Honey Bee, Apis mellifera Toxicity of Five Antibiotic Formulations Used against the Fire Blight Control in Apple Orchard
Abstract
The use of antibiotics in agriculture to combat bacterial plant diseases has become a critical area of study due to its implications for environmental safety and agricultural sustainability. One major concern is the potential impact of these antibiotics on pollinators, particularly honey bees. In this study, the acute toxicity of five antibiotics on adult honey bees was evaluated using oral and contact spray bioassays, with six serial dilutions based on the recommended concentrations. The results showed that copper hydroxide was toxic to honey bees when administered orally, with an LD50 of 9.5 μg/bee. In contrast, the other four antibiotics-oxolinic acid, streptomycin, Bacillus subtilis, and oxytetracycline dehydrate-exhibited no toxicity in either the oral or spray bioassays. The high oral toxicity of copper hydroxide indicates that its application should be carefully managed during apple blooming to minimize harm to pollinators. These findings highlight the variability in antibiotic toxicity and underscore the need for careful consideration in their agricultural use to protect pollinator health.
Keywords:
Antibiotic, Honey bee, Toxicity, Oral, Spray, Copper hydroxideINTRODUCTION
Since the 1950s, antibiotics have been used to manage specific bacterial diseases affecting high-value crops such as fruits, vegetables, and ornamental plants (McManus et al., 2002). Antibiotics are defined as substances produced by microorganisms that inhibit the growth of or kill other microorganisms (Madigan et al., 1997). Some microbial compounds with antifungal properties-used, for instance, to control rice blast and other plant fungal diseases-have also been classified as antibiotics (Yamaguchi, 1995).
Regulatory approaches to antibiotic use in agriculture vary globally. Europe enforces strict regulations that significantly limit their use in crop production. In contrast, the United States permits the application of antibiotics such as streptomycin in orchards. In China and several Asian countries, antibiotics are widely applied in rice and vegetable farming (Batuman et al., 2024). In South Korea, antibiotics including streptomycin, oxytetracycline, oxolinic acid, and copper compounds are commonly used to control plant diseases (Awad et al., 2010).
Oxolinic acid, a quinolone antibiotic that inhibits DNA gyrase, has been employed in South Korea since 2015 to combat fire blight, a bacterial disease affecting apple and pear trees (Ham et al., 2022). Streptomycin, used since the 1950s to target various plant-pathogenic bacteria, is most commonly applied to manage Erwinia amylovora, the causative agent of fire blight that infects leaves, flowers, and shoots of apple and pear trees (McManus et al., 2002). In South Korea, E. amylovora-the pathogen responsible for black fire blight-was first reported in 1995, and since then, streptomycin has been a principal method of disease management in orchards (Lee et al., 2023).
Bacillus species, particularly Bacillus subtilis, have emerged as important biological control agents due to their antibiotic production and plant growth-promoting properties (Shafi et al., 2017). B. subtilis serves as both a biofertilizer and a biocontrol agent and is among the most widely utilized microbial products in agriculture (Wu et al., 2019).
Copper hydroxide is another widely used agricultural chemical, applied as a fungicide to control bacterial and fungal diseases in crops, trees, and ornamental plants. Produced via a chemical reaction between sodium hydroxide and copper sulfate, copper hydroxide forms fine particles that rapidly release copper ions, offering effective protection against pathogens (Rusjan, 2012). In South Korea, it is commonly used to protect crops from various fungal infections (Um et al., 2021).
Tetracyclines are broad-spectrum antibiotics active against bacteria, protozoa, parasites, and fungi, and are among the most commonly used in South Korea. Oxytetracycline, in particular, is widely used and is officially registered for agricultural use on crops (Kim et al., 2023).
In agricultural practice, antibiotics are typically applied through spraying onto tree canopies or by direct trunk injection, especially during the blooming period to prevent pathogen infection through flowers (Avila et al., 2024). Notably, the timing of these applications often overlaps with the period when fruit growers introduce honey bee colonies for pollination (Lawrence et al., 2022), potentially exposing pollinators to antibiotics and raising concerns about their impact on bee health.
Therefore, the objective of this study was to evaluate the acute toxicity of five agricultural antibiotics-oxolinic acid, streptomycin, B. subtilis, copper hydroxide, and oxytetracycline dihydrate-on honey bees through oral and spray bioassays under laboratory conditions.
MATERIALS AND METHODS
1. Honey bees
To evaluate the acute effects of antibiotics, Apis mellifera workers of similar life stages were collected from three healthy colonies maintained at the experimental apiary of Andong National University. Bees were carefully gathered by gently brushing individuals from brood frames without emerging bees, minimizing physical stress or injury. The collected bees were then placed in 120×80 mm insect breeding cages (SPL- TDS-ISBDJ, Pocheon, South Korea), following the procedure outlined by Williams et al. (2013). After collection, the bees were transferred to the laboratory and maintained under controlled conditions at 25±2°C with 50-60% relative humidity.
2. Antibiotics
The antibiotics were serially diluted in 50% sugar syrup from the producer’s recommended concentration (Giske et al., 2022) to 10-6 times, with active ingredient (a.i.) concentrations of oxolinic acid at 200 mg/L, streptomycin at 100 mg/L, copper hydroxide at 1540 mg/L, oxytetracycline dihydrate at 85 mg/L, and B. subtilis at 5×109 cfu/g, respectively.
3. Acute oral toxicity
Honey bees were released into the plastic cages with dimensions of 90 mm of bottom diameter×120 mm of top diameter×80 mm in height, with side ventilation slots (10 bees per cage) using a small amount of CO2 for anaesthetization, as detailed by Human et al. (2013), and starved for 2 hours in experimental room maintained at 25±2°C and 50-60% relative humidity. A plastic Kovax Syringe (5 mL) was used as feeder unit (Bisrat et al., 2020), containing 2 mL of the prepared dose. A 50% sugar solution alone was used as control. Afterward, the feeders containing antibiotics were replaced with feeders containing only the sugar solution, immediately after 2 hours. Each treatment had three replications. Bee mortality was observed at 6, 12, 24, 48 and 72 hours.
4. Spray toxicity
Honey bees (10 bees per group) were anesthetized using a small amount of CO2 and placed onto a Petri dish (15 cm in diameter). The bees were then sprayed 10 times from a distance of 15 cm using a 600 mL hand sprayer (KOMAX G600, Sansoo Co., LTD, Korea) containing the antibiotic solution. After spraying, the bees transferred into the plastic cages with feeders containing only a sugar solution. Each treatment had three replications. Bee mortality was observed at 6, 12, 24, 48 and 72 hours after spraying.
5. Statistical analysis
The median lethal concentration (LC50) values for 72 hours post-exposure to antibiotic were calculated using Probit analysis in SPSS version 16. Assuming a daily feeding amount of 35 μL (Laurino et al., 2011), the cumulative median lethal dose (LD50) for repeated treatments was determined based on the corresponding LC50 values. A volume of 1.575 mg/bee was used to estimate the LD50 from the corresponding LC50 value in the spray bioassay (Zhu et al., 2015).
RESULTS
Honey bees were subjected to acute oral and spray bioassays using six serial concentrations of five commonly used agricultural antibiotics (Table 1), starting from the recommended field concentration.

Percentage of active ingredient, type of commercial formulation, field recommended concentration, dilution factor, and mode of action of tested antibiotics
The results of the toxicity assessments are summarized in Table 2 for both oral and spray exposure routes. In the oral bioassay, four antibiotics-oxolinic acid, streptomycin, Bacillus subtilis, and oxytetracycline dihydrate-were found to be non-toxic to honey bees, with LD50 values exceeding 50 μg/bee. In contrast, copper hydroxide demonstrated moderate toxicity, with an LD50 of 9.5 μg/bee after 72 hours of exposure (Table 2).

Lethal dose (LD50 in μg/bee) of antibiotics to honey bees via oral and spray bioassays at 72 hours, with reference LD50 values (Rf.LD) from the literature
In the spray bioassays, all tested antibiotics-including copper hydroxide-exhibited low toxicity. Honey bee mortality remained below 10% across all tested concentrations, and LD50 values were greater than 50 μg/bee, indicating minimal risk from dermal exposure.
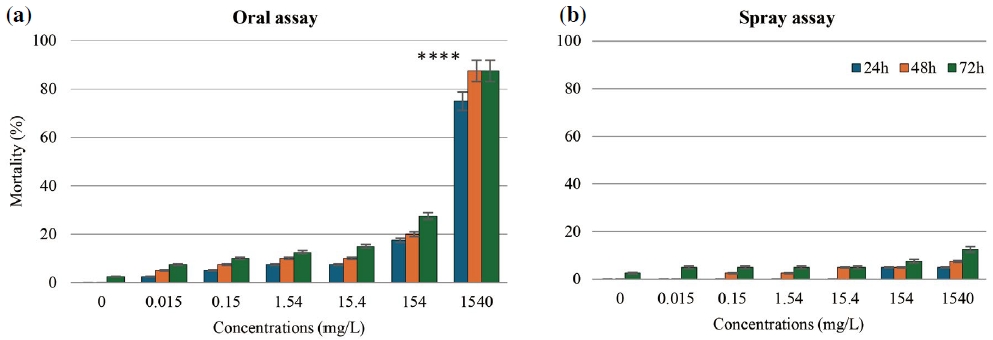
In the case of copper hydroxide, Fig. 2 illustrates a clear contrast in honey bee mortality between oral and spray exposure routes. Oral exposure at the highest concentration (1540 mg/L) led to mortality rates exceeding 80% at both 48 and 72 hours (Fig. 2a). In comparison, spray exposure at the same concentration resulted in less than 10% mortality (Fig. 2b). Statistical analysis confirmed a significant difference in mortality between the oral and spray bioassays at 1540 mg/L (p<0.0001), highlighting the critical role of exposure route in determining toxicity.
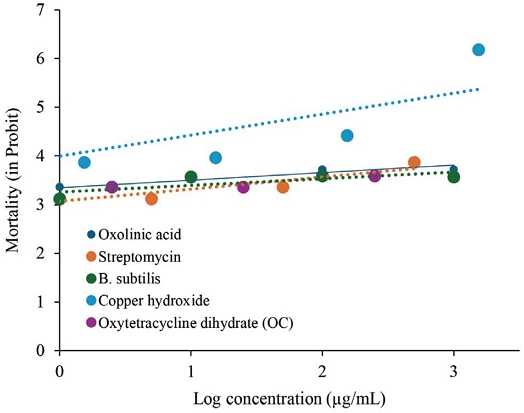
Log concentration lines of the mortality of bees to antibiotics (oxolinic acid, streptomycin, B. subtilis and oxytetracycline dehydrate).
DISCUSSION
This study assessed the acute toxicity of five agricultural antibiotics-oxolinic acid, streptomycin, Bacillus subtilis, copper hydroxide, and oxytetracycline dihydrate-on honey bees using oral and spray bioassays, with serial dilutions prepared from recommended field concentrations. The results revealed significant differences in toxicity profiles, with copper hydroxide exhibiting notably higher oral toxicity, while the other four compounds showed no adverse effects under the test conditions.
Our findings demonstrated that oxolinic acid, streptomycin, B. subtilis, and oxytetracycline dihydrate were non-toxic to honey bees in both oral and contact spray bioassays. All exhibited LD50 values exceeding 100 μg/bee (Table 2), consistent with prior studies and data from the EPA Pesticide Ecotoxicity Database. Specifically, oxolinic acid (Lewis et al., 2016), streptomycin (Ludvigsen et al., 2018), B. subtilis (Dedej et al., 2004), and oxytetracycline dihydrate have all been previously reported to exhibit low toxicity to honey bees. According to the U.S. Environmental Protection Agency (USEPA) classification, substances with LD50 values greater than 11 μg/bee are considered non-toxic (Fitzpatrick et al., 2017). Thus, these antibiotics pose minimal acute risk to pollinators at recommended concentrations.
However, while acute toxicity may be low, the long-term and sublethal effects of these substances, particularly on honey bee physiology and microbiota, remain important areas for further study (Hladun et al., 2015). Streptomycin, for example, has been shown to disrupt gut microbial communities, potentially compromising immune function and increasing vulnerability to pathogens (Ludvigsen et al., 2018). Such disruptions can negatively impact honey bee nutrition, energy metabolism, and overall colony health.
In contrast, copper hydroxide, when administered orally at the recommended concentration of 1540 mg/L, caused over 80% mortality within 48 to 72 hours, with an LD50 of 9.5 μg/bee after 72 hours-classifying it as moderately toxic by USEPA standards. Copper-based compounds are widely used in South Korean apple and pear orchards to manage bacterial diseases such as fire blight caused by Erwinia amylovora. Ryu et al. (2023) reported that copper hydroxide at 2000 ppm was highly effective against fire blight in apples, yet the toxicity risk to honey bees at these concentrations raises concerns (Glavan et al., 2024).
Copper hydroxide (Cu(OH)2) functions through the release of copper ions (Cu2+), which possess antimicrobial properties (Sinisi et al., 2018; Burandt et al., 2024). New formulations of copper compounds are partially systemic, increasing the risk of accumulation in plant tissues. In apples, repeated applications can lead to copper concentrations in flowers and fruits that exceed the EU maximum residue limit of 5 mg/kg (Kurnik et al., 2012). For instance, in Switzerland, seasonal applications elevated copper residues to 15 mg/kg (Kurnik et al., 2012), while in vivo studies have shown that copper can accumulate in foraging bees, reaching levels as high as 22.6 mg/kg (Roman, 2010). Notably, copper accumulation in bee tissues has been linked to reduced brain dopamine levels (Nisbet et al., 2018), which could impair cognitive functions.
Chronic exposure to copper has been associated with a range of detrimental effects on honey bee health. High concentrations can reduce lifespan, impair reproduction, and hinder hive maintenance behaviors (Burden, 2016; Di et al., 2016; Nikolić et al., 2019). Di et al. (2016) observed that copper exposure delayed larval development, reduced pupal weights, and decreased survival rates in a dose-dependent manner. Additionally, it affected feeding behavior and led to metal accumulation in both larvae and adult bees. Nikolić et al. (2019) further reported reduced activity of glutathione S-transferase, a key detoxification enzyme, under high copper exposure.
In contrast, the spray bioassay revealed mortality below 10% for copper hydroxide, indicating relatively low toxicity via contact exposure. The statistically significant difference between oral and spray bioassay outcomes (p<0.0001) underscores the importance of exposure route in determining risk. While ingestion of copper residues via nectar or water appears highly hazardous, dermal contact under typical field conditions may pose less immediate threat. Nonetheless, even at environmentally relevant concentrations, copper has been shown to impair appetitive learning and reduce long-term memory in honey bees (Monchanin et al., 2021), which could affect foraging efficiency and colony survival.
These findings highlight the need for careful management of antibiotic application in agriculture, particularly for compounds like copper hydroxide that exhibit high oral toxicity. Strategies such as restricting application during peak foraging periods, adjusting timing relative to bloom, and developing pollinator-safe formulations should be considered. Additionally, future studies should explore chronic toxicity, sublethal behavioral effects, and impacts on bee microbiota to fully assess the ecological consequences of agricultural antibiotic use.
References
-
Avila, L., C. McCullough, A. Schiffer, J. Moreno, N. Ganjur, Z. Ofenloch, T. DuPont, L. Nottingham, N. M. Gerardo and B. J. Brosi. 2024. Effects of a field-sprayed antibiotic on bee foraging behavior and pollination in pear orchards. Agric. Ecosyst. Environ. 359: 108757.
[https://doi.org/10.1016/j.agee.2023.108757]

-
Awad, Y. M., S. S. Lee, S. C. Kim, J. E. Yang and Y. S. Ok. 2010. Novel approaches to monitoring and remediation of veterinary antibiotics in soil and water: a review. Korean J. Environ. Agric. 29(4): 315-327.
[https://doi.org/10.5338/kjea.2010.29.4.315]

-
Batuman, O., K. Britt-Ugartemendia, S. Kunwar, S. Yilmaz, L. Fessler, A. Redondo, K. Chumachenko, S. Chakravarty and T. Wade. 2024. The use and impact of antibiotics in plant agriculture: A review. Phytopathol. 114(5): 885-909.
[https://doi.org/10.1094/phyto-10-23-0357-ia]

-
Benfenati, E., E. Boriani, M. Craciun, L. Malazizi, D. Neagu and A. Roncagioni. 2011. Databases for pesticide ecotoxicity. Quantitative Structure-activity Relationships (QSAR) for Pesticide Regulatory Purposes. Elsevier. Oct 13: 59-81.
[https://doi.org/10.1016/b978-044452710-3/50004-5]

-
Bisrat, D., D. Ulziibayar, T. Begna and C. Jung. 2020. Differential effects of the detoxicant on the honey bee intoxicated with seven pesticides and its chemical characterization. J. Apic. Res. 35(2): 91-98.
[https://doi.org/10.17519/apiculture.2020.06.35.2.91]

-
Burandt, Q. C., H. B. Deising and A. von Tiedemann. 2024. Further Limitations of Synthetic Fungicide Use and Expansion of Organic Agriculture in Europe Will Increase the Environmental and Health Risks of Chemical Crop Protection Caused by Copper-Containing Fungicides. Environ. Toxicol. Chem. 43(1): 19-30.
[https://doi.org/10.1002/etc.5766]

- Burden, C. M. 2016. Sublethal effects of heavy metal and metalloid exposure in honey bees: behavioral modifications and potential mechanisms. Arizona State University.
-
Dedej, S., K. S. Delaplane and H. Scherm. 2004. Effectiveness of honey bees in delivering the biocontrol agent Bacillus subtilis to blueberry flowers to suppress mummy berry disease. Biological Control 31(3): 422-427.
[https://doi.org/10.1016/j.biocontrol.2004.07.010]

-
Di, N., K. R. Hladun, K. Zhang, T. X. Liu and J. T. Trumble. 2016. Laboratory bioassays on the impact of cadmium, copper and lead on the development and survival of honey bee (Apis mellifera L.) larvae and foragers. Chemosphere 152: 530-538.
[https://doi.org/10.1016/j.chemosphere.2016.03.033]

-
Fitzpatrick, J., R. Schoeny, K. Gallagher, K. Deener, C. Dockins, M. Firestone, W. Jordan, M. McDonough, D. Murphy, M. Olsen and K. Raffaele. 2017. US Environmental Protection Agency̓s framework for human health risk assessment to inform decision making. IJRAM. 20(1-3): 3-20.
[https://doi.org/10.1504/ijram.2017.082558]

-
Giske, C. G., J. Turnidge, R. Cantón and G. Kahlmeter. 2022. Update from the European committee on antimicrobial susceptibility testing (EUCAST). J. Clin. Microbiol. 60(3): 00276-21.
[https://doi.org/10.1128/jcm.00276-21]

-
Glavan, G., G. Benko and J. Božič. 2024. Impact of copper and zinc oral chronic exposure on Carniolan honey bee survival and feeding preference. J. Econ. Entomol. 117(4): 1485-1492.
[https://doi.org/10.1093/jee/toae108]

-
Ham, H., G. R. Oh, D. S. Park and Y. H. Lee. 2022. Survey of oxolinic acid-resistant Erwinia amylovora in Korean apple and pear orchards, and the fitness impact of constructed mutants. Plant Pathol J. 38(5): 482.
[https://doi.org/10.5423/ppj.oa.04.2022.0059]

-
Hladun, K. R., D. R. Parker and J. T. Trumble. 2015. Cadmium, copper, and lead accumulation and bioconcentration in the vegetative and reproductive organs of Raphanus sativus: implications for plant performance and pollination. J. Chem. Ecol. 41: 386-395.
[https://doi.org/10.1007/s10886-015-0569-7]

-
Human, H., R. Brodschneider, V. Dietemann, G. Dively, J. Ellis, E. Forsgren, I. Fries, F. Hatjina, F. L. Hu, R. Jaffé and A. B. Jensen. 2013. Miscellaneous standard methods for Apis mellifera research. J. Apic. Res. 52(4): 1-53.
[https://doi.org/10.3896/ibra.1.52.4.10]

-
Kim, S. H., S. Y. Park, G. E. Kim and E. H. Jho. 2023. Effect of pH and temperature on the biodegradation of oxytetracycline, streptomycin, and validamycin A in soil. Appl. Biol. Chem. 66(1): 1-8.
[https://doi.org/10.1186/s13765-023-00822-1]

-
Kurnik, V., V. Gaberšek, T. Unuk, S. Tojnko, A. Vogrin, S. Vajs and M. Lešnik. 2012. Influence of alternative copper fungicide formulations on copper content in apple fruits. Erwerbs-Obstbau 54: 161-170.
[https://doi.org/10.1007/s10341-012-0172-9]

- Laurino, D., M. Porporato, A. Patetta and A. Manino. 2011. Toxicity of neonicotinoid insecticides to honey bees: laboratory tests. Bull. Insectology 64(1): 107-113.
-
Lawrence, M. G., S. Williams, P. Nanz and O. Renn. 2022. Characteristics, potentials, and challenges of transdisciplinary research. One Earth 5(1): 44-61.
[https://doi.org/10.1016/j.oneear.2021.12.010]

-
Lee, M. H., H. Ham, H. W. Choi and D. S. Park. 2023. Isolation of streptomycin-resistant Erwinia pyrifoliae in Korea. Plant Dis. 107(3): 616-619.
[https://doi.org/10.1094/pdis-03-22-0553-sc]

-
Lewis, K. A., J. Tzilivakis, D. J. Warner and A. Green. 2016. An international database for pesticide risk assessments and management. Hum. Ecol. Risk Assess: Int. J. 22(4): 1050-1064.
[https://doi.org/10.1080/10807039.2015.1133242]

-
Ludvigsen, J., G. V. Amdam, K. Rudi and T. M. L’Abée-Lund. 2018. Detection and characterization of streptomycin resistance (strA-strB) in a honey bee gut symbiont (Snodgrassella alvi) and the associated risk of antibiotic resistance transfer. Microb. Ecol. 76: 588-591.
[https://doi.org/10.1007/s00248-018-1171-7]

- Madigan, M. T., J. M. Martinko and J. Parker. 1997. Brock biology of microorganisms. Vol. 11. Upper Saddle River, NJ. 490: 1-10.
- McManus, P. S., V. O. Stockwell, G. W. Sundin and A. L. Jones. 2002. Antibiotic use in plant agriculture. Annu. Rev. Phytopathol. 40(2002): 443-465.
-
Monchanin, C., E. Drujont, J. M. Devaud, M. Lihoreau and A. B. Barron. 2021. Metal pollutants have additive negative effects on honey bee cognition. J. Exp. Biol. 224: 241869.
[https://doi.org/10.1242/jeb.241869]

-
Nikolić, T. V., D. Kojić, S. Orčić, E. L. Vukašinović, D. P. Blagojević and J. Purać. 2019. Laboratory bioassays on the response of honey bee (Apis mellifera L.) glutathione S-transferase and acetylcholinesterase to the oral exposure to copper, cadmium, and lead. Environ. Sci. Pollut. Res. 26: 6890-6897.
[https://doi.org/10.1007/s11356-018-3950-6]

- Nisbet, C., A. Guler, N. OrmancÄ and S. Cenesiz. 2018. Preventive action of zinc against heavy metals toxicity in honeybee. Afr. J. Biochem. Res. 12: 1-6.
- Roman, A. 2010. Levels of Copper, Selenium, Lead, and Cadmium in Forager Bees. Pol. J. Environ. Stud. 19: 663-669.
- Rusjan, D. 2012. Copper in horticulture. In Fungicides for plant and animal diseases. IntechOpen.
-
Ryu, D. K., M. Adhikari, D. H. Choi, K. J. Jun, D. H. Kim, C. R. Kim, M. K. Kang and D. H. Park. 2023. Copper-Based Compounds against Erwinia amylovora: Response Parameter Analysis and Suppression of Fire Blight in Apple. Plant. Pathol. J. 39(1): 52.
[https://doi.org/10.5423/ppj.oa.07.2022.0100]

-
Shafi, J., H. Tian and M. Ji. 2017. Bacillus species as versatile weapons for plant pathogens: a review. Biotechnol. Biotechnol. Equip. 31(3): 446-459.
[https://doi.org/10.1080/13102818.2017.1286950]

-
Sinisi, V., P. Pelagatti, M. Carcelli, A. Migliori, L. Mantovani, L. Righi, G. Leonardi, S. Pietarinen, C. Hubsch and D. Rogolino. 2018. A green approach to copper-containing pesticides: antimicrobial and antifungal activity of brochantite supported on lignin for the development of biobased plant protection products. ACS Sustain. Chem. Eng. 7(3): 3213-3221.
[https://doi.org/10.1021/acssuschemeng.8b05135]

-
Um, N., S. O. Park, C. W. Yoon and T. W. Jeon. 2021. A pretreatment method for effective utilization of copper product manufacturing waste. J. Environ. Chem. Eng. 9(4): 105509.
[https://doi.org/10.1016/j.jece.2021.105509]

- US Environmental Protection Agency, 2006. Biopesticide registration action document. Bacillus subtilis strain QST 713 (PC code 006479).
-
Williams, M. L., G. Ilas, M. A. Jessee, B. T. Rearden, D. Wiarda, W. Zwermann, L. Gallner, M. Klein, B. Krzykacz-Hausmann and A. Pautz. 2013. A statistical sampling method for uncertainty analysis with SCALE and XSUSA. Nucl. Technol. 183(3): 515-526.
[https://doi.org/10.13182/nt12-112]

-
Wu, H., Q. Gu, Y. Xie, Z. Lou, P. Xue, L. Fang, C. Yu, D. Jia, G. Huang, B. Zhu and A. Schneider. 2019. Cold-adapted Bacilli isolated from the Qinghai-Tibetan Plateau are able to promote plant growth in extreme environments. Environ. Microbiol. 21(9): 3505-3526.
[https://doi.org/10.1111/1462-2920.14722]

- Yamaguchi, I. 1995. Modern selective fungicides: properties, applications, mechanisms of action. No. Ed. 2: 595.
-
Zhu, Y. C., J. Adamczyk, T. Rinderer, J. Yao, R. Danka, R. Luttrell and J. Gore. 2015. Spray toxicity and risk potential of 42 commonly used formulations of row crop pesticides to adult honey bees (Hymenoptera: Apidae). J. Econ. Entomol. 108(6): 2640-2647.
[https://doi.org/10.1093/jee/tov269]