
Mineral Profiling of Honey Samples from Uzbekistan and Russia through Chemometric Analysis
Abstract
Mineral profiles of honey reflect the floral and environmental factors of nectar plants and play a role in determining its authenticity. However, systematic studies applying chemometric approaches to characterize these profiles are still in the early stages. This study evaluated five macroelements (Ca, K, Mg, Na, and P) and four microelements (Cu, Fe, Mn, and Zn) in 31 samples (29 honeys and 2 syrups) from Uzbekistan and Russia, using ICP-OES and ICP-MS. K and Ca were found most abundant in the honey samples, ranging from 97.3-977.3 mg/kg and 80.9-414.1 mg/kg. The concentrations of individual elements and the total mineral content differed significantly among floral origins, with the lowest levels observed in the syrup samples. Chemometric analyses, including Pearson’s correlation analysis, revealed positive correlations between honey color and each of Ca, Cu, Mg, and P. Principal Component analysis clearly discriminated authentic honeys from syrup. Clustering Uzbek and Russian honeys together indicated that floral source exerts a stronger influence than geographical origin. This study provides the first comprehensive assessment of honey minerals in Uzbekistan and establishes a reference framework for developing a mineral-based database to support authenticity verification of honeys distributed in Uzbekistan.
Keywords:
Macroelements, Microelements, Authenticity, Syrup, ICP, ColorINTRODUCTION
Honey (blossom honey) is a widely consumed natural sweetener produced by honey bees from floral nectar (Codex Alimentarius, 2001). It is a highly complex substance, comprising nearly 200 compounds, including sugars, water, proteins, organic acids, vitamins, and minerals (Alvarez-Suarez et al., 2013; da Silva et al., 2016). The characteristics of honey are influenced by bee species, seasonality, environmental parameters, beekeeping practices, storage conditions, and most notably, its geographical location and botanical source (Leite et al., 2000; Bogdanov et al., 2008; Kaškonienė et al., 2010; da Silva et al., 2016).
In our previous studies, the botanical origins of honey samples collected from the Tashkent region in Uzbekistan were first identified through melissopalynological analysis, followed by a comprehensive investigation into their physicochemical properties. These studies highlighted the significant contribution of floral origin to honey composition and confirmed the overall high quality of Uzbek honey, thereby prompting further investigation into its mineral content (Jang et al., 2025a; Sun et al., 2025).
Minerals in honey primarily originate from the soil type and nectar-secreting plants (Bogdanov et al., 2007; Schmidlová et al., 2024). Solayman et al. (2016) identified 54 mineral elements previously reported in honey, comprising seven macroelements and 47 microelements. The total mineral content of honey has been reported to range from 126 to 4060 mg/kg, depending on the type of honey (Vanhanen et al., 2011). Several studies have shown that mineral levels and compositions differ significantly according to botanical and regional origins (Nalda et al., 2005; Ajtony et al., 2007; Bogdanov et al., 2007; Czipa et al., 2018; Bodó et al., 2020; Vukašinovič-Pešić et al., 2020). Dark-colored honey, whose color is largely determined by its floral origin, contains higher mineral content than light-colored ones (Alqarni et al., 2014; Bodó et al., 2021; Pavlin et al., 2023). Moreover, Liu et al. (2021) reported that honey exhibited higher levels of elements than syrup, and that syrup adulteration could be distinguished from authentic honey through chemometric analysis. Taken together, these findings underscore the significance of mineral profiling not only for evaluating the botanical origin and quality of honey but also for identifying potential adulteration.
The Republic of Uzbekistan is a Central Asian country bordered by Kazakhstan to the north, Kyrgyzstan and Tajikistan to the east and southeast, Turkmenistan to the west, and Afghanistan to the south. It has a predominantly continental and arid climate, which varies across regions (Belolipov et al., 2013). Beekeeping in the country is essential for supporting local economic growth through pollination services and honey production (Farmanov, 2025). Uzbek people have been consuming honey and honey-based products as a food resource for a long time (Abdiniyazova et al., 2016). However, despite its importance, comprehensive studies on the chemical characteristics of honey, particularly its mineral content, remain limited.
This study aims to expand the understanding of the mineral composition of honey collected from Tashkent, Uzbekistan. Specifically, this study focuses on determining the levels of macro- and microelements in honey samples and employs chemometric analyses to investigate the relationships among mineral composition, color, floral source and geographical origin, as well as to verify honey authenticity. We hypothesized that the floral origin of honey would have a stronger influence on its mineral composition than its geographical source. Through this approach, the study not only establishes baseline data for assessing the quality and authenticity of Uzbek honey but also offers valuable insights into the distinctive mineral signatures that characterize honey produced in Central Asia.
MATERIALS AND METHODS
1. Honey samples
A total of 31 samples, including 30 honeys from Apis mellifera collected in Tashkent, Uzbekistan, in 2022, were analyzed. Among the collected samples, 27 were produced in Tashkent, while 4 were produced in Russia. Among the Uzbek honeys, 7 were Alhagi, 4 Helianthus, 4 Medicago, 1 Salvia, 1 Ferula, 1 Onobrychis, and 1 Tilia. 6 samples were classified as multifloral honey, comprising 2 dominated by Helianthus and 1 each dominated by Alhagi, Calligonum, Ferula, and Onobrychis. 2 samples were classified as artificial syrup prepared from fructose, glucose, and sucrose. From Russia, 3 samples were Helianthus and 1 was Medicago. Although the number of samples was relatively small due to the limited market availability, all honey products available in Tashkent during the sampling period were collected, ensuring that the dataset represents the overall honey market in Tashkent. Detailed information on the botanical origins of these honey samples is available in previous studies (Jang et al., 2025a; Sun et al., 2025), and a summary is presented in Table 1.
2. Determination of honey color
The Pfund scale, which is the most commonly used method, is widely applied to determine honey color based on color intensity (Bogdanov et al., 2004). A portable honey color photometer (HI96785, Hanna Instruments, Seoul, South Korea), calibrated using a glycerol standard, was used for color measurement. After calibration, approximately 4 mL of honey was transferred into a cuvette, and the color was measured carefully to avoid air bubbles. The values were classified according to the USDA honey color standards (1985) as follows: 0-8 mm-water white, 8-17 mm-extra white, 17-34 mm-white, 34-50 mm-extra light amber, 50-85 mm-light amber, 85-114 mm-amber, and >114-140 mm-dark amber. The results are summarized in Table 1.
3. Mineral analysis
Sample preparation for mineral quantification was performed according to the method previously described by Jang et al. (2025b). A 0.5 g portion of homogenized honey was treated with 5 mL of concentrated nitric acid (HNO3) and digested in a high-pressure microwave digestion system equipped with a single reaction chamber (UltraWAVE, Milestone, Sorisole, Italy) at 180℃ for 30 min. After digestion, the solution was allowed to cool to room temperature and then diluted with distilled water to a final volume of 50 mL. Ca, Fe, K, Mg, Na, and P in the honey samples were analyzed using inductively coupled plasma optical emission spectroscopy (ICP-OES; Agilent 5110, Agilent Technologies, Santa Clara, CA, USA), whereas Cu, Mn, and Zn were determined by inductively coupled plasma mass spectrometry (ICP-MS; Agilent 7800, Agilent Technologies, Santa Clara, CA, USA). For ICP-OES, standard instrumental conditions were applied as described by Gąsecka et al. (2021), while the ICP-MS parameter settings were adopted from Alhagri and Albeshry (2023). Contents of both macroelements (Ca, K, Mg, Na, and P) and microelements (Cu, Fe, Mn, and Zn) in the samples were expressed in mg/kg. All measurements were performed in triplicate to ensure reproducibility and accuracy.
4. Statistical analysis
All statistical and chemometric analyses were performed using R software (version 4.4.1; R Core Team, Vienna, Austria). Normality and homogeneity of variances were assessed using the Shapiro-Wilk and Levene’s tests, respectively. Since the data met the assumption of normality but violated the assumption of homogeneity of variances (p<0.05), differences among floral origins for macroelements, microelements, and total mineral content were analyzed using Welch’s ANOVA, followed by the Games-Howell post hoc test. Principal component analysis (PCA) was applied to explore the relationships between the honey samples and their mineral compositions. PCA score and loading plots were generated to visualize the clustering patterns of the samples and the contribution of variables, respectively. Pairwise correlations among mineral elements and between the mineral content and color intensity of the honey samples were evaluated using Pearson’s correlation analysis. The significance level was set at α=0.05, and all results are presented as mean±standard deviation (SD).
RESULTS AND DISCUSSION
1. Elemental composition of honey samples
The concentrations of 5 macroelements (Ca, K, Mg, Na, and P) and 4 microelements (Cu, Fe, Mn, and Zn) were analyzed in a total of 31 samples, including 25 Uzbek honeys of distinct floral origins (Alhagi, Helianthus, Medicago, Ferula, Onobrychis, Salvia, Tilia, and multi-floral types), 4 Russian honeys, and 2 artificial syrup samples. These data are summarized in Tables 2 and 3, and the variations in total mineral content, which was calculated as the sum of the 9 measured elements, are depicted in Fig. 1. All elements, as well as the total mineral content, showed statistically significant differences among the samples (p<0.001). Similarly, previous studies have also reported significant differences in mineral composition depending on geographical and botanical origins (Bogdanov et al., 2007; Valverde et al., 2022; Pavlin et al., 2023; Mongi, 2024). These differences are thought to be influenced by agricultural practices and the mineral composition of soils, as plants absorb and transfer these elements into nectar and subsequently into honey (Hemalatha and Satyanarayana, 2015; Lanjwani and Channa, 2019; Mongi, 2024). Furthermore, Table 4 provides reference data on the macroelement and microelement contents of honeys with different floral origins from various countries, including Poland and Italy (Grembecka and Szefer, 2013), Serbia (Jovetić et al., 2017), China (Zhou et al., 2013), Spain (González-Miret et al., 2005), and India (Nayik and Nanda, 2016), for comparison with the present results. The mineral concentrations observed in the present study were generally comparable to those reported in honeys from other parts of the world (González-Miret et al., 2005; Grembecka and Szefer, 2013; Zhou et al., 2013; Jovetić et al., 2017).
Contents of macroelements (mg/kg) in honey and syrup samples from different floral origins collected in Tashkent, Uzbekistan, in 2022

Contents of microelements (mg/kg) in honey and syrup samples from different floral origins collected in Tashkent, Uzbekistan, in 2022
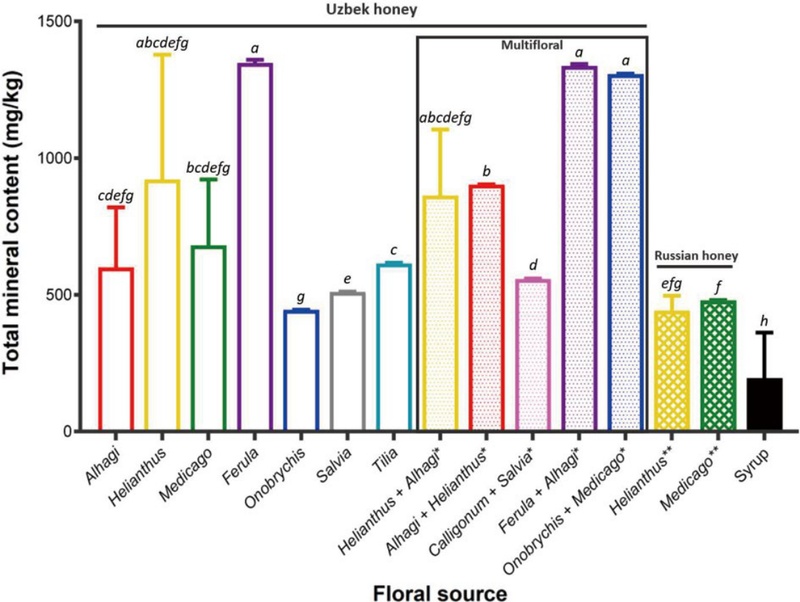
Total mineral contents (mg/kg) in honey and syrup samples from different floral origins collected in Tashkent, Uzbekistan, in 2022. Values are presented as mean±standard deviation. Different superscript letters indicate significant differences among floral groups (Welch’s ANOVA followed by the Games-Howell post hoc test for multiple comparisons, p<0.05). Asterisks (*) indicate multi-floral honeys, and double asterisks (**) denote honeys produced in Russia.

Comparative summary of macroelement and microelement contents (mg/kg) in monofloral honey samples of various floral origins from different countries, as reported in previous studies
K was the most abundant element across all honey samples, with the exception of the syrup samples (Table 2). Comparable findings have been documented for honeys from Serbia (Sakač et al., 2019), New Zealand (Vanhanen et al., 2011), Malaysia (Moniruzzaman et al., 2014), Slovenia (Pavlin et al., 2023), Tanzania (Mongi, 2024), Romania (Pop et al., 2022), Ethiopia (Gebeyehu and Jalata, 2023), and Italy (Pisani et al., 2008), although the levels of K differed considerably among countries and also varied with the botanical origins of the honeys produced in each country.
In the Uzbek honey samples, the concentrations of macroelements varied within the following ranges (mg/kg): Ca, 80.93-414.06; K, 97.33-977.29; Mg, 17.49-78.65; Na, 58.67-130.84; and P, 23.10-120.19 (Table 2). The Russian honeys contained Ca, 130.08-145.35; K, 108.12-196.57; Mg, 29.99-35.40; Na, 57.63-65.60; and P, 45.70-77.75 mg/kg. Overall, the macroelement contents of these honeys were within the wide ranges documented in other countries. For instance, Romanian honeys contained Ca, 15.7-429.8; K, 46-1507; Mg, 10.8-339.0; Na, 28.4-292.6; and P, 21.8-145.1 mg/kg (Pop et al., 2023). Similarly, Argentine honeys showed Ca, 33-83; K, 126-1490; Mg, 8-44; Na, 22-62; and P, 33-150 mg/kg (Cabrera and Santander, 2022), while Hungarian honeys exhibited Ca, 9.12-124; K, 102-2212; Mg, 3.83-60.2; Na, 3.02-17.9; and P, 32.0-138 mg/kg (Czipa et al., 2018).
Across all honey samples from Uzbekistan and Russia, the concentrations of trace elements ranged as follows (mg/kg): Cu, 0.098-0.558; Fe, 1.05-6.70; Mn, 0.016-0.194; and Zn, 0.100-1.140 (Table 2). Similar concentration ranges have been reported in honeys from other regions, as observed for the macroelements, including Argentina (Cu, <0.1-0.27; Fe, 0.38-4.21; Mn, 0.12-1.62; Zn, 0.45-4.82 mg/kg; Cabrera and Santander, 2022), Switzerland (Cu, 0.051-1.966; Fe, 0.136-9.852; Mn, 0.125-12.354; Zn, 0.016-4.133 mg/kg; Bogdanov et al., 2007), Slovenia (Cu, 0.015-0.626; Fe, 0.00-134.59; Mn, 0.17-15.77; Zn, 0.01-4.53 mg/kg; Pavlin et al., 2023), and Italy (Cu, 0.172-5.9; Fe, 0.97-13.7; Mn, 0.13-16.9; Zn, 0.72-3.66 mg/kg; Pisani et al., 2008).
2. Differences between countries and among floral origins
Fig. 1 demonstrate significant differences (p<0.001) in total mineral content among honeys of different origins, ranging from 386.02 to 1630.01 mg/kg in Uzbek honeys, and 401.18 to 516.08 mg/kg in Russian honeys. Pavlin et al. (2023) reported total mineral contents of honeys with various botanical and geographical origins ranging from 267.80 to 4423.37 mg/kg, produced from different floral sources in Slovenia, Bulgaria, Turkey, Morocco, and Croatia.
However, comparing the present findings with previously published data remains challenging. Variations in sample decomposition approaches (such as microwave, wet, or dry ashing) and analytical methodologies could account for inconsistencies among studies (Pisani et al., 2008). Therefore, establishing standardized procedures for these methods is essential to ensure consistency across future investigations.
Meanwhile, the syrup samples exhibited significantly lower concentrations of most elements than the authentic honeys, and their total mineral content was the lowest among all samples (Tables 2 and 3; Fig. 1). This finding is consistent with the results reported by Liu et al. (2021), who observed that syrup samples showed significantly lower mineral concentrations than honeys, except for Ba. In their study, Ca, Na, P, Ba, and Mg were identified as the most abundant elements in syrups, whereas in our results, Na, K, Ca, Mg, and Fe predominated. This discrepancy is likely attributed to the fact that syrups are mainly produced from sugar beet or sugarcane, whose intrinsic chemical composition markedly affects the elemental profile of the final product (Pohl and Stecka, 2011).
Ferula, multifloral (Ferula), and Onobrychis+Medicago honey samples exhibited high values in most analyzed elements as well as in total mineral content (Tables 2 and 3; Fig. 1). The genus Ferula, which includes around 180-185 species globally (Kadereit and Bittrich, 2018), is found throughout the Mediterranean, Siberia, Central Asia, and northern Africa (Pimenov and Leonov, 1993; Wu et al., 2003; Ajani and Ajani, 2008; Qin et al., 2023). Numerous Ferula species possess medicinal properties and are widely utilized in both folk and traditional pharmacy practices (Qin et al., 2023). Furthermore, Ferula species exhibited the highest concentrations of macroelements (P, K, Ca, Mg, and S) and microelements (Cu and Zn) among 21 plant species from different genera, as reported by Ghafoor et al. (2019). This may explain, at least partially, the elevated mineral levels observed in the honey derived from regions containing Ferula species.
In contrast to Helianthus and Helianthus+Alhagi or Ferula and Ferula+Alhagi honeys, significant differences in total mineral content were observed between Alhagi and Alhagi+Helianthus honeys, as well as between Onobrychis and Onobrychis+Medicago honeys (Fig. 1). In Helianthus+Alhagi honey, Helianthus pollen accounted for 37.9% and Alhagi 11.6% (Table 1), while the total mineral contents of the respective monofloral honeys were 922.2 and 601.0 mg/kg, correspondingly (Fig. 1). Therefore, the slightly lower, but not statistically different, total mineral content of the multifloral sample (863.7 mg/kg; Fig. 1) is likely due to the minor contribution of Alhagi with a lower mineral content. A similar trend was also observed for Ferula+Alhagi honey. Additionally, Alhagi+Helianthus honey contained 26.2% Alhagi and 25.0% Helianthus pollen, showing nearly equal proportions of the two major floral sources (Table 1). Since the total mineral contents of Alhagi and Helianthus monofloral honeys did not differ significantly, the Alhagi+Helianthus honey also exhibited no significant difference in mineral composition compared with the corresponding monofloral honeys (Fig. 1). In the case of Onobrychis+Medicago honey, the two dominant pollen types, Onobrychis and Medicago, accounted for 39.4% and 33.1%, respectively (Table 1). However, this sample differed statistically from both corresponding monofloral honeys (Fig. 1), which is likely attributable to the contribution of minor floral sources with relatively higher mineral contents.
Helianthus and Medicago honeys were present in both Uzbekistan and Russia, showing no statistical differences in the concentrations of most individual elements and the total mineral content between the two countries (Tables 2 and 3; Fig. 1). A previous study also reported no significant difference in total mineral content between acacia honeys produced in Slovenia and those from Bulgaria (Pavlin et al., 2023). Furthermore, Bogdanov et al. (2007) highlighted that the trace element profile of honeys is primarily determined by their botanical origin rather than by geographical or environmental factors.
3. Chemometric and correlation analysis
Fig. 2 shows the PCA biplot illustrating the relationships among honey samples and the contribution of each element to the total variance based on the loadings of the variables. The first and second principal components explained 48.3% and 19.4% of the total variance, respectively, accounting for 67.7% in total. PC1 showed positive correlations with Na, Zn, and Mn, whereas PC2 was negatively correlated only with P. The PCA plot revealed a clear separation between the syrup and honey samples, with the syrups located distinctly on the positive side of PC1, mainly influenced by high Na and Zn contents. A similar pattern was observed by Liu et al. (2021), who also found distinct clustering between authentic honeys and syrup samples in PCA based on their mineral compositions. In contrast, most honey samples clustered toward the negative side of PC1, characterized by higher levels of Ca, K, Mg, Cu, and P. Uzbek and Russian honeys showed no distinct separation from each other, suggesting that the element compositions from the two regions are similar. These PCA results are consistent with the statistical comparisons in Tables 2 and 3 and Fig. 1, further supporting that mineral patterns are primarily influenced by botanical rather than geographical origin. This finding is also in agreement with the results reported by Bogdanov et al. (2007), as discussed earlier.
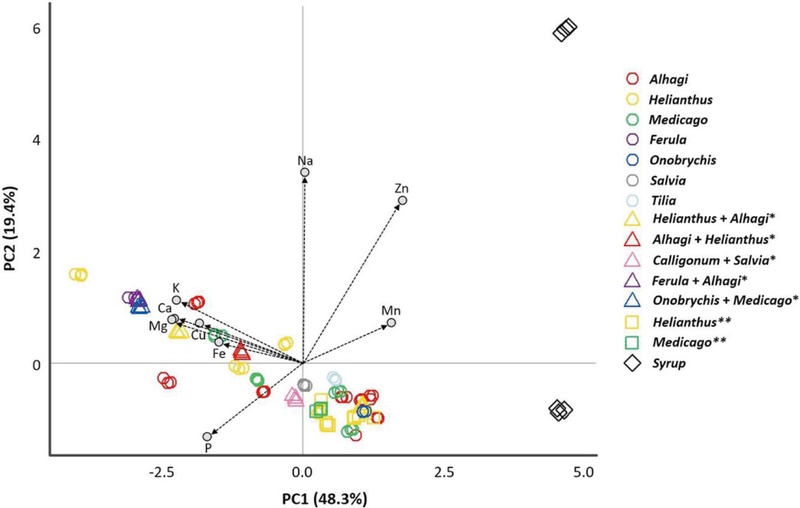
Principal component analysis (PCA) biplot based on the concentrations of macro- and microelements in honey and syrup samples from different floral origins collected in Tashkent, Uzbekistan, in 2022. Arrows represent mineral loading vectors, and symbols indicate individual samples grouped by floral origin. Asterisks (*) indicate multifloral honeys, and double asterisks (**) denote honeys produced in Russia.
The Pearson correlation coefficients among the elemental concentrations and color in the honey samples are presented in Fig. 3. Strong positive correlations were observed among the major elements, particularly between Ca and Mg (r=0.95, p<0.001), Ca and K (r=0.88, p<0.001), and K and Mg (r=0.83, p<0.001). Similarly, notable correlations were also found between Cu and K (r=0.71, p<0.001) and between Na and Zn (r=0.69, p<0.001). Moderate but significant correlations were detected between Ca and Cu (r=0.59, p<0.001), Cu and Mg (r=0.58, p<0.001), Fe and P (r=0.53, p<0.01), Mg and P (r=0.49, p<0.01), Mn and Zn (r=0.47, p<0.01), and Ca and P (r=0.44, p<0.05). In contrast, moderate negative correlations were observed between P and Zn (r= -0.54, p<0.01), Ca and Zn (r= -0.41, p<0.01), and Mg and Zn (r= -0.41, p<0.01). Kędzierska-Matysek et al. (2018) found a strong positive correlation between K and Mg, and a moderate correlation between Ca and Mg in Polish honey samples. In another study, Slovakian honeys exhibited a strong positive correlation between Cu and Mg, as well as a moderate positive correlation between Ca and Mg (Kacaniová et al., 2009). A strong positive correlation between Mn and Zn was found in Swiss honeys (Bogdanov et al., 2007). These correlations collectively suggest that the mineral balance of honey is a complex outcome of floral origin, soil composition, and surrounding environmental factors. In particular, cations such as K+, Ca2+ and Mg2+ are often taken up concurrently due to overlapping transport mechanisms in plant roots (Marschner, 2012). This may partially explain the strong positive correlations observed among these elements in the honey samples. Additionally, the color of the honey samples showed significant positive correlations with Ca (r=0.48, p<0.01), Cu (r=0.40, p<0.05), Mg (r=0.40, p<0.05), and P (r=0.37, p<0.05), while exhibiting negative correlations with Mn (r= -0.58, p<0.001) and Zn (r= -0.51, p<0.01). Several studies have reported that darker-colored honeys generally contain higher levels of specific minerals and total mineral content compared to lighter-colored honeys (Terrab et al., 2003; González-Miret et al., 2005; Nalda et al., 2005; Osman et al., 2007; Pisani et al., 2008; Alqarni et al., 2014; Solayman et al., 2016; Bodó et al., 2021; Pavlin et al., 2023).
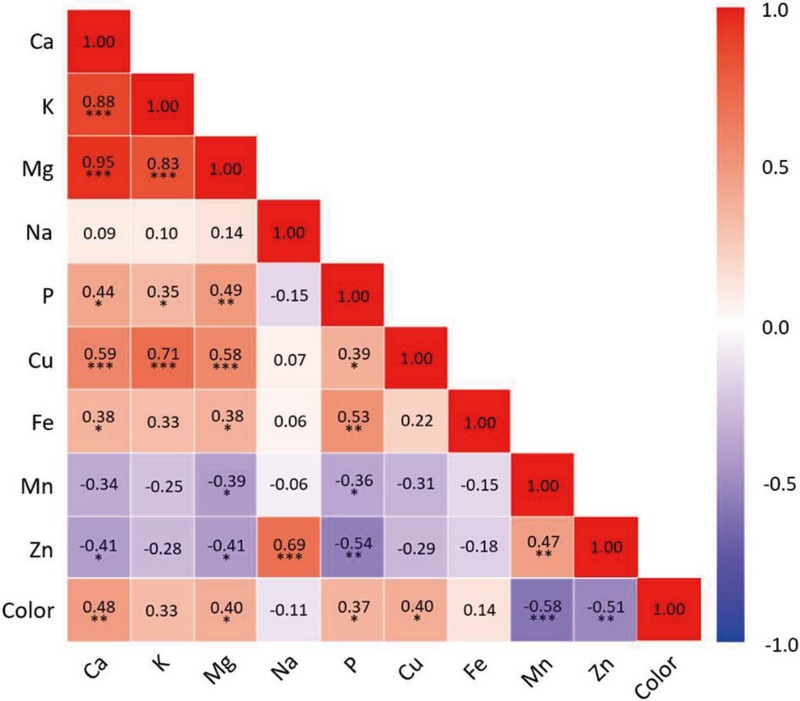
Pearson correlation matrix among the mineral elements and color in honey and syrup samples from different floral origins collected in Tashkent, Uzbekistan, in 2022. The numbers indicate Pearson correlation coefficients, and asterisks denote significance levels: p<0.05 (*), p<0.01 (**), and p<0.001 (***).
CONCLUSION
This study represents the first comprehensive assessment of mineral composition of honeys from Tashkent, Uzbekistan, using chemometric techniques. The integration of multivariate statistical analysis with mineral quantification provided clear discrimination between authentic honeys and syrup samples, underscoring the robustness of this approach for authenticity evaluation. Furthermore, the consistent clustering of Uzbek and Russian honeys indicates that floral source exerts a stronger influence than geographical origin. Due to limited sample availability in the local market of Tashkent, particularly for Russian honeys, the generalizability of geographic trends remains restricted. Future studies should expand sample collection beyond Tashkent to other regions of Uzbekistan to overcome this limitation. Nevertheless, this study establishes a reference framework for developing a mineral-based database to support authenticity verification of honey distributed in Tashkent, Uzbekistan.
Acknowledgments
We sincerely thank Dr. Mojtaba Esmeily (Agriculture Research Institute, Gyeongkuk National University, Andong, Republic of Korea) for his assistance with English editing. Following are results of a study on the “Glocal University Project Group in Gyeongkuk National University-Gyeongbuk Provincial College” Project, supported by the Ministry of Education and National Research Foundation of Korea.
References
-
Abdiniyazova, G. J., O. K. Khojimatov and V. V. Pak. 2016. Honey in traditional cuisine of Uzbekistan and analysis of melliferous flora of Karakalpakstan. J. Ethn. Foods 3(3): 222-227.
[https://doi.org/10.1016/j.jef.2016.07.002]

-
Ajani, Y. and M. Ajani. 2008. A new species of Ferula (Umbelliferae) from southern Iran. Edinb. J. Bot. 65(3): 425-431.
[https://doi.org/10.1017/S0960428608005052]

-
Ajtony, Z., L. Bencs, R. Haraszi, J. Szigeti and N. Szoboszlai. 2007. Study on the simultaneous determination of some essential and toxic trace elements in honey by multi-element graphite furnace atomic absorption spectrometry. Talanta 71: 683-690.
[https://doi.org/10.1016/j.talanta.2006.05.023]

-
Alhagri, I. A. and M. Albeshry. 2023. Microwave-assisted digestion using dilute nitric acid and hydrogen peroxide for multi-element determination in wheat flour by ICP-MS. J. Chem. 2023(1): 2349838.
[https://doi.org/10.1155/2023/2349838]

-
Alqarni, A. S., A. A. Owayss, A. A. Mahmoud and M. A. Hannan. 2014. Mineral content and physical properties of local and imported honeys in Saudi Arabia. J. Saudi Chem. Soc. 18(5): 618-625.
[https://doi.org/10.1016/j.jscs.2012.11.009]

-
Alvarez-Suarez, J. M., F. Giampieri and M. Battino. 2013. Honey as a source of dietary antioxidants: structures, bioavailability and evidence of protective effects against human chronic diseases. Curr. Med. Chem. 20: 621-638.
[https://doi.org/10.2174/092986713804999358]

-
Belolipov, I. V., D. E. Zaurov and S. W. Eisenman. 2013. The geography, climate and vegetation of Uzbekistan. pp. 5-7. in Medicinal Plants of Central Asia: Uzbekistan and Kyrgyzstan, eds. by Eisenman, S. W., D. E. Zaurov and L. Struwe, Springer, New York, NY.
[https://doi.org/10.1007/978-1-4614-3912-7_2]

-
Bodó, A., L. Radványi, T. Kőszegi, R. Csepregi, D. U. Nagy, Á. Farkas and M. Kocsis. 2020. Melissopalynology, antioxidant activity and multielement analysis of two types of early spring honeys from Hungary. Food Biosci. 35: 100587.
[https://doi.org/10.1016/j.fbio.2020.100587]

-
Bodó, A., L. Radványi, T. Kőszegi, R. Csepregi, D. U. Nagy, Á. Farkas and M. Kocsis. 2021. Quality evaluation of light- and dark-colored Hungarian honeys, focusing on botanical origin, antioxidant capacity and mineral content. Molecules 26(9): 2825.
[https://doi.org/10.3390/molecules26092825]

-
Bogdanov, S., K. Ruoff and L. P. Oddo. 2004. Physico-chemical methods for the characterisation of unifloral honeys: a review. Apidologie 35(Suppl. 1): S4-S17.
[https://doi.org/10.1051/apido:2004047]

-
Bogdanov, S., M. Haldimann, W. Luginbühl and P. Gallmann. 2007. Minerals in honey: environmental, geographical and botanical aspects. J. Apic. Res. 46(4): 269-275.
[https://doi.org/10.1080/00218839.2007.11101407]

-
Bogdanov, S., T. Jurendic, R. Sieber and P. Gallmann. 2008. Honey for nutrition and health: a review. J. Am. Coll. Nutr. 27(6): 677-689.
[https://doi.org/10.1080/07315724.2008.10719745]

-
Cabrera, M. and E. Santander. 2022. Physicochemical and sensory analysis of honeys from eastern Formosa province (Argentina) and its relationship with their botanical origin. Food Chem. Adv. 1: 100026.
[https://doi.org/10.1016/j.focha.2022.100026]

- Codex Alimentarius. 2001. Revised standard for honey. Codex Standard 12-1981 (Rev. 2). FAO/WHO, Rome.
-
Czipa, N., L. Alexa, C. J. Phillips and B. Kovács. 2018. Macro-element ratios provide improved identification of the botanical origin of monofloral honeys. Eur. Food Res. Technol. 244(8): 1439-1445.
[https://doi.org/10.1007/s00217-018-3057-9]

-
da Silva, P. M., C. Gauche, L. V. Gonzaga, A. C. O. Costa and R. Fett. 2016. Honey: chemical composition, stability and authenticity. Food Chem. 196: 309-323.
[https://doi.org/10.1016/j.foodchem.2015.09.051]

- Farmanov, J. 2025. Current status of the development of the beekeeping industry in our republic. Mod. Am. J. Bus. Econ. Entrep. 1(2): 139-147.
-
Gąsecka, M., M. Siwulski, S. Budzyńska, Z. Magdziak, P. Niedzielski, A. Budka, P. Kalač and M. Mleczek. 2021. The importance of Cu×Pb interactions to Lentinula edodes yield, major/trace elements accumulation and antioxidants. Eur. Food Res. Technol. 247: 2799-2812.
[https://doi.org/10.1007/s00217-021-03833-8]

-
Gebeyehu, H. R. and D. D. Jalata. 2023. Physicochemical and mineral contents of honey from Fitche and Addis Ababa districts in Ethiopia. Food Chem. Adv. 2: 100177.
[https://doi.org/10.1016/j.focha.2022.100177]

-
Ghafoor, K., S. Doğu, I. A. Mohamed Ahmed, G. J. Fadimu, Ü. Geçgel, F. Al Juhaimi and M. M. Özcan. 2019. Effect of some plant species on fatty acid composition and mineral contents of Ferulago, Prangos, Ferula, and Marrubium seeds and oils. J. Food Process. Preserv. 43(5): e13939.
[https://doi.org/10.1111/jfpp.13939]

-
González-Miret, M. L., A. Terrab, D. Hernanz, M. Á. Fernández-Recamales and F. J. Heredia. 2005. Multivariate correlation between color and mineral composition of honeys and their botanical origin. J. Agric. Food Chem. 53: 2574-2580.
[https://doi.org/10.1021/jf048207p]

-
Grembecka, M. and P. Szefer. 2013. Evaluation of honeys and bee products quality based on their mineral composition using multivariate techniques. Environ. Monit. Assess. 185: 4033-4047.
[https://doi.org/10.1007/s10661-012-2847-y]

- Hemalatha, K. and P. Satyanarayana. 2015. Elemental analysis of different floral honeys of East Godavari Dist., Andhra Pradesh, India. J. Environ. Sci. Toxicol. Food Technol. 9(12): 56-60.
-
Jang, H., S. Sun, B. A. Kakhramanov and C. Jung. 2025a. Quality assessment of honey samples collected in Tashkent, Uzbekistan. J. Apic. 40(2): 155-164.
[https://doi.org/10.17519/apiculture.2025.06.40.2.155]

-
Jang, H., T. Begna, S. Sun, D. Ulziibayar and C. Jung. 2025b. Possible interactive risk of copper hydroxide, inorganic bactericide of fire blight with dinotefuran and triflumizole on honey bee. J. Asia-Pac. Entomol. 28(4): 102475.
[https://doi.org/10.1016/j.aspen.2025.102475]

-
Jovetić, M., J. Trifković, D. Stanković, D. Manojlović and D. Milojković-Opsenica. 2017. Mineral content as a tool for the assessment of honey authenticity. J. AOAC Int. 100(4): 862-870.
[https://doi.org/10.5740/jaoacint.17-0145]

-
Kacaniová, M., V. Knazovicka, M. Melich, M. Fikselova, P. Massanyi, R. Stawarz, P. Hascik, T. Pechociak, A. Kuczkowska and A. Putała. 2009. Environmental concentration of selected elements and relation to physicochemical parameters in honey. J. Environ. Sci. Health B 44(4): 414-422.
[https://doi.org/10.1080/10934520802659802]

-
Kadereit, J. W. and V. Bittrich. 2018. Flowering plants. Eudicots: Apiales, Gentianales (except Rubiaceae). in The Families and Genera of Vascular Plants, Springer, Berlin, Germany.
[https://doi.org/10.1007/978-3-319-93605-5]

-
Kaškonienė, V., P. R. Venskutonis and V. Čeksterytė. 2010. Carbohydrate composition and electrical conductivity of different origin honeys from Lithuania. LWT-Food Sci. Technol. 43: 801-807.
[https://doi.org/10.1016/j.lwt.2010.01.007]

-
Kędzierska-Matysek, M., M. Florek, A. Wolanciuk, J. Barłowska and Z. Litwińczuk. 2018. Concentration of minerals in nectar honeys from direct sale and retail in Poland. Biol. Trace Elem. Res. 186(2): 579-588.
[https://doi.org/10.1007/s12011-018-1315-0]

-
Lanjwani, M. F. and F. A. Channa. 2019. Minerals content in different types of local and branded honey in Sindh, Pakistan. Heliyon 5(7): e02042.
[https://doi.org/10.1016/j.heliyon.2019.e02042]

-
Leite, J. M. C. C., L. C. Trugo, L. S. M. Costa, L. M. C. Quinteiro, O. M. Barth and V. M. L. Dutra. 2000. Determination of oligosaccharides in Brazilian honeys of different botanical origin. Food Chem. 70: 93-98.
[https://doi.org/10.1016/S0956-7135(99)00115-2]

-
Liu, T., K. Ming, W. Wang, N. Qiao, S. Qiu, S. Yi and L. Luo. 2021. Discrimination of honey and syrup-based adulteration by mineral element chemometrics profiling. Food Chem. 343: 128455.
[https://doi.org/10.1016/j.foodchem.2020.128455]

- Marschner, H. 2012. Marschner’s mineral nutrition of higher plants. 3rd ed. Academic Press, London.
-
Mongi, R. J. 2024. Influence of botanical origin and geographical zones on physicochemical properties, mineral contents and consumer acceptance of honey in Tanzania. Food Chem. Adv. 4: 100731.
[https://doi.org/10.1016/j.focha.2024.100731]

-
Moniruzzaman, M., M. A. Z. Chowdhury, M. A. Rahman, S. A. Sulaiman and S. H. Gan. 2014. Determination of mineral, trace element, and pesticide levels in honey samples originating from different regions of Malaysia compared to Manuka honey. Biomed. Res. Int. 2014(1): 359890.
[https://doi.org/10.1155/2014/359890]

-
Nalda, M. N., J. B. Yagüe, J. D. Calva and M. M. Gómez. 2005. Classifying honeys from the Soria Province of Spain via multivariate analysis. Anal. Bioanal. Chem. 382: 311-319.
[https://doi.org/10.1007/s00216-005-3161-0]

-
Nayik, G. A. and V. Nanda. 2016. A chemometric approach to evaluate the phenolic compounds, antioxidant activity and mineral content of different unifloral honey types from Kashmir, India. LWT 74: 504-513.
[https://doi.org/10.1016/j.lwt.2016.08.016]

- Osman, K. A., M. A. Al-Doghairi, S. Al-Rehiayani and M. I. Helal. 2007. Mineral contents and physicochemical properties of natural honey produced in Al-Qassim region, Saudi Arabia. J. Food Agric. Environ. 5: 142-146.
-
Pavlin, A., D. Kočar, J. Imperl, M. Kolar, G. Marolt and P. Petrova. 2023. Honey origin authentication via mineral profiling combined with chemometric approaches. Foods 12(15): 2826.
[https://doi.org/10.3390/foods12152826]

-
Pisani, A., G. Protano and F. Riccobono. 2008. Minor and trace elements in different honey types produced in Siena County (Italy). Food Chem. 107(4): 1553-1560.
[https://doi.org/10.1016/j.foodchem.2007.09.029]

- Pimenov, M. G. and M. V. Leonov. 1993. The genera of Umbelliferae. Royal Botanic Gardens, Kew, UK.
-
Pohl, P. and H. Stecka. 2011. Elemental composition of white refined sugar by instrumental methods of analysis. Crit. Rev. Anal. Chem. 41(2): 100-113.
[https://doi.org/10.1080/10408347.2011.538585]

-
Pop, I. M., D. Simeanu, S. M. Cucu-Man, A. Pui and A. Albu. 2022. Quality profile of several monofloral Romanian honeys. Agriculture 13(1): 75.
[https://doi.org/10.3390/agriculture13010075]

-
Qin, H. H., J. Cai, C. K. Liu, Y. L. Zhang, S. Y. Li, Q. Y. Li and X. L. Zhang. 2023. The plastid genome of twenty-two species from Ferula, Talassia, and Soranthus: comparative analysis, phylogenetic implications, and adaptive evolution. BMC Plant Biol. 23: 9.
[https://doi.org/10.1186/s12870-022-04027-4]

-
Sakač, M. B., P. T. Jovanov, A. Z. Marić, L. L. Pezo, Ž. S. Kevrešan, A. R. Novaković and N. M. Nedeljković. 2019. Physicochemical properties and mineral content of honey samples from Vojvodina (Republic of Serbia). Food Chem. 276: 15-21.
[https://doi.org/10.1016/j.foodchem.2018.09.149]

-
Schmidlová, S., Z. Javůrková, B. Tremlová, J. Hernik, B. Prus, S. Marcinčák and M. Pospiech. 2024. Exploring the influence of soil types on the mineral profile of honey: implications for geographical origin prediction. Foods 13(13): 2006.
[https://doi.org/10.3390/foods13132006]

-
Solayman, M., M. A. Islam, S. Paul, Y. Ali, M. I. Khalil, N. Alam and S. H. Gan. 2016. Physicochemical properties, minerals, trace elements, and heavy metals in honey of different origins: a comprehensive review. Compr. Rev. Food Sci. Food Saf. 15 (1): 219-233.
[https://doi.org/10.1111/1541-4337.12182]

-
Sun, S., H. Jang and C. Jung. 2025. Botanical origin and diversity of pollen in honey samples collected from Uzbekistan determined by the melissopalynological analysis. J. Apic. 40(2): 165-173.
[https://doi.org/10.17519/apiculture.2025.06.40.2.165]

-
Terrab, A., A. G. Gonzalez, M. J. Díez and F. J. Heredia. 2003. Mineral content and electrical conductivity of the honeys produced in northwest Morocco and their contribution to the characterisation of unifloral honeys. J. Sci. Food Agric. 83: 637-643.
[https://doi.org/10.1002/jsfa.1341]

- USDA. 1985. United States standards for grades of extracted honey. Agricultural Marketing Service, Fruit and Vegetable Division, Processed Products Branch.
-
Valverde, S., A. M. Ares, S. J. Elmore and J. Bernal. 2022. Recent trends in the analysis of honey constituents. Food Chem. 387: 132920.
[https://doi.org/10.1016/j.foodchem.2022.132920]

-
Vanhanen, L. P., A. Emmertz and G. P. Savage. 2011. Mineral analysis of monofloral New Zealand honey. Food Chem. 128(1): 236-240.
[https://doi.org/10.1016/j.foodchem.2011.02.064]

-
Vukašinovič-Pešić, V., N. Blagojević, S. Brašanac-Vukanović, A. Savić and V. Pešić. 2020. Using chemometric analyses for tracing the regional origin of multifloral honeys of Montenegro. Foods 9(2): 210.
[https://doi.org/10.3390/foods9020210]

- Wu, Z. Y., A. M. Lu, Y. C. Tang, Z. D. Chen and D. Z. Li. 2003. The families and genera of Angiosperms in China: a comprehensive analysis. Science Press, Beijing, China.
-
Zhou, J., Z. Suo, P. Zhao, N. Cheng, H. Gao, J. Zhao and W. Cao. 2013. Jujube honey from China: physicochemical characteristics and mineral contents. J. Food Sci. 78(3): C387-C394.
[https://doi.org/10.1111/1750-3841.12049]
