
Comparative Evaluation of Acaricidal Effects on Varroa destructor during the Spring Season
Abstract
This study evaluated the corrected efficacy of three acaricide treatments, amitraz spray, oxalic acid fumigation, and fluvalinate strip for controlling Varroa destructor in honey bee (Apis mellifera) colonies during the spring season. In this experiment, all capped brood frames were removed prior to treatment to create a broodless condition, which is typical in early spring honey bee colony condition after overwintering. Colonies received one of the three treatments at 3 times at 1-week intervals, and mite densities were assessed before and after treatment using sticky board and sugar shaking method. Results showed that oxalic acid treatment was the most effective, followed by amitraz treatment, and fluvalinate treatment did not show sufficient level of control. The corrected efficacy obtained using sugar shaking and sticky boards were 33.0-69.5 for fluvalinate, 73.9-91.6 for oxalic acid fumigation, 58.7-74.6% for amitraz spray. All the treatments did not influence the honey bee population size in the colony, indicating the safety of the treatments to the honey bee colonies. Under broodless conditions, oxalic acid fumigation and amitraz spray have been found to be effective, while maintaining colony health, and the use of these acaricides is recommended.
Keywords:
Sugar shaking, Sticky boards, Amitraz spray, Oxalic acid fumigation, Fluvalinate stripINTRODUCTION
Varroa destructor (hereafter “Varroa”) is widely regarded as one of the most critical pests affecting Apis mellifera (hereafter “honey bee”) colonies worldwide (McMenamin and Genersch, 2015). Since shifting hosts from its natural host, Apis cerana, to A. mellifera, it has severely impacted apiculture and contributed significantly to colony losses globally (Brown et al., 2018). With its nearly global distribution (Rosenkranz et al., 2010), untreated infestations can weaken or collapse colonies (Frey and Rosenkranz, 2014). As an ectoparasite, Varroa infests both adult bees and brood, feeding on hemolymph and fat bodies, which weakens bee immunity (Ramsey et al., 2019). Its infestations reduce bee lifespan, impair metabolism and reproduction, and increase vulnerability to viral infections such as deformed wing virus (DWV) and acute bee paralysis virus (ABPV) (Martin et al., 2012). Additionally, Varroa indirectly harm colonies by decreasing population sizes, foraging and pollination efficiency, and honey production, while making colonies more susceptible to environmental stress. Given its rapid reproduction, colonies can be overwhelmed within approximately six months (Jung, 2009).
Most commercial beekeepers manage Varroa by treating highly infested colonies with synthetic acaricide (Haber et al., 2019). Chemical control remains the most reliable strategy for mitigating Varroa infestations (Jack and Ellis, 2021). Accordingly, a substantial proportion of both small- and large-scale beekeepers employ acaricide derived from either natural or synthetic origins (Jung and Kim, 2008; Haber et al., 2019). Among the naturally occurring compounds, thymol, oxalic acid, and formic acid are commonly used. Synthetic treatments include fluvalinate, amitraz, and coumaphos; however, their safety for the colony and effectiveness against Varroa are influenced by environmental factors and the physiological state of the colony, such as the extent of capped brood (Haber et al., 2019). However, resistance to these chemicals has developed over recent decades (Mitton et al., 2022). Although nonsynthetic chemicals and mechanical interventions are also used, they are rarely effective on their own (Jack and Ellis, 2021). Consequently, beekeepers often rely on the same chemical treatments year after year, further promoting resistance (Jeong et al., 2016).
Amitraz, a formamidine compound, exerts its acaricidal effects by binding to insect and acarine octopamine (OAR) and tyramine (TAR) receptors (Kumar, 2019). Prolonged use of amitraz in beekeeping has imposed significant selective pressure on mite populations. This pressure favors the development of resistance, as documented in several regions across USA (Rinkevich, 2020) and Europe (Kamler et al., 2016). Although no reports of amitraz resistance have been documented in Korea, resistance has been observed internationally. This discrepancy underscores the need for domestic studies to monitor resistance development and to inform effective Varroa management strategies.
Oxalic acid, a natural compound extracted from plants, has gained widespread use (van der Steen and Vejsnæs, 2021). Its low residue levels in hives and high efficacy make it widely used for controlling Varroa (Rademacher and Harz, 2006). Oxalic acid has been used since the 1980s (Takeuchi and Harada, 1983), and studies have shown that it acts through contact toxicity to control mites (Aliano and Ellis, 2008). Despite decades of use, no resistance to oxalic acid has been reported.
Spring Varroa control significantly influences the initial growth and honey production of honey bee colonies (Jung, 2012). Prior study indicates that treatment applied at an infestation threshold of 2% (i.e., 2 mites per 100 bees) during the spring increases honey yield (Ostermann and Currie, 2004). Once the honey flow period commences, the application of treatments is not feasible; hence, early intervention is critical (Ostermann and Currie, 2004). Failure to implement timely spring treatment may result in colony collapse before autumn (Goodwin and Van Eaton, 2001). Therefore, early spring treatment is essential.
In the spring, when beekeepers routinely remove capped brood from the hives prior to initiating Varroa control, the dynamics of mite infestation differ significantly from those in brood-present colonies. Under these broodless conditions, the effectiveness of oxalic acid is expected to be enhanced, as it typically shows superior performance when brood is absent. Based on this seasonal practice, we hypothesize that oxalic acid fumigation will demonstrate a higher treatment effect than amitraz and fluvalinate in the early spring. By focusing on spring treatments, this experiment seeks to provide targeted insights into the acaricidal effects of these compounds during the critical period of initial Varroa management, ultimately informing more effective seasonal strategies in apiculture.
MATERIALS AND METHODS
1. Experimental colonies
To investigate effective spring management of ectoparasitic mites, a field experiment was conducted between March 3 and April 4, 2025, at the Gyeongkuk National University apiary (36°32ʹ47ʺN, 128°48ʹ08ʺE). Prior to treatment, spring activity in the colonies was initiated on February 14, and all sealed brood frames were removed. The test colonies included an average of 3,860 adult bees and 330 larvae. Four treatment groups were established, each consisting of four replicates. Notably, these colonies had been treated with oxalic acid prior to the winter season.
2. Chemicals treatment
This study evaluated the treatment effect of three acaricides-fluvalinate, amitraz, and oxalic acid. Formic acid was utilized as a follow-up treatment for comparative analysis. Fluvalinate (JB’S, Seoul, Republic of Korea) was applied in the form of strips, each containing 30 mg of active compound. Amitraz was used in a commercial solution (Soksalman 10ho, LATTICE, Gwangju, Republic of Korea) at a concentration of 125 g per 1,000 mL. Oxalic acid (CAS No. 6153-56-6; Bepoch Master Global Business Inc., Nanjing, China) was applied with a purity of 99.6%. For the follow-up assessment, 60% formic acid (CAS No. 64-18-6; Feicheng Acid Chemicals Co., Ltd., Feicheng, China) was employed.
For fluvalinate application, a single strip was placed between the second and third frames in each hive. Amitraz was diluted by mixing 1 mL of the concentrate with 1,000 mL of water, and 4 mL of the resulting solution was sprayed onto each frame. Oxalic acid was administered via fumigation using a gas-torch-style vaporizer (Henan Bee Healthy Bio-Technology Co., Ltd., Henan, China) positioned at the hive entrance. 1 g of oxalic acid was loaded into the vaporizer, and treatment continued until smoke emission ceased. Following fumigation, the entrance was sealed for 10 minutes.
All treatments were repeated 3 times at 1-week intervals. In the subsequent control phase, formic acid was applied using a vaporizer (Dabee Co., Ltd., Iksan, Republic of Korea). 6 mL of the acid were absorbed into the device’s internal pad, the adjustment cap was closed, and the vaporizer was positioned on the hive’s inner cover. During this procedure, the hive entrance remained fully open (Table 1).
3. Evaluation of acaricide treatment
Prior to the application of acaricides, baseline mite density was measured using two complementary techniques. First, to assess natural mites falling, a Vaseline-coated sticky board was placed on the bottom of each hive to collect mites that dropped naturally (Kim and Jung, 2010; Dietemann et al., 2013). These boards were positioned before treatment began and retrieved after 72 hours to quantify the fallen mites.
To evaluate infestation levels on adult bees, the sugar shaking method was implemented in accordance with the procedure described by Kim and Jung (2008). Approximately 100 adult bees were sampled and placed in a ventilated container with a mesh lid. 20 g of powdered sugar were added, and the container was gently shaken for 1 minute to ensure uniform coating. The container was then inverted, and mites dislodged by the sugar were counted as they passed through the mesh. This procedure was repeated three times for each colony.
Mite density was reassessed after 2 months following the completion of the treatment. The same assessment methods, sugar shaking and 72-hour sticky board monitoring, were employed. No acaricides were administered during this post-treatment evaluation.
When using acaricide and subsequent follow-up treatments, treatment effects were calculated following the method described by Dietemann et al. (2013). The formula used is as follows:
To account for significant natural mite drops in control groups, treatment effect was corrected by the mite drops in the control treatment proposed by Abbott (1925) and Rashid et al. (2020). The formula used is as follows:
4. Colony strength evaluation
To evaluate the impact of the treatments on overall colony health, measurements of colony strength were performed. The key indicators included the number of adult bees and larvae. Adult bee population was estimated based on the assumption that three cells accommodate one bee; thus, a frame fully covered with bees was calculated to hold approximately 1,150 individuals. The area occupied by larvae measured following the protocol described by Delaplane et al. (2013), in which each frame was segmented into 32 equal parts, and 103 cells were counted within each segment. Colony strength assessments were carried out twice - once before treatment application and again after the final chemical treatment.
5. Statistical analysis
The numbers of adult bees and larvae per colony adult bee infestation, recorded before and after treatment, were analyzed using a paired t-test when the data were normally distributed; otherwise, the nonparametric Wilcoxon signed-rank test was applied. The comparison of the number of mites fallen on the sticky board after each treatment and percentage change in adult and larval populations between treatment after acaricide application were performed using one-way ANOVA or Kruskal Wallis Test. The comparison between treatment groups for corrected efficacy was also analyzed using ANOVA test. This analysis was applied according to the normality of the data. Post-hoc test were performed using Tukey’s test. All analyses were conducted using SPSS software (IBM, Armonk, NY, USA).
RESULTS
1. Ectoparasitic mite density evaluation
Among the samples collected from adult bees, only Varroa were identified. In the control group that received no treatment, the infestation rate exhibited a significant increase following the experimental period (Paired t-test, t=-5.509, p<0.05; Fig. 1). In contrast, colonies treated with fluvalinate or oxalic acid via fumigation showed no notable change in infestation levels between the pre- and post-treatment assessments. While the amitraz-treated group demonstrated a decrease in mite prevalence after treatment, the reduction was not statistically significant.
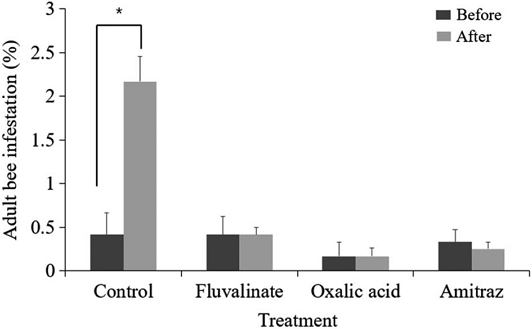
Varroa infestation rates (mean±SD, %) on adult honey bees measured before and after treatments by sugar shaking method in the spring. Treatments (control, fluvalinate strip, oxalic acid fumigation, amitraz spray) were applied 3 times at 1-week intervals. Sugar shakings were done before and 1-week after the last treatment. Asterisks indicate a statistically significant difference between before and after treatment (Paired t-test, p<0.05).
Mite drops onto sticky boards were monitored after each application of acaricides. Initial Varroa densities prior to treatment did not differ significantly among the treatment groups. However, after the first round of applications, a significant treatment effect was observed (One-way ANOVA, F=6.916, p<0.01; Fig. 2). The average number of mites (mean±SD) collected in the first application was as follows: 9.25±15.76 in the control group, 5.75±8.70 for fluvalinate, 24.75±10.90 for oxalic acid, and 29.50±16.52 for amitraz. After the second application, mite drop averaged 10.25±2.50 (control), 6.75±2.00 (fluvalinate), 9.25±8.30 (oxalic acid), and 12.00±3.59 (amitraz), with no statistically significant differences among groups. Similarly, results after the third application were 10.75±2.89 (control), 6.50±2.83 (fluvalinate), 6.75±4.35 (oxalic acid), and 8.25±1.29 (amitraz), again showing no significant variation among treatments.
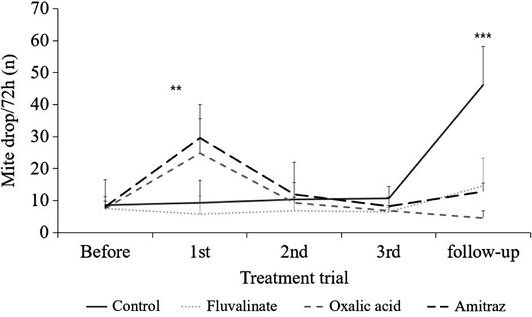
Number of fallen Varroa (Mean±SD/colony, n) collected on sticky boards before and after treatments in the spring. Treatments (control, fluvalinate strip, oxalic acid fumigation, amitraz spray) were applied 3 times at 1-week intervals. Asterisks indicate a statistically significant difference (p<0.001) by ANOVA.
Further evaluation following the final treatment revealed notable differences between groups (One-way ANOVA, follow-up; F=23.396, p<0.001). The average number of fallen mites was highest in the control group (46.3±12.0), followed by amitraz (12.8±2.6), fluvalinate (14.5±8.7), and oxalic acid (4.5±2.4). A paired t-test comparing pre-treatment and 2-month post-treatment mite numbers revealed statistically significant increases in both the control group (Paired t-test, t= -4.498, p<0.05) and the fluvalinate group (Paired t-test, t=-4.788, p<0.05).
2. Treatment effect
Mite survival rates before and after acaricide application were determined using the sugar shaking method, while treatment effect was assessed based on mite counts collected from sticky boards. To evaluate the longer-term effects, data collected 2 months of post-treatment were used to estimate both survival rates and treatment effect at that time point. The values were then corrected using Abbott’s formula, with the control group serving as the baseline for adjustment. Among the tested treatments, oxalic acid fumigation demonstrated the highest treatment effect against Varroa, followed by amitraz, whereas fluvalinate exhibited the lowest performance. Significant differences among treatments were observed in corrected efficacy, as measured by sticky board (Table 2). Additionally, all corrected efficacy showed the same trend, with oxalic acid exhibiting the highest corrected efficacy, followed by amitraz, and finally fluvalinate.

Corrected efficacies (mean±SD, %) of acaricide treatments on Varroa measured by sugar shaking and sticky board methods at 1 and 2 months after treatment
With respect to Tropilaelaps, mite presence was confirmed in only a limited number of colonies: two in the control group, one in the oxalic acid group, and two in the amitraz group. No Tropilaelaps were detected in the fluvalinate-treated colonies. Due to the small sample size, treatment effect analysis for Tropilaelaps was not conducted.
3. Evaluation of colony strength
Post-treatment assessments revealed a general decline in the number of adult bees across all treatment groups (Table 3). Despite this trend, no statistically significant differences were identified when comparing pre- and post-treatment values within each group. Conversely, larval counts showed an upward trend following treatment in all groups. Notably, significant changes in brood area were observed, particularly in the control (t=-3.350, p<0.05), fluvalinate (t=-5.894, p<0.05), oxalic acid fumigation (t=-6.363, p<0.01), and amitraz spray (t=-4.394, p<0.05), as determined by paired t-tests.

Population changes (Mean±SD, Log10 percent change) of adult bees and larvae before and after 28 days treatments in the spring experiment
To further evaluate the variation in adult bees and larval populations, the net increase or decrease post-treatment was calculated, and a Kruskal-Wallis test was performed to compare differences among treatment groups. The analysis did not reveal any statistically significant differences in either adult bee numbers (H=2.149, p>0.05) or larval counts (H=2.776, p>0.05).
DISCUSSION
Through a spring experiment evaluating the effectiveness of mite control treatments in honey bee colonies, the following results were obtained. Oxalic acid fumigation and amitraz spray showed strong control effects, while fluvalinate did not demonstrate any noticeable treatment effect. When comparing mite infestation rates before and after treatment, the untreated group showed a clear increase in infestation. In the fluvalinate and oxalic acid groups, infestation rates remained similar before and after treatment. Although the amitraz group showed a decrease in infestation, the difference was not statistically significant.
Previous summer trials conducted in Korea showed control rates of 72.2% in Gunwi and 66.8% in Andong (Oh and Jung, 2025). In contrast, this spring trial achieved a higher control rate of 73.9-91.6%. All treatment effects and corrected efficacy showed similar trends. This difference may be due to two main factors: the presence of brood frame and the amount of oxalic acid used. In the summer, 2 g of oxalic acid was applied to colonies with about 6,000 adult bees, while in spring, 1 g was used for colonies with around 3,860 bees. Since the amount of oxalic acid per bee was proportionally similar in both cases, the difference in dosage likely did not affect the results. However, the presence of brood frames may have played a major role. The summer trial was conducted with brood frame present, while in the spring trial, all brood frame was removed. Previous studies have shown that oxalic acid is not effective against mites inside capped brood cells (Gregorc et al., 2016) and works better when no brood is present (Oh et al., 2024). This supports the finding that oxalic acid was more effective during the broodless spring period.
For amitraz, the control effect was slightly lower than that of oxalic acid, although the difference was not statistically significant. The average temperature during the trial was 7.6±6.6°C. According to previous studies (Meilkle et al., 2017), the effect of amitraz tends to weaken at temperatures below 20°C, suggesting that the lower temperature during the spring trial may have reduced its effectiveness. Therefore, using amitraz at low temperatures can be ineffective in controlling mites and may lead to overuse. Varroa have evolved resistance to many synthetic acaricides as a result of their excessive and improper use, which has consequently diminished the overall effectiveness of these chemical treatments (Sammataro et al., 2005). Although no cases of amitraz resistance have been reported in Korea to date, international studies have documented such occurrences under conditions of repeated misuse and over-application (Kamler et al., 2016; Rinkevich, 2020). This suggests that even in the absence of current domestic resistance, cautious and judicious use of amitraz is warranted to prevent future resistance development. It is therefore recommended that beekeepers adhere strictly to the recommended application protocols and incorporate integrated pest management strategies to mitigate potential risks. It is necessary to use them at the appropriate temperature.
When comparing the number of mites before treatment and two months after treatment, statistically significant increases were observed in the control and fluvalinate groups. But no statistically significant changes were observed in the oxalic acid fumigation and amitraz spray groups. This suggests that oxalic acid and amitraz effectively controlled mites, resulting in the maintenance of mite density two months after treatment.
Compared to pre-treatment, a slight decrease in the number of adult bees was observed after treatment; however, these differences were not statistically significant. Prior to treatment, all brood frame within the colonies was removed, so the chemicals affected only the adult bees. Furthermore, the increase or decrease in the number of adult bees after treatment was not statistically significant. This suggests that the chemicals may have exerted a minor impact on adult bees. Previous studies have reported that fluvalinate, amitraz spray, and oxalic acid fumigation can influence adult bees (Rademacher et al., 2017); nonetheless, no statistically significant differences were observed in this study. Regarding the number of larvae, an increase was detected post-treatment compared with pre-treatment levels. In the untreated control, as well as in the oxalic acid fumigation and amitraz treatment groups, this increase was statistically significant. In addition, post-treatment comparisons of larval numbers in treated versus untreated areas did not reach statistical significance. Since spring is a period of colony growth, the observed increase in larval area is natural (Bodenheimer, 1937). It is determined that there is no effect from the treatment.
In this study, our findings clearly demonstrate that seasonal conditions play a crucial role in the effectiveness of acaricide treatments in honey bee colonies. The significantly higher corrected efficacy of oxalic acid fumigation in the broodless spring environment supports the importance of early Varroa management, before brood is present. In contrast, the reduced performance of amitraz at low temperatures - potentially leading to its overuse and the subsequent risk of resistance development - highlights the need for careful adherence to recommended application protocols. Furthermore, fluvalinate failed to achieve effective mite control. These results underscore the necessity for integrated pest management approaches that consider both environmental conditions and proper dosage to ensure sustainable Varroa control in apiculture.
Acknowledgments
This research was partly funded by the National Research Foundation of Korea (NRF-2018R1A6A1A03024862), and RDA agenda on Smart beekeeping (RS-2023-00232847).
References
- Abbott, W. S. 1925. A method of computing the effectiveness of an insecticide, J. Econ. Entomol. 18: 265-267.
-
Aliano, N. P. and M. D. Ellis. 2008. Bee-to-bee contact drives oxalic acid distribution in honey bee colonies. Apidologie 39(5): 481-487.
[https://doi.org/10.1051/apido:2008030]

-
Bodenheimer, F. S. 1937. Studies in animal populations. II. Seasonal population-trends of the honey-bee. Q Rev. Biol. 12(4): 406-425.
[https://doi.org/10.1086/394540]

-
Brown, P., L. E. Newstrom-Lloyd, B. J. Foster, P. H. Badger and J. A. McLean. 2018. Winter 2016 honey bee colony losses in New Zealand. J. Apic. Res. 57(2): 278-291.
[https://doi.org/10.1080/00218839.2018.1430980]

-
Delaplane, K. S., J. Van Der Steen and E. Guzman-Novoa. 2013. Standard methods for estimating strength parameters of Apis mellifera colonies. J. Apic. Res. 52(1): 1-12.
[https://doi.org/10.3896/ibra/1.52.1.03]

-
Dietemann, V., F. Nazzi, S. J. Martin, D. L. Anderson, B. Locke, K. S. Delaplane and J. D. Ellis. 2013. Standard methods for varroa research. J. Apic. Res. 52(1): 1-54.
[https://doi.org/10.3896/ibra.1.52.1.09]

-
Frey, E. and P. Rosenkranz. 2014. Autumn invasion rates of Varroa destructor (Mesostigmata: Varroidae) into honey bee (Hymenoptera: Apidae) colonies and the resulting increase in mite populations. J. Econ. Entomol. 107(2): 508-515.
[https://doi.org/10.1603/ec13381]

- Goodwin, M. and C. Van Eaton. 2001. Control of varroa. A guide for New Zealand Beekeepers. New Zealand Ministry of Agriculture and Forestry (MAF). Wellington.
-
Gregorc, A., J. Adamczyk, S. Kapun and I. Planinc. 2016. Integrated Varroa control in honey bee (Apis mellifera carnica) colonies with or without brood. J. Apic. Res. 55: 253-258.
[https://doi.org/10.1080/00218839.2016.1222700]

-
Haber, A. I., N. A. Steinhauer and D. vanEngelsdorp. 2019. Use of chemical and nonchemical methods for the control of Varroa destructor (Acari: Varroidae) and associated winter colony losses in U S beekeeping operations. J. Econ. Entomol. 112(4): 1509-1529.
[https://doi.org/10.1093/jee/toz088]

-
Jack, C. J. and J. D. Ellis. 2021. Integrated pest management control of Varroa destructor (Acari: Varroidae), the most damaging pest of Apis mellifera L. (Hymenoptera: Apidae) colonies. J. Insect Sci. 21(5): 6.
[https://doi.org/10.1093/jisesa/ieab058]

-
Jeong, S., C. Lee, D. Kim and C. Jung. 2016. Questionnaire study on the overwintering success and pest management of honey bee and damage assessment of Vespa hornets in Korea. J. Apic. 31: 201-210.
[https://doi.org/10.17519/apiculture.2016.09.31.3.201]

- Jung, C. 2009. Frame Work of the Adaptive Population Dynamic Model of Varroa Mite in Korean Beekeeping Environment. Kor. J. Apic. 24: 67-74.
- Jung, C. 2012. Incorporation of damage function of varroa mites into Honeybee varroa mite interaction model. J. Apic. 27: 215-221.
- Jung, C. and D. Kim. 2008. A population model of the Varroa mite, Varroa destructor on adult honeybee in the colony I. Exponential population growth. J. Apic. 23(4): 269-273.
-
Kamler, M., M. Nesvorna, J. Stara, T. Erban and J. Hubert. 2016. Comparison of tau-fluvalinate, acrinathrin, and amitraz effects on susceptible and resistant populations of Varroa destructor in a vial test. Exp. Appl. Acarol. 69: 1-9.
[https://doi.org/10.1007/s10493-016-0023-8]

- Kim, D. and C. Jung. 2008. Evaluation of chemical susceptibility for Varroa destructor in honeybee (Apis mellifera). J. Apic. 23(4): 259-268.
- Kim, D. and C. Jung. 2010. Comparison of population monitoring techniques for the ectoparasitic mite Varroa destructor in honeybee (Apis mellifera L.). J. Apic. 25(3): 201-209.
-
Kumar, R. 2019. Molecular markers and their application in the monitoring of acaricide resistance in Rhipicephalus microplus. Exp. Appl. Acarol. 78(2): 149-172.
[https://doi.org/10.1007/s10493-019-00394-0]

-
Martin, S. J., A. C. Highfield, L. Brettell, E. M. Villalobos, G. E. Budge, M. Powell and D. C. Schroeder. 2012. Global honey bee viral landscape altered by a parasitic mite. Science 336(6086): 1304-1308.
[https://doi.org/10.1126/science.1220941]

-
McMenamin, A. J. and E. Genersch. 2015. Honey bee colony losses and associated viruses. Curr. Opin. Insect Sci. 8: 121-129.
[https://doi.org/10.1016/j.cois.2015.01.015]

-
Meikle, W. G., M. Meiss, P. W. Maes, W. Fitz, L. A. Snyder, T. Sheehan and K. E. Anderson. 2017. Internal hive temperature as a means of monitoring honey bee colony health in a migratory beekeeping operation before and during winter. Apidologie 48(5): 666-680.
[https://doi.org/10.1007/s13592-017-0512-8]

-
Mitton, G. A., F. Meroi Arcerito, H. Cooley, G. Fernández de Landa, M. J. Eguaras, S. R. Ruffinengo and M. D. Maggi. 2022. More than sixty years living with Varroa destructor: A review of acaricide resistance. Int. J. Pest Manag. 68: 1-18.
[https://doi.org/10.1080/09670874.2022.2094489]

-
Oh, H. and C. Jung. 2025. Evaluation of the efficacy of formic acid and oxalic acid against Varroa destructor and Tropilaelaps mercedesae in Apis mellifera colonies during summer season. J. Apic. 40(1): 1-12.
[https://doi.org/10.17519/apiculture.2025.04.40.1.1]

-
Oh, H., T. Begna and C. Jung. 2024. Evaluating the Efficacy of Registered Acaricides on Varroa destructor and Tropilaelaps mercedesae, in Apis mellifera Colonies. J. Apic. 39(1): 21-30.
[https://doi.org/10.17519/apiculture.2024.04.39.1.21]

-
Ostermann, D. J. and R. W. Currie. 2004. The effect of formic acid formulations on honey bee, Apis mellifera L., colonies, and the influence of colony and ambient conditions on formic acid concentration in the hive. J. Econ. Entomol. 97(5): 1500-1508.
[https://doi.org/10.1603/0022-0493-97.5.1500]

-
Rademacher, E. and M. Harz. 2006. Oxalic acid for the control of varroosis in honey bee colonies - a review. Apidologie 37(1): 98-120.
[https://doi.org/10.1051/apido:2005063]

-
Rademacher, E., M. Harz and S. Schneider. 2017. Effects of oxalic acid on Apis mellifera (Hymenoptera: Apidae). Insects 8(3): 84.
[https://doi.org/10.3390/insects8030084]

-
Ramsey, S. D., R. Ochoa, G. Bauchan, C. Gulbronson, J. D. Mowery, A. Cohen and D. vanEngelsdorp. 2019. Varroa destructor feeds primarily on honey bee fat body tissue and not hemolymph. Proc. Natl. Acad. Sci. U S A. 116(5): 1792-1797.
[https://doi.org/10.1073/pnas.1818371116]

-
Rashid, B., A. Khani, V. Ghasemi, N. Sahebzadeh and S. Moharramipour. 2020. Evaluation of a new plant-based formulation for the treatment of varroosis in the honey bee colonies: efficacy and safety. Apidologie 51: 1074-1090.
[https://doi.org/10.1007/s13592-020-00786-x]

-
Rinkevich, F. D. 2020. Detection of amitraz resistance and reduced treatment efficacy in the Varroa mite, Varroa destructor, within commercial beekeeping operations. PLoS One 15(1): e0227264.
[https://doi.org/10.1371/journal.pone.0227264]

-
Rosenkranz, P., P. Aumeier and B. Ziegelmann. 2010. Biology and control of Varroa destructor. J. Invertebr. Pathol. 103(Suppl 1): S96-S119.
[https://doi.org/10.1016/j.jip.2009.07.016]

-
Sammataro, D., P. Untalan, F. Guerrero and J. Finley. 2005. The resistance of Varroa mites (Acari: Varroidae) to acaricides and the presence of esterase. Int. J. Acarol. 31(1): 67-74.
[https://doi.org/10.1080/01647950508684419]

- Takeuchi, K. and K. Harada. 1983. Control of Varroa jacobsoni mites with oxalic acid spray. Honeybee Sci. 4: 113-116.
-
Van Der Steen, J. and F. Vejsnæs. 2021. Varroa control: A brief overview of available methods. Bee World 98(2): 50-56.
[https://doi.org/10.1080/0005772x.2021.1896196]
