
Comparative Analysis of Morphological Characteristics among Subspecies of Honey Bee (Apis mellifera) in Different Regions of Eastern Uzbekistan
Abstract
Identifying the subspecies of honey bees (Apis mellifera) and understanding their morphological characteristics is essential for improving colony productivity, particularly in regions with suboptimal yields. This study aimed to determine the subspecies distribution and compare morphometric traits among honey bees managed in 6 regions of eastern Uzbekistan. A total of 3,200 workers were collected from 32 apiaries and analyzed. Subspecies classification was conducted using 19 wing landmarks processed through IdentiFly software, and key morphological traits-including body length, head width, thorax width, lengths and widths of fore and hindwings, as well as the tarsus were measured using ImageJ. 9 morphotypes presumed to belong to known Apis mellifera subspecies were inferred based on wing morphometric analysis, with A. m. remipes (26.7%), A. m. carpatica (24.2%), and A. m. caucasica (17.4%) being the most prevalent. Statistical analysis revealed significant differences among subspecies for all traits except body length. Notably, A. m. cecropia exhibited the largest values in all wing-related parameters. Given the established link between wing size and foraging capacity, the presence of high-performing subspecies such as A. m. cecropia suggests potential for enhancing apicultural productivity in Uzbekistan. The findings of this study provide baseline data on subspecies diversity and morphological variation in the region and may support future breeding strategies tailored to local environmental conditions and production goals.
Keywords:
Honey bee subspecies, Morphometrics, Morphological characteristics, Beekeeping, UzbekistanINTRODUCTION
Beekeeping, which relies on the management of honey bees, has sustained a long standing and evolving relationship with human civilization for over 10,000 years (Ruttner, 1988; Crane, 1990). Although it centered historically on honey production, modern apiculture has diversified significantly alongside scientific and technological advancements. The beekeeping industry has expanded from a primary focus on honey production to include diverse activities such as processing bee products-royal jelly, propolis, beeswax, and venom- and providing pollination services, thereby enhancing agricultural productivity and farm income (Lee et al., 2010; Jeong et al., 2016; Kang et al., 2024). Honey bees are vital pollinators for numerous wild plant species and cultivated crops, facilitating plant reproduction and thereby playing a critical role in maintaining ecosystem stability and enhancing food security on a global scale (Jung, 2008; Aryal et al., 2020; Papa et al., 2022; Jung and Shin, 2022). As a result of their ecological and agricultural significance, honey bees are actively managed worldwide not only for their pollination services but also for the economic value of their hive products. Apiculture thus serves as both a tool for biodiversity conservation and a means of income generation for beekeepers (Jung, 2008; Garibaldi et al., 2017; Minaud et al., 2024). Globally, an estimated 75% of the top 100 food crops rely, at least in part, on pollinators, and approximately 30% of total agricultural production is dependent on pollination (Klein et al., 2007; Jung, 2008). The economic value of pollination by honey bees has attracted global attention; for instance, in the United States, it has been estimated to exceed the value of direct hive products by more than 140-fold (Levin, 1983). These findings underscore the essential role of honey bees in sustaining both natural ecosystems and global agriculture.
The western honey bee (Apis mellifera) is distributed across a wide range of climates globally and has diversified into numerous subspecies (Ruttner, 1988; Meixner et al., 2013). Based on morphological traits, mitochondrial DNA, and nuclear genetic markers, 33 subspecies have been identified and are grouped into 5 major evolutionary lineages: M (Western and Northern Europe), C (Southern and Eastern Europe), A (Africa), O (Middle East and Central Asia), and Y (Arabian Peninsula) (Ruttner, 1988; Ilyasov et al., 2020). Among the available techniques, morphometric approaches remain cost-effective and robust tools for distinguishing subspecies and conducting comparisons within species. Geometric morphometric analysis of wing venation and body structures has proven to be especially effective (Oleksa and Tofilski, 2015). These morphometric methods for subspecies identification can be broadly classified into two main categories: the measurement of body parts and the analysis of wing shape. The first category, known as traditional morphometry, involves the assessment of 36 morphometric traits related to the body (Ruttner, 1988). In contrast, wing shape analysis includes several approaches. The first is DuPraw’s method, which measures 11 angles formed between 18 intersections of wing veins to detect shape variation (DuPraw, 1965). The second is the DAWINO method, which builds upon DuPraw’s approach by adding seven linear measurements, five indices, and one area measurement. The third method is geometric morphometrics, which uses multivariate statistical techniques to analyze landmark coordinates derived from the wing venation pattern (Miguel et al., 2011; Ilyasov et al., 2020). Accurate identification of honey bee subspecies is critical not only for conserving biodiversity but also for ensuring the sustainability of apiculture. Subspecies may differ significantly in various biological traits such as pesticide susceptibility, gut microbiota composition, and thermal tolerance (Suchail et al., 2000; Alattal and Alghamdi, 2015). Therefore, reliable subspecies classification and trait based breeding strategies can play a crucial role in improving beekeeping practices.
Uzbekistan is the most populous landlocked country in Central Asia, with approximately 36.36 million people in 2024 (UN, 2024). The country exhibits a typical continental climate, characterized by dryness and large daily temperature fluctuations (Qoshmatovich, 2022). Beekeeping is an important agricultural industry in Uzbekistan, contributing to the yield of various pollinator-dependent crops such as alfalfa, sunflower, and cotton (Jamolov et al., 2022). In response to its importance, a national program to promote beekeeping was launched in 2017. As a result, the number of honeybee colonies increased from approximately 190,000 in 2011 to 490,000 in 2019 (Djurabaev and Rashidov, 2021; Jamolov et al., 2022; Kwon et al., 2024). Despite such governmental efforts, research on apiculture remains limited, and in 2018, Uzbekistan produced approximately 12,578 tons of honey, ranking 27th globally total honey production (FAO, 2018). To improve productivity, advancements in hive management and breeding strategies are necessary. Larger-bodied honey bees have been shown to fly farther, collect more pollen, and produce more honey (Greenleaf et al., 2007; Ramello et al., 2024). Therefore, identifying the distribution of honey bee subspecies in Uzbekistan and analyzing their morphological characteristics will be essential for selecting superior breeding stock to enhance apicultural productivity.
This study aimed to (1) identify the subspecies of honey bees managed in Uzbekistan and (2) analyze regional variation in morphological traits of the subspecies. The findings are expected to provide a foundation for future breeding programs aimed at improving apicultural productivity in Uzbekistan.
MATERIALS AND METHODS
1. Honey bee sampling
Sampling was conducted from July 28 to August 28, 2022, in 6 Uzbek regions: Yanghikurgan (n=4), Tashkent (n=5), Sukok (n=6), Komarovka (n=5), Gagarin (n=6), and Angren (n=6), total 32 apiaries. From each apiary, 30 worker bees were collected from 5 colonies without considering age. Samples were preserved in 70% ethanol for subsequent analysis.
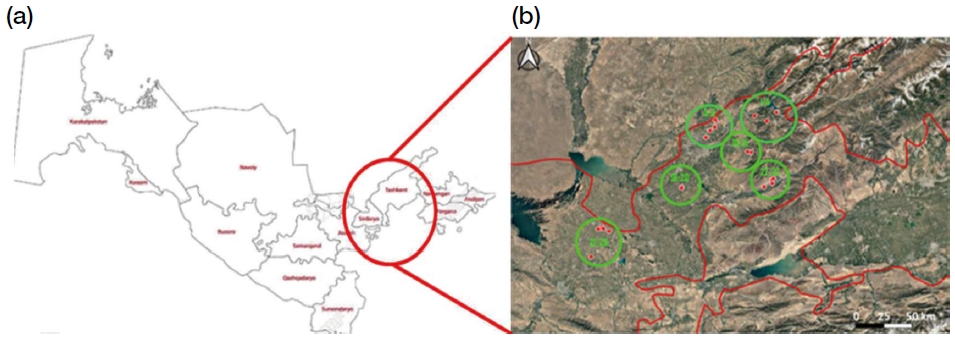
(a) Overview map of Uzbekistan highlighting the eastern region where honey bee samples were collected. (b) Enlarged satellite view displaying the distribution of 32 sampled apiaries across six distinct regions in eastern Uzbekistan, denoted by green circles (Region 1: sites 1-4, Region 2: sites 5-9, Region 3: sites 10-15, Region 4: sites 16-20, Region 5: sites 21-26, Region 6: sites 27-32). Each red dot corresponds to a specific apiary location.
2. Identification of honey bee subspecies and measurement of external morphology
To identify honey bee subspecies, 20 workers were randomly selected from each colony, and photographs of their right forewings were taken. The wings were mounted under a stereomicroscope (KS-208, KOREA LABTECH, Korea) and photographed at 6× magnification. Subspecies identification was performed using the IdentiFly software (Version 1.6.2) (Meixner et al., 2013; Nawrocka et al., 2017; Salehi and Nazemi-Rafie, 2020). Each of the 3,200 workers was identified individually, and subspecies distribution was assessed based on the cumulative results of all identified individuals, rather than on a colony-level basis. The IdentiFly software identifies 20 subspecies from 4 evolutionary lineages (A, C, O, and M) based on 19 geometric morphometric landmarks on the forewing (Dilday, 2022).
After subspecies identification, the right hind wings and hind legs of the same bees were dissected and photographed using a stereomicroscope, as was done for the forewings. Each anatomical structure was mounted and imaged at 6× magnification alongside a 1 cm scale grid. Morphometric measurements-including head width, thorax width, forewing length and width, hind wing length and width, and hind tibia length and width-were obtained from the images using ImageJ software (Java version 1.8), based on the 1 cm reference grid.
3. Data analysis
The distribution of honey bee subspecies in Uzbekistan was summarized using descriptive statistics and expressed as percentages. To compare the morphometric characteristics of the dominant subspecies, normality was first assessed using the Shapiro-Wilk test, followed by one-way analysis of variance (ANOVA) at a 95% confidence level. Post hoc comparisons among colonies were conducted using Tukey’s honestly significant difference (HSD) test. All statistical analyses were performed using R Studio software (Version 4.3.2).
RESULTS
1. Subspecies composition
A total of 9 Apis mellifera subspecies were identified from samples collected at 32 apiaries across 6 regions of eastern Uzbekistan. The subspecies composition of honey bees varied across the surveyed locations. Among all identified groups, A. m. remipes was the most prevalent, accounting for 26.7% overall, with particularly high proportions in Sukok (37.9%) and Komarovka (31.1%). A. m. carpatica ranked second (24.2%), showing major representation in Gagarin (29.6%) and Tashkent (28.6%). A. m. caucasica followed with 17.4%, with notable occurrences in both Tashkent and Angren (21.3% each). Other subspecies-A. m. carnica, A. m. ligustica, A. m. syriaca, A. m. mellifera, A. m. ruttneri, and A. m. cecropia-were detected in lower frequencies and showed more localized distributions.
2. Morphological comparison between subspecies
Morphological traits including body length, head width, thorax width, forewing length and width, hindwing length and width, and metatarsus length and width were analyzed for 9 Apis mellifera subspecies identified from eastern Uzbekistan. Among the traits examined, all except body length showed statistically significant differences among subspecies (p<0.05; Table 2). The longest body length was observed in A. m. cecropia (13.43±1.228 mm), while A. m. ruttneri showed the shortest value (13.04±1.139 mm). However, these differences were not statistically significant (F=1.336, p>0.05). Head width varied significantly across subspecies (F=2.143, p<0.05), with A. m. carnica and A. m. ligustica exhibiting relatively broader heads (3.76±0.235 mm and 3.76±0.261 mm, respectively), whereas A. m. caucasica had the narrowest mean head width (3.71±0.242 mm). Thorax width also differed significantly (F=3.051, p<0.05), with A. m. syriaca having the broadest thorax (3.90±0.427 mm), while A. m. caucasica again showed the smallest measurement (3.77±0.453 mm). In terms of wing morphology, A. m. cecropia consistently displayed the greatest forewing and hindwing dimensions among the subspecies. The shortest forewing length was found in A. m. caucasica (8.92±0.436 mm), and the narrowest forewing width was in A. m. mellifera (2.93±0.257 mm). Hindwing length was also smallest in A. m. caucasica (6.25±0.317 mm), while A. m. ruttneri had the narrowest hindwing width (1.71±0.148 mm). Tarsus length was highest in A. m. syriaca (2.04±0.157 mm) and lowest in A. m. caucasica (1.99±0.151 mm). In terms of metatarsus width, A. m. carnica showed the largest value (1.13±0.138 mm), whereas A. m. carpatica had the smallest (1.10±0.130 mm).
DISCUSSION
This study investigated the distribution and morphometric variations among honey bee (Apis mellifera) subspecies managed in 6 regions of eastern Uzbekistan. Out of 9 identified subspecies, A. m. remipes (syn. Armenica) was predominant, followed by A. m. carpatica and A. m. caucasica. Morphometric analyses revealed significant differences among subspecies for head width, thorax width, lengths and widths of fore- and hindwings, and leg tarsus, excluding only body length. Such variations are consistent with previous studies, which suggested they are influenced by genetic backgrounds and regional environmental factors (Ruttner, 1988; Ilyasov et al., 2020).
In this study, distinct regional patterns were observed in the prevalence of Apis mellifera subspecies across Uzbekistan. The Yanghikurgan population exhibited a relatively balanced presence of four subspecies: A. m. remipes, A. m. carpatica, A. m. caucasica, and A. m. carnica. In Tashkent, A. m. carpatica and A. m. caucasica were predominant, while in Sukok and Komarovka, A. m. remipes was the dominant subspecies. Similarly, in Gagarin, A. m. remipes was the most common, followed by similar proportions of A. m. carpatica and A. m. carnica. In Angren, A. m. remipes also dominated, with A. m. carpatica and A. m. caucasica appearing at comparable levels. A. m. remipes is considered native to the southern Caucasus region, particularly Armenia and Anatolia (Ruttner, 1988). While no detailed studies have specified its altitudinal preferences, its distribution ranges from lowland plains to montane meadows and temperate highlands, indicating a broad ecological tolerance (Kirpik et al., 2010). A. m. carpatica is primarily associated with the Carpathian Mountains of Romania and Moldova and is assumed to be adapted to a wide range of altitudes, approximately 300-2,000 meters above sea level (Ruttner, 1988). Its presence in Uzbekistan has also been documented previously (Kakhramanov et al., 2021). A. m. caucasica, originating from the central Caucasus Mountain regions, is known to occur in Armenia, Azerbaijan, Georgia, the North Caucasus of Russia, northeastern Türkiye, and parts of northwestern Iran (Janashia et al., 2025). A. m. carnica, one of the most widely utilized commercial subspecies, is distributed across Slovenia, southern Austria, Croatia, Bosnia and Herzegovina, Serbia, Hungary, Romania, and Bulgaria (Ilyasov et al., 2020; Balazs et al., 2025). Like A. m. carpatica, A. m. carnica has also been reported in Uzbekistan (Kakhramanov et al., 2021). Globally, Apis mellifera has recently been reclassified into 33 subspecies belonging to five evolutionary lineages, with Central Asia primarily falling under lineages C and O (Ilyasov et al., 2020). The findings of this study are consistent with that classification, as the dominant subspecies identified in Uzbekistan correspond to the C and O lineages.
Morphometric studies of A. m. remipes remain limited. Ruttner (1988) reported a forewing length of 9.068±0.144 mm, which is not substantially different from the value obtained in the present study (9.01±0.442 mm). In the case of A. m. carpatica, morphological and biological differences from A. m. carnica were first described in the early 20th century (Gaini, 1923; Safonkin et al., 2019). Later, A. m. carpatica was recognized as a distinct subspecies rather than a subgroup of A. m. carnica (Zinov’eva et al., 2013). According to Mannapov et al. (2015), the Standard ‘Maikopski’-a reference line established to evaluate the morpho-ecological consistency and breed-specific features of A. m. carpatica-shows a forewing length ranging from 9.27 to 9.63 mm. However, the mean forewing length measured in the present study was 9.00±0.466 mm, which is notably shorter. This may indicate a partial deviation from the standard morphotype of the subspecies. Deviations in morphological traits from expected values can result from hybridization (Kirpik et al., 2010). In a study by Frunze et al. (2020), differences in forewing length within the same subspecies were attributed to uncontrolled mating. A similar pattern was also observed in A. m. caucasica. Significant morphological differences were observed among subspecies across nearly all traits measured, including head and thorax width, lengths and widths of both forewings and hindwings, and the tarsus. Only total body length did not differ significantly across groups. Notably, A. m. caucasica consistently exhibited the smallest values in most traits, which aligns with previous findings that this subspecies, although smaller in body size, is characterized by a relatively elongated proboscis (Ruttner, 1988; Karakaş, 2013).
All 9 A. mellifera subspecies identified in this study were detected across all 6 regions of eastern Uzbekistan, suggesting a high potential for natural hybridization. Typically, honey bee subspecies develop distinct morphological and genetic traits through long-term geographic isolation imposed by physical barriers such as mountain ranges, deserts, and oceans (Ruttner, 1988; Sheppard and Meixner, 2003). However, interbreeding remains feasible when these populations come into contact, either through human-mediated colony movements or natural proximity between regions. Ruttner (1988) classified approximately 26 A. mellifera subspecies as “geographic races” of a single interbreeding species, emphasizing their taxonomic unity. Since these subspecies are reproductively compatible, the co-occurrence of multiple lineages within Uzbekistan may reflect active or historical admixture. Additionally, the introduction of non-native subspecies from neighboring countries likely contributes to the observed diversity. For instance, Nuralieva et al. (2023) reported that beekeepers in Kazakhstan-Uzbekistan’s northern neighbor-imported colonies from diverse regions including the Caucasus, Ukraine, Russia, and Uzbekistan itself. Although these foreign lineages were incorporated in relatively small proportions, their presence supports the possibility of cross-regional gene flow. Collectively, these findings provide a plausible explanation for the mixed distribution of subspecies observed in the present study and point to a high level of hybridization occurring both naturally and anthropogenically in the region. While the use of geometric morphometric analysis provides an effective and cost-efficient method for subspecies inference, it may not yield definitive taxonomic classification. In future studies, we need to incorporate molecular markers and phylogenetic tools to verify and strengthen the morphometric assignments presented here.
Our findings indicate that A. m. cecropia possesses the largest wing dimensions-both in length and width-among the subspecies analyzed in this study. Previous research has demonstrated that wing size in honey bees is closely associated with flight efficiency and foraging ability, with larger wings generally enabling longer-distance travel to floral resources and more effective collection of nectar and pollen (Greenleaf et al., 2007; Dadgostar, 2020). Moreover, wing damage has been shown to significantly impair flight performance and reduce foraging activity, occasionally leading bees to cease foraging altogether (Higginson et al., 2011). These findings collectively suggest that large and intact wings may serve as an important morphological trait that enhances foraging efficiency at the colony level. In this context, Uzbekistan’s relatively low honey production-which ranks 27th globally according to FAO (2018)-may not be solely attributable to limitations in beekeeping practices or environmental factors. The low prevalence of A. m. cecropia, a subspecies characterized by wing morphology potentially advantageous for foraging, could also be a contributing factor. However, it is important to acknowledge that our study did not empirically evaluate the direct relationship between the wing size of A. m. cecropia and actual productivity metrics. Therefore, such interpretations should be regarded as hypothetical and warrant further investigation. Nevertheless, considering its favorable morphological traits and presumed productivity potential, A. m. cecropia may represent a promising candidate for future queen breeding programs aimed at enhancing colony performance and honey yield.
A comparison between the honey bees collected in Uzbekistan and those reported in Korea by Frunze et al. (2022) revealed that Korean bees exhibited larger values across all measured morphometric traits (Table 3). However, it is important to note that the Korean data in that study were not obtained from general apiaries, but from genetically purebred lines maintained at the National Institute of Agricultural Sciences. These colonies were preserved under artificial selection through controlled breeding, rather than under field conditions. As such, the observed size differences may not reflect environmental effects alone but could result from long-term selective pressure and body size stabilization associated with line breeding. According to FAO (2018), honey production in Korea is higher than in Uzbekistan. While multiple factors such as equipment, forage availability, and management practices may also influence productivity, the larger body size of bees in Korean apiaries suggests a potential contribution of morphometric advantage to performance. Thus, the comparatively smaller average body size of Uzbek bees may partially explain differences in productivity. These findings may serve as valuable baseline information for shaping future breeding strategies in Uzbekistan, especially regarding the selection and improvement of local Apis mellifera stocks.
Acknowledgments
This study was funded by the ODA project, Smart AI UZ (NRF-2022K1A3A9A05036394) from NRF, Korea.
References
- Alattal, Y. and A. AlGhamdi. 2015. Impact of temperature extremes on survival of indigenous and exotic honey bee subspecies, Apis mellifera, under desert and semiarid climates. Bull. Insectol. 68(2): 219-222.
-
Aryal, S., S. Ghosh and C. Jung. 2020. Ecosystem services of honey bees; regulating, provisioning and cultural functions. J. Apic. 35(2): 119-128.
[https://doi.org/10.17519/apiculture.2020.06.35.2.119]

-
Balazs, R., T. G. Molnar, E. Edvine Meleg, A. Hidas, E. Zajacz, T. Racz and N. Palinkas-Bodzsar. 2025. Evolutionary Origin and Genetic Diversity of the Pannonian Ecotype of Apis mellifera carnica Colonies in Hungary Based on Mitochondrial DNA and Microsatellite Markers. Biology 14(5): 475.
[https://doi.org/10.3390/biology14050475]

- Crane, E. 1990. Bees and Beekeeping: Science, Practice, and World Resources. Oxford: Heinemann Newnes. 614pp.
- Dadgostar, S. 2020. Wing characters for morphological study on the honey bee (Apis mellifera L.) populations among six provinces of Iran. Arthropods 9(4): 129.
- Dilday, S. E. 2022. Using morphometric analysis of honey bee (Apis mellifera) colonies to identify lineages and subspecies on the isolated island of Newfoundland (Doctoral dissertation, Memorial University of Newfoundland).
-
Djurabaev, O. D. and J. K. Rashidov. 2021. The main directions of effective management and development of the beekeeping industry. In: E3S Web of Conferences. EDP Sciences 282: 02002.
[https://doi.org/10.1051/e3sconf/202128202002]

-
DuPraw, E. J. 1965. The recognition and handling of honeybee specimens in non-Linnean taxonomy. J. Apic. Res. 4: 71-84.
[https://doi.org/10.1080/00218839.1965.11100107]

- FAO (Food and Agriculture Organization). 2018. Non-wood forest products: Honey and beeswax production statistics. https://www.fao.org/forestry/nwfp/statistics/honey-and-beeswax/en, .
-
Frunze, O., D. W. Kim, E. J. Kang, K. Kim, B. S. Park and Y. S. Choi. 2022. The accuracy of morphometric characteristic analysis depends on the type of the assessed traits of honey bees (Apis cerana F. and Apis mellifera L.). J. Asia-Pac. Entomol. 25(4): 101991.
[https://doi.org/10.1016/j.aspen.2022.101991]

-
Frunze, O., Y. S. Сhoi, D. W. Kim, B. S. Park, H. G. Park and E. J. Kang. 2020. Morphometric Characterization of Honey Bee, Apis mellifera Linnaeus, Inbred Lines in Korea. Korean J. Appl. Entomol. 59(4): 371-382.
[https://doi.org/10.5656/KSAE.2020.10.0.056]

- Gaini, M. 1923. Beekeeping in Carpathian Rus, Uzhhorod. http://bdjilka.in.ua/index.php/ru/component/content/article/79-pchely/90-istoriya-vozniknoveniya-karpatskojporodyna-zakarpate, .
-
Garibaldi, L. A., F. Requier, O. Rollin and G. K. Andersson. 2017. Towards an integrated species and habitat management of crop pollination. Curr. Opin. Insect Sci. 21: 105-114.
[https://doi.org/10.1016/j.cois.2017.05.016]

-
Greenleaf, S. S., N. M. Williams, R. Winfree and C. Kremen. 2007. Bee foraging ranges and their relationship to body size. Oecologia 153(3): 589-596.
[https://doi.org/10.1007/s00442-007-0752-9]

-
Higginson, A. D., C. J. Barnard, A. Tofilski, L. Medina and F. Ratnieks. 2011. Experimental wing damage affects foraging effort and foraging distance in honeybees Apis mellifera. Psyche: J. Entomol. 7(6): 419793.
[https://doi.org/10.1155/2011/419793]

-
Ilyasov, R. A., M. L. Lee, J. I. Takahashi, H. W. Kwon and A. G. Nikolenko. 2020. A revision of subspecies structure of western honey bee Apis mellifera. Saudi J. Biol. Sci. 27(12): 3615-3621.
[https://doi.org/10.1016/j.sjbs.2020.08.001]

- Jamolov, R., D. M. Xatamova and M. A. Xolmatova. 2022. Asalarichilik va uning ahamiyati. Oriens Renaissance 2(1): 644-649.
-
Janashia, I., L. Westover and G. Japoshvili. 2025. A review of Apis mellifera caucasica (Hym., Apidae): History, taxonomy and distribution. J. Insect Biodivers. Syst. 11(2): 455-468.
[https://doi.org/10.61186/jibs.11.2.455]

-
Jeong, S., C. Lee, D. Kim and C. Jung. 2016. Questionnaire study on the overwintering success and pest management of honeybee and damage assessment of Vespa hornets in Korea. Korean J. Apic. 31(3): 201-210.
[https://doi.org/10.17519/apiculture.2016.09.31.3.201]

- Jung, C. 2008. Economic value of honeybee pollination on major fruit and vegetable crop in Korea. Korean J. Apic. 23: 147-152.
- Jung, C. and J. H. Shin. 2022. Evaluation of crop production increase through insect pollination service in Korean agriculture. Korean J. Appl. Entomol. 61: 229-238.
-
Kakhramanov, B., S. Isamukhamedov, F. Kuldasheva, S. Doniyorov and N. Rakhimjanova. 2021. Breeding indicators of Carniolan (Apis mellifera carnica Pollm) and Carpathy (Apis mellifera carpatica) honey bees. In E3S Web of Conferences. EDP Sciences 244: 02008.
[https://doi.org/10.1051/e3sconf/202124402008]

-
Kang, Y., S. H. Kwon and C. Jung. 2024. Correlation Analysis between Honeybee Pest Occurrences and Winter Failure Based on the Questionnaire from Beekeepers in Korea. Korean J. Apic. 39(3): 185-193.
[https://doi.org/10.17519/apiculture.2024.09.39.3.185]

- Karakaş, E. G. 2013. Genetic Impact of Migratory Beekeeping: Genetic Variation Between Stationary and Migratory Populations of Honey Bee (Apis mellifera L.) in Turkey. M.Sc. Thesis, Department of Biological Sciences, Middle East Technical University, Ankara, Turkey. 56pp.
-
Kirpik, M. A., O. Batutaki and D. Tanrmkulu. 2010. Determining the relative abundance of honey bee (Apis mellifera L.) races in Kars plateau and evaluating some of their characteristics. Kafkas. Univ. Vet. Fak. Derg. 16: 278-282.
[https://doi.org/10.9775/kvfd.2010.2081]

-
Klein, A. M., B. E. Vaissière, J. H. Cane, I. Steffan-Dewenter, S. A. Cunningham, C. Kremen and T. Tscharntke. 2007. Importance of pollinators in changing landscapes for world crops. Proc. R Soc. B. 274(1608): 303-313.
[https://doi.org/10.1098/rspb.2006.3721]

-
Kwon, M., R. N. Zakhidovna, K. B. Abdiazizovich, C. Jung and E. J. Kil. 2024. First metagenomic analysis of virome in Uzbekistan honey bee (Apis mellifera): Investigating basic information on honey bee viruses. J. Invertebr. Pathol. 206: 108171.
[https://doi.org/10.1016/j.jip.2024.108171]

- Lee, M. Y., I. P. Hong, Y. S. Choi, N. S. Kim, H. K. Kim, K. G. Lee and M. L. Lee. 2010. Present status of Korean beekeeping industry. Korean J. Apic. 25(2): 137-144.
- Levin, M. D. 1983. Value of bee pollination to U.S. agriculture. Bull. Ent. Soc. Am. 29: 50-51.
- Mannapov, A. G., L. I. Khoruzhii, N. A. Simoganov and L. A. Red’kova. 2015. Tekhnologiya proizvodstva produktsii pchelovodstva po zakonam prirodnogo standarta (Beekeeping Production Technology according to Natural Standard Laws), Moscow: Prospekt. 24-30.
-
Meixner, M. D., M. A. Pinto, M. Bouga, P. Kryger, E. Ivanova and S. Fuchs. 2013. Standard methods for characterising subspecies and ecotypes of Apis mellifera. J. Apic. Res. 52(4): 1-28.
[https://doi.org/10.3896/ibra.1.52.4.05]

-
Miguel, I., M. Baylac, M. Iriondo, C. Manzano, L. Garnery and A. Estonba. 2011. Both geometric morphometric and microsatellite data consistently support the differentiation of the Apis mellifera M evolutionary branch. Apidologie 42(2): 150-161.
[https://doi.org/10.1051/apido/2010048]

-
Minaud, E., F. Rebaudo, G. Mainardi, P. Vardakas, F. Hatjina, I. Steffan-Dewenter and F. Requier. 2024. Temperature in overwintering honey bee colonies reveals brood status and predicts colony mortality. Ecol. Indic. 169: 112961.
[https://doi.org/10.1016/j.ecolind.2024.112961]

-
Nawrocka, A., I. Kandemir, S. Fuchs and A. Tofilski. 2017. Computer software for identification of Honey Bee subspecies and evolutionary lineages. Apidologie 49: 172-184.
[https://doi.org/10.1007/s13592-017-0538-y]

-
Nuralieva, U., K. Tajiyev, Z. Sheralieva, M. Toishimanov, G. Moldakhmetova, K. Temirbayeva and A. Tajieva. 2023. Geometric morphometric characteristics of Apis mellifera honeybee in Kazakhstan. Biodiversitas 24(8).
[https://doi.org/10.13057/biodiv/d240821]

-
Oleksa, A. and A. Tofilski. 2015. Wing geometric morphometrics and microsatellite analysis provide similar discrimination of honey bee subspecies. Apidologie 46: 49-60.
[https://doi.org/10.1007/s13592-014-0300-7]

-
Papa, G., R. Maier, A. Durazzo, M. Lucarini, I. K. Karabagias, M. Plutino, E. Bianchetto, R. Aromolo, G. Pignatti, A. Ambrogio, M. Pellecchia and I. Negri. 2022. The honey bee Apis mellifera: An insect at the interface between human and ecosystem health. Biology 11(2): 233.
[https://doi.org/10.3390/biology11020233]

- Qoshmatovich, J. R. 2022. Technology of feeding families of bees in different types of households in uzbekistan. Galaxy Int. Interdiscip. Res. J. 10(3): 295-301.
-
Ramello, P. J., V. Almada, L. Ashworth, L. J. Alvarez and M. Lucia. 2024. Bee size increases pollen deposition in Cucurbita maxima (Cucurbitaceae) crops. Apidologie 55(2): 23.
[https://doi.org/10.1007/s13592-024-01065-9]

- Ruttner, F. 1988. Biogeography and Taxonomy of Honeybees. Springer-Verlag, Berlin.
-
Safonkin, A. F., T. A. Triseleva and T. O. Bykova. 2019. Intraracial diversity of the Carpathian race of honeybees Apis mellifera carpatica. Biol. Bull. 46: 492-499.
[https://doi.org/10.1134/s1062359019050091]

-
Salehi, S. and J. Nazemi-Rafie. 2020. Discrimination of Iranian honeybee populations (Apis mellifera meda) from commercial subspecies of Apis mellifera L. using morphometric and genetic methods. J. Asia-Pac. Entomol. 23(2): 591-598.
[https://doi.org/10.1016/j.aspen.2020.04.009]

-
Sheppard, W. S. and M. D. Meixner. 2003. Apis mellifera pomonella, a new honey bee subspecies from Central Asia. Apidologie 34(4): 367-375.
[https://doi.org/10.1051/apido:2003037]

-
Suchail, S., D. Guez and L. P. Belzunces. 2000. Characteristics of imidacloprid toxicity in two Apis mellifera subspecies. Environ. Toxicol. Chem. 19(7): 1901-1905.
[https://doi.org/10.1897/1551-5028(2000)019<1901:coitit>2.3.co;2]

- UN. 2024. World Population Prospects 2024: Summary of Results. Available at: https://population.un.org/wpp/, (accessed May 25, 2025).
- Zinov’eva, N. A., M. S. Fornara, A. V. Borodachev, E. A. Gladyr’, V. I. Lebedev, S. N. Akimova, N. I. Krivtsov and L. K. Ernst. 2013. Differentiation of the Carpathian and Krajina bees using microsatellites. Pchelovodstvo 1: 14-17.
