Manufacturing Methods and Standards for Drone Pupae, a Novel Food Ingredient from South Korea
Abstract
Drone pupae of honeybee (Apis mellifera) have been used as food in many countries to boost the immune system. In this study, we evaluated the nutritional characteristics of drone pupae, which have been registered as a novel food in South Korea. In order to use drone pupae as a food ingredient, regulations regarding the manufacturing process, heavy metals, and acid value must be complied with. We found that the optimal period for pupae collection was 17 to 22 days after spawning. Carbohydrate, crude fat, and protein content in proximate composition of drone pupae were measured as 15.89±0.11%, 26.19±0.05%, and 51.78±0.08%, respectively. Overall 18 amino acids, including 11 essential amino acids, were identified as components, with glutamic acid in the highest amount (38.28±6.2 g/kg). Drone pupae contained 13 minerals and were rich in potassium (1438.74±1.12 mg/100 g) and phosphorus (714.00±2.44 mg/100 g). Furthermore, they comprised four water-soluble vitamins (B1, B2, B9, and C) and one fat-soluble vitamin (D). Drone pupae collected for the current study did not exhibit bacterial or fungal contamination. These results suggest that honeybee drone pupae are a rich source of protein and other essential nutrients and could be used as a food ingredient. Furthermore, drone pupae could be safe to be consumed as a food supplement.
Keywords:
Drone pupae, Apis mellifera, Composition, Foodborne pathogens, MycotoxinsINTRODUCTION
Securing food supply for the growing global population is one of the most urgent challenges, as the latest United Nations population projections estimate that the global population will reach approximately 9.7 billion by 2050 (UN, 2022). Accordingly, pressures on global food systems have heightened the need to explore new alternative foods, especially meat, dairy, seed, and fish substitutes (FAO, 2023). In order to meet this challenge, edible insects have been proposed as a promising food source due to their high protein and fat content (Melgar-Lalanne et al., 2019). Furthermore, recent global assessments indicate that edible insect represent a long-standing and culturally across 128 countreis (Omuse et al., 2024), and they continue to serve as an important dietary resource for large populations globally. Insect farming has been widely recognized as an environmentally sustainable protein-production system (Smetana, 2023). Additionally, insects reproduce and grow faster than conventional livestock. Moreover, it is expected that their use in the food industry will continue to grow due to their economic and nutritional value. The Korean government recently announced new legislation to authorize seven insect species as food materials: Bombyx mori, Oxyachinensis sinuosa, Batryticatus bombyx, Tenebrio molitor larva, Protaetia brevitarsis larva, Allomyrina dichotoma larva, and Gryllus bimaculatus (MFDS, 2019a).
Drone pupae of honeybee (Apis mellifera) can be an alternative source of animal protein due to their high nutritional value (Adamkova et al., 2017). The drone pupae are known to have rich protein, vitamin, microelement, carbohydrate, and hormone content (Zhou et al., 2011; Tian and Hines, 2018). The potential of drone pupae as a nutritional food is also further supported by its high flavonoid content (Xi et al., 2018).
We evaluated the nutrient composition data and safety profile of honeybee drone pupae based on article 7-2(2) of the 「Food Sanitation」 Act (MFDS, 2019b) and registered it as a novel food ingredient. Through 「Temporary Standards and Standard Accreditation for Food」 in South Korea on July 9th, 2020 (MFDS & RDA, 2020), honeybee drone pupae have been acknowledged as a food ingredient for the first time. In this paper, we propose the manufacturing method and standards for drone pupae, registered as a temporary food ingredient.
MATERIALS AND METHODS
1. Drone pupae samples
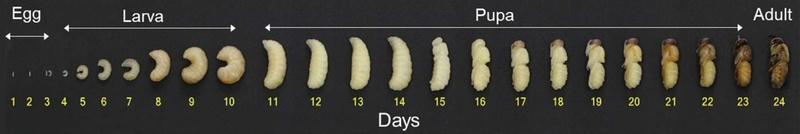
In order to employ drone pupae as a temporary food ingredient, we used European honeybees bred in apiaries and inserted a drone-only brood frame for the queen bee to spawn drone pupae. The honeybee drone pupae produced in apiaries from four colonies that naturally forage in the Korean landscape were collected and immediately frozen at -20℃. Next, the samples were subjected to freeze-drying using a freeze dryer (SFDSM24L, Sam Won Freezing Engineering Co., Busan, Korea) for 72 h. We measured the weight and shape of drone pupae from their pupa (Day 11) to imago stages, to establish the optimum production time of drone pupae to be used as a food ingredient.
2. Microbiological analysis
Microbiological analyses were performed according to the International Organization for Standardization (ISO, 1999, 2002, 2003, 2009, 2018). Briefly, the enumeration of coliform microorganisms was carried out on VRBL agar (Merck, Darmstadt, Germany) according to the standard ISO (ISO, 2006). Each sample was analyzed using specific ISO methodologies for the detection of Salmonella spp. (ISO, 2002). All presumptive Salmonella isolates were biochemically confirmed using lysine iron agar, Kligler’s iron agar, and motility indole ornithine agar. For the enumeration of coagulase-positive staphylococci (Staphylococcus aureus and other species), the Baird Parker with RPF supplement agar (Biokar Diagnostics, Allonne, France) was used (ISO, 1999). The uncertainty of measurement associated with the standardized inoculum was evaluated using the procedure described in the ISO/TS (ISO, 2009). Samples were extracted according to the ISO procedure with minor modifications for the detection of aflatoxin (ISO, 2003). The standard ISO was used for the detection and determination of ochratoxin in cereals and cereal products (ISO, 2018). Deoxynivalenol was determined according to the method described by Piacentini et al. (2015), with some modifications. Samples were extracted according to the procedure described by Pascari et al. (2019), with minor modifications for the detection of zearalenone. Samples were injected into the HPLC-FLD system (Shiseido Nanospace SI-2, Osaka Soda, Osaka, Japan).
3. Analysis of nutritional profile
The nutritional profile of samples was presented as means ± standard deviation (SD) of three replicates for each experiment. The samples were analyzed for moisture, ash, crude protein, and crude fat content using the AOAC method (AOAC, 2005). Moisture content was determined using the hot air oven (105±2℃, and 2 h) method, ash content by incineration of the dried sample at 500-550℃, crude protein of the dried sample (5 g) using the Kjeldahl method, and crude fat of the dried sample (2 g) using a soxhlet apparatus. Carbohydrate content was calculated by difference, using the formula: carbohydrate (%)=100-[(moisture%)+(ash%)+(crude protein%)+(crude fat%)].
Moisture (%)=(W1-W2)/(W1-W)×100
(W g)=Tared weight of the dish
(W1 g)=Weight of dish with samples
(W2 g)=Weight of dish+sample after drying in an oven
Ash (%)=W2/W1×100
(W1 g)=Weight of sample
(W2 g)=Weight of ash
Nitrogen (%)=(A-B×0.001)/W×V/V1×100
Crude protein (%)=N×conversion factor (6.25)
A=Volume of 0.1 N H2SO4 for sample
B=Volume of 0.1 N H2SO4 for blank
W=weight (g) of sample
V=Volume made (100 mL)
V1=Aliquot distilled (10 mL)
Crude fat (%)=W/(W2-W1)×100
W=Weight of sample
W1=Weight of flask
W2=Weight of flask+fat
4. Determination of amino acid content
Amino acids were determined using the methodology proposed by AOAC (AOAC, 2005). The amino acid profile was analyzed using an ion-exchange amino acid analyzer(L-8900, Hitachi, Tokyo, Japan).
5. Mineral content
The mineral content of mineral ash was analyzed by atomic absorption spectrometry using an inductively coupled plasma mass spectrometer (iCAP 7400 ICP-OES Duo, Thermo Fisher Scientific, MA, USA), and the sample was prepared and performed according to the AOAC method (AOAC, 2005).
6. Vitamin analysis
The vitamin content was determined according to the AOAC (AOAC, 2005) and calculated using HPLC (Shiseido Nanospace SI-2, Osaka Soda, Osaka, Japan).
7. Evaluation of fatty acid composition
The fatty acid content was determined according to the AOAC (AOAC, 2005). The analysis was performed using a gas chromatograph (7890A, Agilent Technologies, CA, USA) equipped with a capillary column (SP-2560, Sigma Aldrich, MO, USA).
8. Acidity measurements
To determine acid value, the sample was placed in a conical flask with a stopper, dissolved in a 100 mL mixture of ethanol and ether (1 : 2, v/v), and titrated with a 0.1 N ethanolic potassium hydroxide solution using phenolphthalein solution as an indicator, until a pale red color persisted for 30 seconds.
Acid value (mg/g)=5.611×(a-b)×f×(1/s)
a: Consumption of 0.1 N ethanolic potassium hydroxide solution for the sample (mL)
b: Consumption of 0.1 N ethanolic potassium hydroxide solution for blank test [ethanol : ether mixture (1 : 2) 100 mL] (mL)
f: Titer of 0.1 N ethanolic potassium hydroxide solution (=1.0)
s: Sample collection (g)
9. Analysis of heavy metal residues
Upon our request, the Korea Functional Food Research Institute (Seongnam, Korea) analyzed the heavy metals according to the Restriction of Hazardous Substances in food products listed in the Korean Food Standard Codex of the Ministry of Food and Drug Safety (MFDS, 2019a). To prepare samples for analysis, 5 g of freeze-dried pupae was decomposed by placing it in a microwave digestion system, treated with nitric acid, and filtered using Whatman No. 6 filter paper. The analysis sample was measured using the inductively coupled plasma (ICP) method. Element wavelengths were 208 amu, 111 amu, and 75 amu for lead (Pb), cadmium (Cd), and arsenic (As), respectively. For mercury (Hg) analysis, the sulfuric acid-nitric acid reflux method was used. Briefly, 10 g of freeze-dried drone pupae was added to a decomposition flask; 10 mL water and 20 mL nitric acid were added to the flask and mixed. After resting for some time, 20 mL sulfuric acid was added and the flask was carefully connected to a reflux cooler and heated until no more nitrogen dioxide was produced. When the decomposition solution turned citrous and transparent, it was cooled and 50 mL of water and 10 mL of 10% urea solution were added. The mixture was boiled for 10 min; after cooling, 1 g of potassium permanganate was added and mixed by shaking occasionally for 10 min. When the magenta color disappeared, 1 g of potassium permanganate was added and shaken to mix. This process was repeated until the magenta color permanently remained. The mixture was then carefully titrated with 10% hydrogen peroxide solution until the solution became colorless and transparent. After cooling, the inside and connecting part of the device was washed with 20 mL sulfuric acid (carefully added to the flask to make up the required volume of test solution). A mercury analyzer was used to perform measurements.
10. Statistical analysis
The data were presented as means±SD of three independent experiments and analyzed using SAS enterprise guide 9.2 software (North Carolina, USA). Differences between data sets were assessed by one-way analysis of variance (ANOVA) followed by Tukey’s multiple comparison test. A p-value of <0.05 was considered statistically significant.
RESULTS
1. Optimal time for the production of drone pupae as a temporary food ingredient
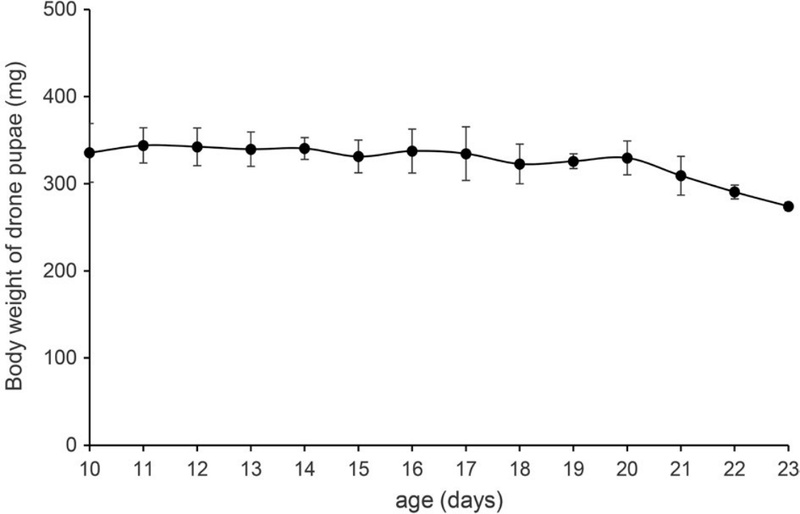
A drone honeybee goes through the egg stage up to day 3 from spawning by the queen bee. The larval stage occurs between days 4-10 and the pupal stage between days 11-23. Pupae do not consume any food and are cut off from the external environment as the honeycomb is sealed with wax. Although pre-pupal stages can be used as a food ingredient, the outer shell is not hard enough to withstand the collecting behavior; therefore, pre- or early pupal stages are easily crushed and the characteristic pupal shape is lost. Hence, it is preferable to collect pupae from day 17 (Fig. 1). However, since pupal weight decreases towards the latter half of this stage, before turning into an imago on day 24, the optimal collection time for food ingredient use is around day 20 (Fig. 2).
2. Establishment of a manufacturing method for temporary use of food ingredients
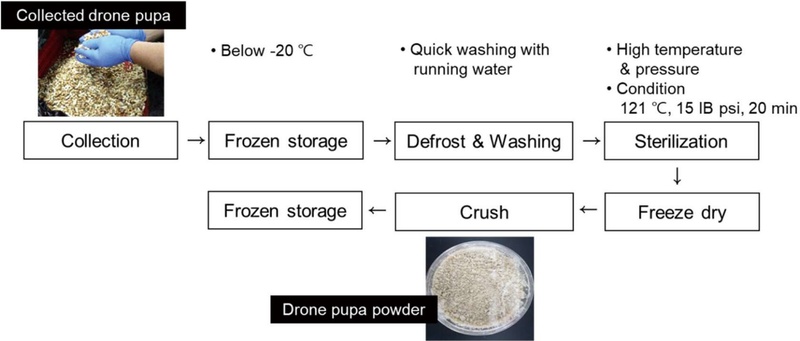
The method of manufacturing drone pupae for food ingredient use was set as standard (Fig. 3). In order to satisfy the acid value criterion, it is important to immediately transfer the collected pupae to a freezer and store them at -20℃ even during transportation. To prevent frozen pupae from contamination, they were placed in a perforated tray and washed quickly with running water (cold or room temperature water) to remove foreign substances. They were air-dried to remove moisture. As a part of the microbial sterilization process, we examined the sterilization status based on the presence of coliform and general bacteria. While no coliform was detected before the sterilization, we found general bacteria in some of the drone pupae. However, after autoclave sterilization, no microorganisms were qualitatively detected in the drone pupae. The standardized conditions of freeze-drying included maintaining freezing room temperature at -35℃ or less and performing quick-freezing for 12 h or more. The vacuum conditions included a vacuum level of 0.5 Torr or less, vacuum period of at least 36 h, and temperature of -15℃ or less. The pupae were slowly dried using a dryer with a temperature of 45℃ or less and left still for 2-3 h. The freeze-dried drone pupae can be easily pulverized and kept frozen for storage.
3. Acidity standard of drone pupae
The acid value criterion is one of the most important factors in using drone pupae as a food ingredient. The main factor affecting the acid value of pupae is temperature change. At room temperature, drone pupae acid value rises significantly, which makes them unsuitable for food ingredient use. Therefore, the acid value standard of drone pupae for use as a food ingredient was determined as 5.0 mg/g or less (Table 1). According to the Korean Food Standards Codex, the acid value of pupae among edible insects is less than 5.0 mg/g.
4. The safety profile of drone pupae
Additional studies were carried out to determine the safety profile of honeybee drone pupae, based on foodborne pathogens and mycotoxins. The same four colonies for the chemical composition study were used for the collection of drone pupae. None of the collected drone pupae contained food poisoning bacteria such as Coliform, Salmonella spp, S. aureus, and enterohemorrhagic Escherichia coli (Table 2).
5. Chemical composition of drone pupae
As shown in Table 3, the ash content in drone pupae were 4.00±0.01%, while the total moisture content was 2.14±1.21%. Moisture is one of the fundamental components of drone pupae and affects its quality and preservation against bacteria and fungi. Carbohydrate, crude fat, and crude protein content of drone pupae were 15.89±0.11%, 26.19±0.05%, and 51.78±0.08%, respectively.
Amino acids, the building blocks for proteins, are crucial components of all cells and tissues. Most notably, essential amino acids are integral to health and must be included in an organism’s diet to maintain physiological functions. As shown in Table 4, drone pupae have all 11 essential amino acids (cysteine, histidine, isoleucine, leucine, lysine, methionine, phenylalanine, threonine, tryptophan, tyrosine, and valine). Drone pupae possess large amounts of glutamic acid (38.28±6.2 g/kg) and leucine (32.02±4.1 g/kg). Therefore, given the indispensable amino acid requirements for a human adult (FAO, 2023), drone pupae can provide an adequate amount of essential amino acids for human consumption.
Micronutrients such as minerals and vitamins are required in relatively small quantities. Minerals play an important role in growth, bone health, and fluid balance, while vitamins are necessary for energy production, immune function, and blood clotting. Drone pupae were found to contain 13 minerals; total potassium and phosphorus contents were 1438.74±1.12 mg/100 g and 714.00±2.44 mg/100 g, respectively (Table 5). Since drone pupae are unable to produce minerals, it is assumed that most minerals are derived from the honey and pollen that drone pupae feed on. Furthermore, drone pupae contained four water-soluble vitamins (B1, B2, C, and folate) and one fat-soluble vitamin (D) (Table 6). As shown in Table 7, it is evident that saturated and unsaturated fatty acid content is moderately different in drone pupae. Total saturated fatty acid content was 13.20±0.03 g/100 g, while total unsaturated fatty acid content was 10.90±0.01 g/100 g. According to Table 8, the main unsaturated fatty acid component found in oil extracted from larvae was oleic acid (10.22±0.2 g/100 g), followed by linolenic acid (0.27±0.06 g/100 g), linoleic acid (0.15±0.03 g/100 g) and palmitoleic acid (0.12±0.01 g/100 g). Likewise, the main saturated fatty acid was palmitic acid (9.13±0.08 g/100 g), followed by stearic acid (2.90±0.01 g/100 g), myristic acid (0.59±0.01 g/100 g) and arachidic acid (0.16±0.0 g/100 g).
6. Heavy metal standards
The amount of residual heavy metals in beekeeping products such as honey and royal jelly has not yet been defined. However, the Korean Food Standards Codex provides the following threshold standards for heavy metals in edible insects: 0.1 mg/kg or less lead, 0.05 mg/kg or less cadmium, and 0.1 mg/kg or less arsenic for mealworm larvae; 0.3 mg/kg or less lead and 0.3mg/kg or less cadmium for Gryllus bimaculatus and Korean rhinoceros beetle larvae; 0.3 mg/kg or less lead, 0.05 mg/kg or less cadmium, and 0.1 mg/kg or less arsenic for whitespotted flower chaffer beetle larvae (MFDS, 2019a). Among the heavy metals that enter the human body via food products, typical hazardous heavy metals regulated by the government due to their high degree of harmfulness include lead, cadmium, mercury, and arsenic (MFDS, 2019a). Edible insects are bred indoors in accordance with the ‘Insect Industry Promotion and Support Act’, which established safe breeding standards such as food source and breeding environment to prevent contamination by harmful substances such as heavy metals.
We analyzed the heavy metal content of drone pupae and identified an infinitesimal amount of cadmium (0.001 mg/kg or less) in all pupae (Table 9). Very small amounts of mercury (0.003 mg/kg or less), lead (0.02 mg/kg), and arsenic (0.017 mg/kg or less) were also present. Table 9 shows the heavy metal standards for drone pupae. Since detected values of heavy metals in these drone pupae were less than the standard values for edible insects, we could demonstrate that drone pupae are a very safe food product. Table 9 shows the standard heavy metal content in drone pupae of honeybees for use as food ingredients.
DISCUSSION
Our study suggests that drone pupae, which have a high nutritional value, are safe food ingredients. There are several reasons for using insects as food or alternative sources of protein. Insects have a high feed conversion rate and fast production of protein mass compared with animals, which are a conventional protein source. Unlike livestock, insects gain weight faster, produce fewer greenhouse gases as they grow, use less water, and are less soil-dependent (Sere et al., 2018). As such, a study by Oonincx et al. concluded that insects could serve as a more environment-friendly alternative for the production of animal protein, with respect to greenhouse gas emissions (Oonincx et al., 2010). Little is known about the microbiological hazards associated with edible insect consumption (Grabowski and Klein, 2017). Insect consumption could potentially ameliorate diet-related public health problems (Payne et al., 2016); however, nutrient bioavailability in edible insects needs to be further investigated for sustainable human consumption. Insects have been shown to have a balanced essential amino acid composition. We found that drone pupae contain all essential amino acids with relatively large amounts of leucine, lysine, and valine. Due to their high protein content and digestibility, edible insects cannot be replaced by other nutrients. Crude protein content in insects was found to be within the range of 40-75 g/100 g in dry matter, which is comparable to the crude protein content in common commodities of animal origin (Adamkova et al., 2017).
As an energy source, fat content exhibits great variability among edible insects. The giant mealworm larva reared on the Island of Sumatra recorded a high fat content of 35% (Adamkova et al., 2017). The measured value of 36% for T. molitor larva in the current study is in accordance with that of the referenced larva (Ghosh et al., 2017) and may be a suitable source of nutritional energy. Our study demonstrates the potential of edible insects to provide specific micronutrients such as potassium, magnesium, and phosphorus.
Acknowledgments
This research was funded by the Research Program for Agricultural Science and Technology Development (grant number PJ01512902), National Institute of Agricultural Sciences, Rural Development Administration, Republic of Korea.
References
-
Adamkova, A., J. Mlcek, L. Kourimska, M. Borkovcova, T. Busina, M. Adamek, M. Bednarova and J. Krajsa. 2017. Nutritional potential of selected insect species reared on the island of sumatra. Int. J. Environ. Res. Public Health 14(5): 541.
[https://doi.org/10.3390/ijerph14050521]

- Association of Official Analytical Chemists (AOAC). 2005. Official methods of analysis of AOAC international. AOAC, Washington, DC, USA.
- Food and Agriculture Organization (FAO). 2023. The state of food security and nutrition in the world 2023. FAO, Rome. Italy.
-
Ghosh, S., S. M. Lee, C. Jung and V. B. Meyer-Rochow. 2017. Nutritional composition of five commercial edible insects in South Korea. J. Asia-Pac. Entomol. 20(2): 686-694.
[https://doi.org/10.1016/j.aspen.2017.04.003]

-
Grabowski, N. T. and G. Klein. 2017. Bacteria encountered in raw insect, spider, scorpion, and centipede taxa including edible species, and their significance from the food hygiene point of view. Trends Food Sci. Technol. 63: 80-90.
[https://doi.org/10.1016/j.tifs.2017.01.007]

- International Organization for Standardization (ISO). 1999. Microbiology of food and animal feeding stuffs-Horizontal method for the enumeration of coagulase-positive staphylococci (Staphylococcus aureus and other species). ISO 6888-1:1999. ISO. Geneva, Switzerland.
- International Organization for Standardization (ISO). 2002. Microbiology of food and animal feeding stuffs. Horizontal method for the detection of Salmonella spp. ISO 6579: 2002. ISO. Geneva, Switzerland.
- International Organization for Standardization (ISO). 2003. Foodstuffs-determination of aflatoxin b1, and the total content of aflatoxins b1, b2, g1 and g2 in cereals, nuts and derived products-high-performance liquid chromatographic method. ISO 16050:2003. ISO. Geneva, Switzerland.
- International Organization for Standardization (ISO). 2006. Microbiology of food and animal feeding stuffs - horizontal method for the enumeration of coliforms - colony-count technique. ISO 4832:2006. ISO, Geneva, Switzerland.
- International Organization for Standardization (ISO). 2009. Microbiology of food and animal feeding stuffs-Guidelines for the estimation of measurement uncertainty for quantitative determinations. ISO/TS 19036:2006. ISO. Geneva, Switzerland.
- International Organization for Standardization (ISO). 2018. Cereals and cereal products-determination of ochratoxin a-high performance liquid chromatographic method with immunoaffinity column cleanup and fluorescence detection. ISO 15141:2018. ISO. Geneva, Switzerland.
-
Melgar-Lalanne, G., A-J. Hernández-Ãlvarez and A. Salinas-Castro. 2019. Edible insects processing: Traditional and innovative technologies. Compr. Rev. Food Sci. Food Saf. 18(4): 1166-1191.
[https://doi.org/10.1111/1541-4337.12463]

- Ministry of Food and Drug Safety (MFDS). 2019a. Food code, Annex 1 The list of raw materials allowed in food. MFDS, Cheongwon, Korea.
- Ministry of Food and Drug Safety (MFDS). 2019b. Food Sanitation Act. MFDS, Cheongju, Korea.
- Ministry of Food and Drug Safety & Rural Development Administration. (MFDS & RDA). 2020. Drone pupae approved as a new food ingredient. Press release, MFDS & RDA, Cheongju/Jeonju, Korea, July 9.
-
Omuse, E. R., H. E. Z. Tonnang, A. A. Yusuf, H. Machekano, J. P. Egonyu, E. Kimathi, S. F. Mohamed, M. Kassie, S. Subramanian, J. Onditi, S. Mwangi, S. Ekesi and S. Niassy. 2024. The global atlas of edible insects: Analysis of diversity and commonality contributing to food systems and sustainability. Sci. Rep. 14: 5045.
[https://doi.org/10.1038/s41598-024-55603-7]

-
Oonincx, D. G. A. B., J. Van Itterbeeck, M. J. W. Heetkamp, H. Van Den Brand, J. J. A. Van Loon and A. Van Huis. 2010. An exploration on greenhouse gas and ammonia production by insect species suitable for animal or human consumption. PLoS One 5(12): e14445.
[https://doi.org/10.1371/journal.pone.0014445]

-
Pascari, X., S. Gil-Samarra, S. Marín, A. Ramos and V. Sanchis. 2019. Fate of zearalenone, deoxynivalenol and deoxynivalenol-3-glucoside during malting process. LWT-Food Sci. Technol. 99: 540-546.
[https://doi.org/10.1016/j.lwt.2018.10.030]

-
Payne, C. L. R., P. Scarborough, M. Rayner and K. Nonaka. 2016. Are edible insects more or less ‘healthy’ than commonly consumed meats? A comparison using two nutrient profiling models developed to combat over- and undernutrition. Eur. J. Clin. Nutr. 70(3): 285-291.
[https://doi.org/10.1038/ejcn.2015.149]

-
Piacentini, K. C., G. D. Savi, G. Olivo and V. M. Scussel. 2015. Quality and occurrence of deoxynivalenol and fumonisins in craft beer. Food Control 50: 925-929.
[https://doi.org/10.1016/j.foodcont.2014.10.038]

-
Sere, A., A. Bougma, J. T. Ouilly, M. Traore, H. Sangare, A. M. Lykke, A. Ouedraogo, O. Gnankine and I. H. N. Bassole. 2018. Traditional knowledge regarding edible insects in burkina faso. J. Ethnobiol. Ethnomed. 14: 59.
[https://doi.org/10.1186/s13002-018-0258-z]

-
Smetana, S. 2023. Circulartiy and environmental impact of edible insects. J. Insects Food Feed 9(3): 1111-1124.
[https://doi.org/10.3920/JIFF2023.x004]

-
Tian, L. and H. M. Hines. 2018. Morphological characterization and staging of bumble bee pupae. PeerJ 6: e6089.
[https://doi.org/10.7717/peerj.6089]

- United Nations (UN). 2022. World population prospects 2022: Summary of results. United Nations, New York, USA.
-
Xi, X. Z., S. S. Guo, H. Guo, X. W. Cui, H. Cao, F. X. Xu, M. M. Zheng, Y. J. Li and C. C. Han. 2018. Anti-exercise-fatigue and promotion of sexual interest activity of total flavonoids from wasps drone-pupae in male mice. Biomed. Pharmacother. 107: 254-261.
[https://doi.org/10.1016/j.biopha.2018.07.172]

-
Zhou, J. H., Y. T. Qi, Y. L. Hou, J. Zhao, Y. Li, X. F. Xue, L. M. Wu, J. Z. Zhang and F. Chen. 2011. Quantitative determination of juvenile hormone iii and 20-hydroxyecdysone in queen larvae and drone pupae of Apis mellifera by ultrasonic-assisted extraction and liquid chromatography with electrospray ionization tandem mass spectrometry. J. Chromatogr. B-Analyt. Technol. Biomedi. Life Sci. 879(25-26): 2533-2541.
[https://doi.org/10.1016/j.jchromb.2011.07.006]