.
[https://doi.org/10.1023/A:1006456720416]

-
Annoscia, D., F. Del Piccolo and F. Nazzi. 2012. How does the mite Varroa destructor kill the honey bee Apis mellifera? Alteration of cuticular hydrcarbons and water loss in infested honey bees. J. Insect Physiol. 58: 1548-1555.
[https://doi.org/10.1016/j.jinsphys.2012.09.008]

-
Ayad, A. S., S. Benchaabane, T. Daas, G. Smagghe and W. Loucif-Ayad. 2024. Assessment of efficacy of Algerian propolis against the parasitic mite Varroa destructor and safety for honey bees by spray treatment. Insects 15: 75.
[https://doi.org/10.3390/insects15010075]

- Ayari, J., I. J. Karoui and M. Abderrabba. 2020. A comparative study between different Tunisian propolis essential oils and their antioxidant activities. Iran. J. Chem. Chem. Eng. 39: 217-231.
-
Bankova, V., A. Dyulgerov, S. Popov, L. Evstatieva, L. Kuleva, O. Pureb and Z. Zamjansan. 1992. Propolis produced in Bulgaria and Mongolia: phenolic compounds and plant origin. Apidologie 23: 79-85.
[https://doi.org/10.1051/apido:19920109]

-
Bankova, V., M. Popova and B. Trusheva. 2014. Propolis volatile compounds: chemical diversity and biological activity: a review. Chem. Cent. J. 8: 1-8.
[https://doi.org/10.1186/1752-153X-8-28]

-
Bankova, V., R. Christov, S. Popov, O. Pureb and G. Bocari. 1994. Volatile constituents of propolis. Z. Naturforsch. C 49: 6-10.
[https://doi.org/10.1515/znc-1994-1-202]

-
Bankova, V. S., R. Christov and A. D. Tejera. 1998. Lignans and other constituents of propolis from the Canary Islands. Phytochemistry 49: 1411-1415.
[https://doi.org/10.1016/S0031-9422(98)00108-3]

-
Bogdanov, S., V. Kilchenmann and A. Imdorf. 1998. Acaricide residues in some bee products. J. Apic. Res. 37: 57-67.
[https://doi.org/10.1080/00218839.1998.11100956]

-
Borčić, I., A. Radonić and K. Grzunov. 1996. Comparison of the volatile constituents of propolis gathered in different regions of Croatia. Flavour Fragr. J. 11: 311-313.
[https://doi.org/10.1002/(SICI)1099-1026(199609)11:5<311::AID-FFJ581>3.0.CO;2-X]

-
Bragança Castagnino, G. L., A. Meana, M. T. Cutuli de Simón and L. F. Batista Pinto. 2023. Propolis and its effects on bee diseases and pests: a systematic review. J. Apic. Res. 62: 171-184.
[https://doi.org/10.1080/00218839.2022.2154474]

-
Clarkson, C., V. J. Maharaj, N. R. Crouch, O. M. Grace, P. Pillay, M. G. Matsabisa, N. Bhagwandin, P. J. Smith and P. I. Folb. 2004. In vitro antiplasmodial activity of medicinal plants native to or naturalised in South Africa. J. Ethnopharmacol. 92: 177-191.
[https://doi.org/10.1016/j.jep.2004.02.011]

-
Drescher, N., A.-M. Klein, P. Neumann, O. Yañez and S. D. Leonhardt. 2017. Inside honey bee hives: Impact of natural propolis on the ectoparasitic mite Varroa destructor and viruses. Insects 8: 15.
[https://doi.org/10.3390/insects8010015]

-
El-Guendouz, S., B. Lyoussi, M. G. Miguel and A. C. Figueiredo. 2019. Characterization of volatiles from Moroccan propolis samples. J. Essent. Oil Res. 31: 27-33.
[https://doi.org/10.1080/10412905.2018.1520748]

-
Ghisalberti, E. 1979. Propolis: a review. Bee World 60: 59-84.
[https://doi.org/10.1080/0005772X.1979.11097738]

-
Greenaway, W., T. Scaysbrook and F. Whatley. 1989. Headspace volatiles from propolis. Flavour Fragr. J. 4: 173-175.
[https://doi.org/10.1002/ffj.2730040404]

-
Greenaway, W., T. Scaysbrook and F. Whatley. 1990. The composition and plant origins of propolis: a report of work at Oxford. Bee World 71: 107-118.
[https://doi.org/10.1080/0005772X.1990.11099047]

-
Grozinger, C. M. and M. L. Flenniken. 2019. Bee viruses: Ecology, pathogenicity, and impacts. Annu. Rev. Entomol. 64: 205-226.
[https://doi.org/10.1146/annurev-ento-011118-111942]

-
Haile, K., T. Kebede and A. Dekebo. 2012. A comparative study of volatile components of propolis (bee glue) collected from Haramaya University and Assela beekeeping centers, Ethiopia. Bull. Chem. Soc. Ethiop. 26: 353-360.
[https://doi.org/10.4314/bcse.v26i3.4]

-
Hussein, M. A. and Z. N. Ayoub. 2019. Propolis impact on the honey bee life span, Varroa mite infestation and population growth of the colony. J. Duhok Univ. 22: 300-311.
[https://doi.org/10.26682/avuod.2019.22.1.28]

- Janes, K. and V. Bumba. 1974. Composition of bee glue (propolis). Die Pharmazie 29: 544-545.
-
Jihene, A., I. J. Karoui, A. Ameni, M. Hammami and M. Abderrabba. 2018. Volatile compounds analysis of Tunisian propolis and its antifungal activity. J. Biosci. Med. 6: 115.
[https://doi.org/10.4236/jbm.2018.66009]

- Kalsum, N., A. Sulaeman, B. Setiawan and I. W. T. Wibawan. 2016. Phytochemical profiles of propolis Trigona spp. from three regions in Indonesia using GC-MS. J. Biol. Agric. Health 6: 31-37.
-
Kaškonienė, V., P. Kaškonas, A. Maruška and L. Kubilienė. 2014. Chemometric analysis of volatiles of propolis from different regions using static headspace GC-MS. Open Chem. 12: 736-746.
[https://doi.org/10.2478/s11532-014-0521-7]

-
Marcucci, M. C., F. Ferreres, C. Garcıa-Viguera, V. Bankova, S. De Castro, A. Dantas, P. Valente and N. Paulino. 2001. Phenolic compounds from Brazilian propolis with pharmacological activities. J. Ethnopharmacol. 74: 105-112.
[https://doi.org/10.1016/S0378-8741(00)00326-3]

-
Melliou, E., E. Stratis and I. Chinou. 2007. Volatile constituents of propolis from various regions of Greece Antimicrobial activity. Food Chem. 103: 375-380.
[https://doi.org/10.1016/j.foodchem.2006.07.033]

-
Meyer, B., N. Ferrigni, J. Putnam, L. Jacobsen, D. j. Nichols and J. L. McLaughlin. 1982. Brine shrimp: a convenient general bioassay for active plant constituents. Planta Med. 45: 31-34.
[https://doi.org/10.1055/s-2007-971236]

-
Nazzi, F., S. P. Brown, D. Annoscia, F. Del Piccolo, G. Di Prisco, P. Varricchio, G. Della Vedova, F. Cattonaro, E. Caprio and F. Pennacchio. 2012. Synergistic parasite-pathogen interactions mediated by host immunity can drive the collapse of honey bee colonies. PLoS Pathog. 8: e1002735.
[https://doi.org/10.1371/journal.ppat.1002735]

-
Orantes-Bermejo, F. J., A. G. Pajuelo, M. M. Megías and C. T. Fernández-Píñar. 2010. Pesticide residues in beeswax and beebread samples collected from honey bee colonies (Apis mellifera L.) in Spain. Possible implications for bee losses. J. Apic. Res. 49: 243-250.
[https://doi.org/10.3896/IBRA.1.49.3.03]

-
Pellati, F., F. P. Prencipe and S. Benvenuti. 2013. Headspace solid-phase microextraction-gas chromatography - mass spectrometry characterization of propolis volatile compounds. J. Pharm. Biomed. Anal. 84: 103-111.
[https://doi.org/10.1016/j.jpba.2013.05.045]

-
Pusceddu, M., D. Annoscia, I. Floris, D. Frizzera, V. Zanni, A. Angioni, A. Satta and F. Nazzi. 2021. Honey bees use propolis as a natural pesticide against their major ectoparasite. Proc. Roy. Soc. B 288: 20212101.
[https://doi.org/10.1098/rspb.2021.2101]

-
Pusceddu, M., I. Floris, A. Mura, P. Theodorou, G. Cirotto, G. Piluzza, S. Bullitta, A. Angioni and A. Satta. 2018. The effects of raw propolis on Varroa-infested honey bee (Apis mellifera) workers. Parasitol. Res. 117: 3527-3535.
[https://doi.org/10.1007/s00436-018-6050-0]

-
Ramsey, S. D., R. Ochoa, G. Bauchan, C. Gulbronson, J. D. Mowery, A. Cohen, D. Lim, J. Joklik, J. M. Cicero and J. D. Ellis. 2019. Varroa destructor feeds primarily on honey bee fat body tissue and not hemolymph. Proc. Natl. Acad. Sci. 116: 1792-1801.
[https://doi.org/10.1073/pnas.1818371116]

-
Rosenkranz, P., P. Aumeier and B. Ziegelmann. 2010. Biology and control of Varroa destructor. J. Invertebr. Pathol. 103: S96-S119.
[https://doi.org/10.1016/j.jip.2009.07.016]

-
Sabahi, Q., M. M. Hamiduzzaman, J. S. Barajas-Pérez, J. M. Tapia-Gonzalez and E. Guzman-Novoa. 2018. Toxicity of anethole and the essential oils of lemongrass and sweet marigold to the parasitic mite Varroa destructor and their selectivity for honey bee (Apis mellifera) workers and larvae. Psyche 2018.
[https://doi.org/10.1155/2018/6196289]

-
Salatino, A., É. W. Teixeira, G. Negri and D. Message. 2005. Origin and chemical variation of Brazilian propolis. Evid. Based Complement. Alternat. Med. 2: 33-38.
[https://doi.org/10.1093/ecam/neh060]

- Segueni, N., F. Khadraoui, F. Moussaoui, A. Zellagui, N. Gherraf, M. Lahouel and S. Rhouati. 2010. Volatile constituents of Algerian propolis. Ann. Biol. Res. 1: 103-107.
-
Smodiš Škerl, M. I., M. Nakrst, L. Žvokelj and A. Gregorc. 2011. The acaricidal effect of flumethrin, oxalic acid and amitraz against Varroa destructor in honey bee (Apis mellifera Carnica) colonies. Acta Vet. Brno 80: 51-56.
[https://doi.org/10.2754/avb201180010051]

-
Torres, R. N. S., J. A. D. Lopes, J. M. Moita Neto and A. M. d. G. L. Citó. 2008. The volatile constituents of propolis from Piaui. Quim. Nova 31: 479-485.
[https://doi.org/10.1590/S0100-40422008000300003]

-
Trusheva, B., D. Ivanova, M. Popova and V. Bankova. 2017. Insights into the essential oil compositions of Brazilian red and Taiwanese green propolis. Nat. Prod. Commun. 12: 1934578X1701200214.
[https://doi.org/10.1177/1934578X1701200214]

-
Tutun, H., N. Koç and A. Kart. 2018. Plant essential oils used against some bee diseases. Turk. J. Agric. - Food Sci. Technol. 6: 34-45.
[https://doi.org/10.24925/turjaf.v6i1.34-45.1502]

-
Vilarem, C., V. Piou, F. Vogelweith and A. Vétillard. 2021. Varroa destructor from the laboratory to the field: Control, biocontrol and ipm perspectives - a review. Insects 12: 800.
[https://doi.org/10.3390/insects12090800]