Vertical Stratification and Directionality as Determinant of Plant-Pollinator Interactions in Sweet Orange (Citrus sinensis L. Osbeck)
Abstract
A total of 18 species of insect visitors were observed to visit the sweet orange (Citrus sinensis L. Osbeck) flowers. Among the pollinators the Hymenoptera constituted major group of insects followed by Diptera and Lepidoptera. 18 species of insects belonging to 3 orders, 9 families and 13 genera were honeybees Apis florea F., Apis cerana, Apis dorsata F., Apis mellifera L., Carpenter bees Xylocopa fenestrata, X. pubescens. Besides, Musca spp., Eristalis spp., Syrphid spp., and butterflies were other insect visitors observed on citrus flowers. Of all these insects, honeybees were most abundant and comprised more than 63% of the flower visitors. Insect visitors were found to behave differently to different in Inflorescence height. It was interesting to note that honeybees visited more middle branches than the upper or lower ones whereas carpenter bees collected nectar and pollen mostly from the upper branches. The dipteran insects including Musca spp., Syrphus spp. and small Hymenopteran insects like Nomia and Lasioglossum spp. referred to as ‘others’ mostly visited lower branches. The impact of direction of plant had a pronounced effect on abundance of foraging population. Generally, more population was observed in east and west compared to north and west directions. Population of bees were at par in north and south directions. The results revealed that stratification and directionality had a significance influence on the behaviour of flower visiting insects.
Keywords:
Citrus, Honeybees, Vertical stratification, Directionality, Foraging rates, PollinationINTRODUCTION
Sexual reproduction of flowering plants largely depends on successful pollination. During pollination, pollen grains transferred from the anthers to the receptive stigmas of conspecific flowers, either by abiotic and/or biotic factors (Ashman et al., 2004; Gaines-Day and Gratton, 2015). However, biotic pollinators are more reliable mode for pollen transfer as they effect in targeted deposition of pollen among conspecifics. The variation in the abundance and availability of pollinators influences the reproductive success of plants.
Citrus belongs to the family Rutaceae and is the most important fruit crops in the world (Roussos, 2015). It is one of the most horticultural and economically important fruit crops globally (Wu et al., 2018), extensively cultivated in more than 140 countries and regions in the tropics and subtropics (Cuenca et al., 2016). A large number of citrus varieties-particularly grapefruits and mandarins, including oranges-are self-incompatible and either require or benefit from cross-pollination by insects to achieve fruit production or to improve yield and quality (Sanford, 1992; Chacoff and Aizen, 2007). Several studies have revealed increased fruit set and resultant production when managed honey bee colonies are used for pollination services (Sanford, 2003).
Numerous studies have shown that insect-mediated cross-pollination increases fruit set, although the degree of dependence varies by species and cultivar (Hearn et al., 1968; Vithanage, 1991; Wallace and Lee, 1999; Malerbo-Souza et al., 2004; Wallace, 2004; Chao, 2005; Papadakis et al., 2009; Yildiz and Kaplankiran, 2017; da Santos et al., 2021). In addition to higher fruit set, insect pollination has also been associated with improved fruit quality, including larger size, greater weight, increased juice content, and higher sugar levels (Manzoor-ul-Haq et al., 1978; Wallace and Lee, 1999; Malerbo-Souza et al., 2004; Vanlalhmangaiha et al., 2023). Despite this, the role of pollinators in citrus production remains a topic of debate (Sanford, 1992; Gurung and Chettri, 2021).
Among various citrus fruits, sweet orange produces highly attractive flowers that secrete nectar with a high sugar concentration (Hassanein and Ibrahim, 1959). Since the pollen is heavy and not adapted for wind dispersal (Krezdom, 1986), cross-pollination is primarily carried out by pollinators; however, some cultivars are also self-fertile (Free, 1993). Several factors are responsible for efficient production of sweet orange such as lack of high yielding varieties, problems of fruit drop, pests and diseases. Furthermore, lack of sufficient pollination at the right time the crop requires them is a major impediment for optimum crop production.
However, the presence and frequency of interactions between particular plants and pollinators vary in time (Olesen et al., 2008) and space (Espíndola et al., 2011; Newman et al., 2015). At the smallest scale, the position of an inflorescence in the context of the surrounding vegetation may affect the frequency and identity of flower visitors with consequences for plant reproduction.
In communities of multiple plants species, a few observational studies found that different bee species tend to visit flowers at different heights (Gumbert and Kunze, 1999; Hoehn et al., 2008). Junker et al. (2013) found that inflorescence height was the most important species trait after phenology to explain which plant species were visited by which insects. Studies investigating the effects of inflorescence height at the intraspecific level found that inflorescence height is under significant pollinator-mediated selection (Sletvold et al., 2010; Jiang and Li, 2017; Trunschke et al., 2017), although pollinator behaviour with respect to vertical stratification of pollinators is less known.
Most previous research focused on the importance of inflorescence height for plant reproduction, while little attention has been devoted to understanding whether and why foraging insects prefer flowers at certain heights. Some insight can be gained from observations of foraging behaviour of individual insects.
In honeybees, “horizontal movement” characterised by a tendency of individual bees to fly between plants of a similar height has been reported (Levin and Kerster, 1973; Faulkner, 1976). Preference of flowers at a certain height was demonstrated also in solitary bees (Gumbert and Kunze, 1999; Hoehn et al., 2008) and wasps (Peakall and Handel, 1993). Flying at a constant height may be advantageous from an energetic point of view for optimally foraging flower visitors (Heinrich, 1975; Pyke, 1978). Also, flowers close to the ground may be avoided by some insects because their visitation requires the ability to manoeuvre among plant stems, which may be challenging in dense vegetation (Gumbert and Kunze, 1999). In stratified tropical forests, bee species often restrict their foraging activity to certain strata (Frankie and Coville, 1979; Opler et al., 1980; Perry and Starret, 1980; Appanah, 1981; Roubik et al., 1982; Frankie and Haber, 1983; Bawa et al., 1985). This behaviour seems to be correlated to body size, with larger bees foraging in the canopy and smaller species in lower strata.
The present study was directed to test whether total visitation rate and the composition of flower visitor assemblages depend on inflorescence height and directionality of plant floral resources.
MATERIALS AND METHODS
1. Study site
Investigations made on insects visiting citrus trees during flowering period lasted during March-April 2016. The studies were carried out on insects visiting sweet orange Citrus sinensis during march April in citrus orchard of Sher-e-Kashmir University of Agricultural Sciences and Technology of Jammu. The study site is located at Latitude: 32°39ʹ26ʺ North, Longitude: 74°37ʹ58ʺ East and at altitude of 294 m.s.l. Five trees of sweet orange Citrus sinensis were selected at distance of 4-5 m from each other. The study area features a humid subtropical climate with extreme summer highs reaching 46°C (115°F), and temperatures in the winter months occasionally falling below 4°C (39°F). The study was conducted with the objective to determine the abundance and diversity of insects, their foraging behaviour, pollination efficiency and role in qualitative and quantitative improvement of the crop. In a standing crop, it is easier to mark the plot and record observations on insect visitors, but in tree plants it is difficult to obtain data at different canopies. Therefore, to meet the purpose of recording observations on citrus crop, the flowering shoots about 1.5-2.0 m above the ground were categorised into lower, middle and upper branches (located at 1 m from one another). Observation on bee counts for 5 min in each of the 5 fixed plots (size 1×1 m) were made each hour and the number of bees of each species recorded. Observations were made from 07.00 to 18.00 hours.
2. Insect sampling, preservation and morphometrics
In practice, this involved closely observing insects as they visited flowers and then capturing them using small, pollen-free glass tubes. Each individual insect was collected in a separate fresh glass vial. For larger insects or those that flew rapidly, a sweeping net was used for their collection. This comprehensive approach ensured that a wide range of flower-visiting insects were effectively captured and documented, providing a thorough assessment of pollinator diversity and behaviour.
Insects that were captured from flowers were euthanized using a killing bottle containing cotton soaked in a 98% ethyl acetate solution. Subsequently, all the insects were transferred to individual containers. To preserve the specimens, they were stored in 70% ethyl alcohol and, if necessary, card-mounted with a unique code assigned to each specimen. Insects preserved in 70% ethanol showed no alteration in their morphological characteristics. The different skeletal parts were separated under a stereo-binocular microscope and placed on a cavity block with glycerol. All measurements are given in millimeters and were measured using a Leica M205C microscope with auto measurement and analysis system.
The insects were then subjected to identification at the most detailed taxonomic level possible. Recognizable taxonomic units (RTU) were established based on observable morphological characteristics, following the method proposed by Oliver and Beattie (1993). Within the family/genus level, each insect specimen received a serial number. To ensure accurate identification, the latest literature available and representative materials were consulted, and professional taxonomists from various government and non-government research institutions, such as the Division of Entomology at the Indian Agricultural Research Institute in New Delhi, India, and the Department of Entomology at the University of Agricultural Sciences in Bangalore, India, were consulted for authentication purposes. The collected samples were deposited in the insect collection of the Apiculture Laboratory of the Division of Entomology at Sher-e-Kashmir University of Agricultural Sciences and Technology, Jammu.
The percentage of each bee species on upper, middle and lower branches was calculated as follows.
Likewise, the percentage of species ×2 and ×3 was calculated. Temperature and relative humidity were recorded with dry and wet bulb thermometer and solar radiation with a solarimeter (luxomet-300). Data on wind velocity was obtained from field observatory of department of Agrometeorology of the university. Data were recorded for 7 different days commencing from 3rd week of March to 2nd week of April. Floral visitors were observed during different hours of the day right from commencement to cessation at hourly intervals. The number of each visitor was counted in all field observations and then relative abundance of different visitors was calculated as follows:
Foraging rate of the floral visitors was recorded in terms of the number of flowers visited per unit time (Free, 1993). For each visitor, the type of visit the type of floral reward (nectar, pollen or tissue) collected were recorded. The foraging speed and time spent (second)/flower were recorded with stop-watch. For the foraging speed data, replications were unequal and it was analysed complete by randomized design (Snedecor and Cochran, 1989).
3. Statistical analyses
The recorded data were analysed for their variation between different treatments using Statistical Package for the Social Science (SPSS) and O.P Stat. Statistical analyses of the data were conducted to obtain the arithmetic mean, standard deviation and confidence interval at 95% level. Two-way ANOVA followed by Duncan’s multiple range test (DMRT) were used to analyse data and P≤0.05 was considered statistically significant.
RESULTS AND DISCUSSION
1. Diversity of insect pollinators on sweet orange
Sweet orange flowers were found to be highly attractive to flower visitors. The data presented in Table 1 shows that sweet orange citrus sinensis flowers attracted wide variety of insects belonging to 3 orders, 9 families, 13 genera and 18 species. The abundance of different flower visiting insects was in the order: Hymenoptera>Diptera>Lepidoptera. Of all these insects, honeybees Apis dorsata, A. mellifera, A. cerana, and A. florea were the dominant flower visitors and comprised of 63.50% of the total flower visiting insects. Their abundance was in the order: A. dorsata>A. mellifera>A. cerana>A. florea. The other important insects frequenting Citrus flowers were Lassioglosum spp., Halictus spp., Xylocopa fenestrata and X. pubescens. Besides, Musca spp., Eristalis spp., Syrphid spp., and butterflies were other insect visitors observad on citrus flowers. The latter group of insects mostly collected nectar and frequented at interrupted hours and were not considered as dependable pollinators.
In earlier studies also, several insect species have been recorded as pollinators of sweet orange like honeybees, carpenter bees, butterflies, hoverflies, wasps and ladybird beetles. Of all the insect visitors, use of honey bees remains the most consistent, effective and economical means of ensuring adequate yields. These outcomes strongly coincide with results of Chacoff and Aizen (2006) that honeybees are major and active pollinators of grapefruit in Northwestern Argentina. Mehmood et al. (2015) reported that eight insect pollinator species viz. Apis florea, Xylocopa sp., Pieris brassicae, Danaus plexippus, Papilio demoleus, Papilio hyale, Eumerus tuberculatus and Musca domestica were observed visiting regularly on citrus (Citrus limon). Foraging activity of insect pollinators was maximum at 0800-0900 and minimum during 1700-1800 hours. Hymenopteran pollinators were higher in numbers (42%) followed by Lepidopterans (33%) and Dipterans (25%). A. florea, E. tuberculatus, and P. demoleus were consistent and frequent visitor on both fruit plants. Karmakar (2013) found frequent visits of A. florea and Diptera during 0900 hours and Lepidoptera during 1000-1130 hours on Citrus aurantiifolia. Majority of insect pollinators during morning times possibly owing to availability of abundant food resources (nectar) compared to noon and evening. In Kagzi Kalan lemon, 10% plant of pumello should be planted which serve the purpose of pollinizer to get high yield. However extensive study is required on insect mediated pollination for efficient fruit production and increasing yield. The objective of present study was to assess various insect pollinators visiting citrus flowers and their peek foraging time.
Singh (2016) observed diverse group of pollinators of belonging to orders Hymenoptera, Diptera, Lepidoptera and Coleoptera visiting orange flowers. Out of 34 visitors, only 12 were frequently collecting pollens or nectar or both. Out of 12 pollinators, 9 belongs to Hymenoptera, 1 each to Diptera, Lepidoptera and Coleoptera. Free (1993) reported Xylocopa aestuans as the most important pollinator of citrus flowers. Hymenoptera (Apis mellfera and Apis cerana).
Diptera (syrphid) were regular visitors on the flower of Citrus spp. (Ghosh, 1990; Bhatia et al., 1995). The male H. squamosus had pollen adhered on tergum of elytra. The scopa was observed in Apidae, whereas on leg, thorax and abdomen in Xylocopidae, and leg and abdomen in Syryhidae (Michener, 2000; Abrol, 2012). The pollen gatherers are the most effective pollinators (Free, 1962). Citrus was visited by 13 species of bees, where Trigona spinrpes (stingless bee) and. A. mellifera were the most abundant (Castro, 2002).
2. Commencement and cessation of flight activities
Studies on commencement and cessation of flight activities of honeybees revealed that Apis dorsata commenced field activities much earlier than A. cerana followed by A. mellifera and A. florea. Similarly, activities of A. dorsata also ceased earlier than A. mellifera, A. florea and A. cerana, respectively. On an average, A. cerana had a higher foraging duration in the field followed by A. dorsata, A. mellifera and A. florea (Table 2). The data further revealed that peak foraging periods also varied fron one species to another. Peak foraging occurred between 0900-1400 in A. cerana, 1100-1300 in A. mellifera 1000-1100 in A. dorsata and 1300-1500 in A. florea. Similarly foraging rates, duration of foraging trips and population dynamics varied from one bee species to another depending upon their adaptiability. Similar results were obtained by Mehmood et al. (2015) who found that foraging activity of insect pollinators was highest at 0800-0900 and lowest during 1700-1800 hours.
3. Flower visitation rates
The data presented in Table 2 show that Apis cerana on an average visited 5.60±1.20 flowers per minute. A. dorsata, A. mellifera and A. florea visited 4.20±0.90, 6.20±1.30 and 2.60±1.12 flowers per minute respectively. Population dynamics of different bee species in terms of number of bees /100 flowers in case of A. cerana, A. dorsata, A. mellifera and A. florea was 14.60±2.10, 15.80±1.80, 10.40±1.20 and 4.20±1.16 respectively. The duration of foraging trips varied from a minimum of 20.2 (A. cerana) min to a maximum of and 28.6 (A. florea) min. The foraging duration of A. dorsata and A. mellifera was in between the two. Similarly flower visited per foraging trips varied from a minimum of 102.90 (A. mellifera) to a maximum of 130.4 (A. florea). The number of flowers visited per trip in case A. cerana and A. dorsata was in between the two. Time spent per flowers also varied significantly (Table 3) during different days of observation. A. dorsata spent from a minimum of 6.42 seconds/flowers to a maximum of 7.78 flowers with an average of 6.82 flowers/sec. A. florea spent between 20.34 to 26.24 sec/flower with an average of 23.66 seconds/flower.
Abrol (1986) reported that each bee species has a specific ecological threshold for initiation of flight activity which differs inter and intra specifically, depending upon its level of adaptability in a particular environment. Lerer et al. (1982) reported that in case of Megachile rotundata F. no flight activity was observed before the critical air temperature of 16-17°C was surpassed. Likewise, 15-18°C has been reported for A. florea (Kapil and Brar, 1971) and A. dorsata (Kapil and Kumar, 1974; Abrol, 1985, 1986). Cessation of activity in all the bee species, however, occurred even before the temperature dropped to the levels associated with the commencement of their activity. Evidently, cessation of flight activity was independent of prevailing air temperature and may possibly be governed by diminishing light intensity and solar radiation (Lerer et al., 1982; Abrol and Kapil, 1986).
4. Foraging speed of pollinating insects
The foraging speed (time spent/flower) is a significant consequence to analyze the efficiency of insect pollinators. The variation of time spent/flower attribute may be due to foraging attributes and internal competition. Vázquez et al. (2005) reported importance of foraging speed as a proxy for pollination success. The foraging speed varied for each pollinator group across populations (Brunet, 2009). The pollinators’ time spent/flower vary during foraging (Vicen and Bosch, 2000; Monzon et a|., 2004; Stout, 2007).
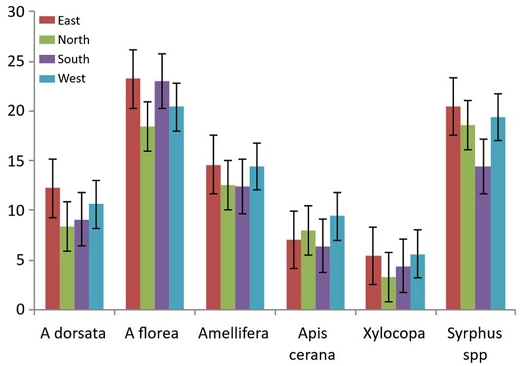
5. Impact of inflorescence direction on abundance of flower visiting insects
The data presented in Table 3, Fig. 1 shows impact of different directions of branches on the number of bees visiting flowers of Citrus sinensis. The studies revealed that population of bees varied with direction of inflorescence with respect to sun. In the earlier hours of the foraging population were more abundant in inflorescence facing east and west and in between north and south. All the honeybee species Apis dorsata, A. florea, A. mellifera, A. cerana, carpenter bee Xylocopa spp. and Syphus spp. were relatively more in numbers on east facing branches with an average of 14.82 of all the visitors. Followed by west with mean of 14.28, and 11.14 and 12.20 for south and north, respectively. The population density of different bee species varied in different directions and was in the order A. dorsata east>west>south>north. The same trend was observed in case of other bee species and Syrphus. Overall, abundance of bees on all branches was 10.47, 21.99, 13.73, 8.36, 4.93 and 18.94 for A. dorsata, A. florea, A. mellifera, Apis cerana, Xylocopa and Syrphus spp., respectively. The study revealed that population dynamics of different species differed significantly from each other. However, the overall differences in population dynamics of different insect visitors on east and west directions were significantly higher compared to north and south directions different directions were not much pronounced.
Analysis of data revealed that abundance of bees of differed significantly from one bee species to another and on different directions of canopy. The east and west directions attracted significantly a greater number of bees compared to north and south directions. Population of bees was at par on north and south branches. Abundance of bees differed significantly depending on both species and inflorescence direction (Two-way ANOVA: bee species, F5,46=393.56, p<0.001; direction, F3,46=46.53, p<0.001; interaction, F15,46=5.09, p<0.001).
This may be due to the fact that pollinator may change the directionality of foraging movements in response to flower orientation (Ginsberg, 1983; Collevatti, 1998), plant density (Handel, 1983; Maasse, 1992; Morris, 1993), prevailing wind condition (Dafni, 1992; Dag and Eisikowitch, 1995) and the occurrence of other pollinators (Stanghellini et al., 2002). A pollinator can easily move across rows if the next closest flower is in that direction instead of down the row.
6. Abundance of insect pollinators at different inflorescence heights
In between initiation and cessation, interesting foraging differences between different flower visitors were observed (Table 4). There was a complete resource partitioning between different flower visitors like honeybees, large sized carpenter bees and flies and number of small solitary bees referred to as ‘others’. All the honeybee species A. cerana, A. mellifera, A. dorsata and A. florea were more abundant on middle branches 2-3 m above ground and relatively few populations reckoned on lower or upper branches on all the days of observations. Their percentage abundance was in the order middle branches>upper branches>lower branches. Whereas in case of Carpenter bees Xylocopa fenestrata and X. pubescens reverse was true. Maximum populations were recorded on upper or top branches. Their abundance was in the order: Upper branches>middle branches>lower branches. In case of dipteran flies such as Musca spp., Syrphus spp. and small solitary bees Lassioglossum spp. and Nomia spp. were more abundant on lower branches as compared to middle or top branches. Their abundance was in the order: Lower branches>upper branches>middle branches.

Percentage proportion Foraging population of insects visiting citrus flowers at different positions of the flowering branches
Abundance of bees at different heights on branches categorized as lower, middle and upper branches differed significantly. One way ANOVA (Table 5) revealed significant differences for all bee species.
The data presented in Table 7 on morphometrics of bees revealed that different insect visitors differed in their body size. Xylocopa pubescens and X. fenestrata were larger in size in terms of body weight, body length, tongue length and length of forewing, followed by A. dorsata, A. mellifera, A. cerana and A. florea in the order: carpenter bees>honeybees>other insects. The differential preference for floral heights seems to be related to their body size and energy requirements. On upper branches competition for floral resources is less compared to middle and lower branches which can meet energy requirements of large pollinators. Similarly, honeybees which are relatively less energy demanding compared to carpenter bees can suffice their energy demands from middle branches. The ‘other’ visitors which included syrphids and smaller insects were more abundant on lower branches and could meet their nutritional requirements from lower branches where resources were depleted at fast rate. Similar results have been reported by earlier investigators (Heinrich, 1975; Abrol, 1992).
The results reveal that visitation pattern is influenced by body size and energy requirements of the floral visitors. This phenomenon of vertical stratification leads to a corresponding stratification of the fauna that is dependent on floral resources in a variety of ways. Changes in inflorescence height are associated with both biotic and abiotic changes, such as floral species composition, temperature, wind, foliage density, and resource abundance (Walther, 2002). All of these changing variables allow for variation in resources and microhabitats, creating a vertical gradient of niche space. Thus, different heights may host different communities of organisms adapted to the conditions and resources of a particular strata. As well, species with the same resource requirements experience reduced competitive interference due to this higher availability of niches (Schulze et al., 2001).
Smaller pollinators mostly confined their visits to nearby and branches close to the ground. Because of their relatively low energy demands as compared to large pollinators, inspite of large population could satisfy their energy needs. Large bee pollinators like Xylocopa species with very higher energy requirements visited top most branches which had lower population of other pollinators and could harvest more energy compared to lower branches where population densities of pollinators were much higher which could deplete nectar sources. Some of the flowers which were not visited by bees may act as lucky hits containing higher amount of nectar (Southwick et al., 1981a, 1981b).
Similar results were obtained by Chauhan (1985) who found that honeybees visiting apple flowers were more abundant on middle branches than the upper or lower ones. Results are in agreement with earlier studies conducted by Abrol (1988) who found that honeybee mostly visited middle branches whereas bumblebees collected nectar and pollen from top most branches. Large bees have relatively higher energy requirements and can forage at lower temperatures compared to small bees (Abrol, 1992). Klecka et al. (2018) also reported pollinator plant interaction influenced by plant stratification. In a similar study, Joshi and Joshi (2015) determined the height preferences of foraging bees on apple flowers. Population count of bees on marked branches of apple trees at different heights showed that A. cerana was more abundant on middle (50.2%) (at a height of about 4 m) than lower (31.7%) and top (19.1%) heights of trees. Similarly, A. mellifera again preferred middle heights (51.6%) followed by top (25.8%) and lower (23.6%). Partitioning of floral resources is attributed to different size and energy requirements of bees.
This behaviour pattern of flower visiting insects into stratification helps resource partitioning and avoids competition for common floral resource. Since the pollinating insects differ in their body size and have different energy requirements. Partitioning of floral resources is beneficial both for pollinators as well as the plant resulting in a mutualistic relationship.
CONCLUSIONS
In conclusion, our results emphasize that special attention needs to be paid to the vertical stratification of habitats that supports various floral and faunal species. The studies reveal that height preferences are related to body size and energetic requirements. The studies shall help to understand the behavioural ecology and physiology of bees. The implications of this research are far-reaching. The quantification of biodiversity allows for a deeper understanding of community structure and of the species that need to be protected, especially if they perform a crucial ecosystem service that maintains the integrity of the whole ecosystem. Further investigations are needed to explore the pollinator behaviour in relation to the height of the plant as well directionality of inflorescences. In tree type plants, it is always advisable to record observations at different strata to obtain clear estimates of pollinator diversity and abundance. Recording observations by observing one set of branches is unlikely to yield good results. The concentration of floral resource composition in the upper canopy supports the greater biodiversity of faunal pollinators.
Another most important conclusion from this study relates to the impact of direction of plant on abundance of foraging population of insects. Generally, more population was observed in east and west compared to north and south directions. Population of bees were at par in north and south directions. This is likely to be related to different intensities of light and radiations, wind velocity and prevailing temperature and other microclimatic conditions. Further observations are required to obtain data on seed set of individual plants corresponding to pollinator-mediated selection of inflorescence height and directionality. However, the study is limited in its ability to provide precise estimates of pollinator abundance and diversity across different heights, as well as their corresponding impacts on seed set and fruit production in various plant species. Additionally, our ability to observe and identify organisms was constrained by the visual limitations and the fixed position of the canopy tower. Since visual observation was only available method, future studies could enhance species identification accuracy through catch-and-release techniques. Moreover, incorporating an additional study site with similar characteristics would help validate the observed trends, as floral composition influences resource availability. Expanding the number of sample sites could also open avenues for further research into the vertical stratification of pollinators and seed dispersers. Given the critical role of pollination, efforts should be directed toward mitigating the loss of pollinators to ensure sustainable pollination services. This can be achieved by recognizing their intrinsic value, emphasizing the essential contribution of pollinators to Citrus productivity, and prioritizing the management of alternative pollinators.
Acknowledgments
The generous funding by the Indian Council of Agricultural Research under HTMM-1 (2.28) is gratefully acknowledged. Thanks, are also due to the Editor and two anonymous reviewers for their critical and very useful suggestions on an earlier draft of this paper. I am highly thankful to Dr. Manish Sharma Professor and Head, Division of Agricultural Statistics & Computer Sciences, Sher-e-Kashmir University of Agricultural Sciences and Technology of Jammu, Jammu, India.
References
- Abrol, D. P. 1992. Bioenergetics in bee flower interrelationships - An analysis of foraging behaviour. Korean J. Apic. 7(1): 39-66.
- Abrol, D. P. 1985. Analysis of biophysical interactions in causing foraging behaviour of some bees - A study in bioenergetics. PhD thesis Haryana Agricultural university, Hissar 286.
- Abrol, D. P. 1986. Eco physiological adaptations between pollinating bees and their flowers. Environ. Ecol. 4: 161-162.
- Abrol, D. P. 1988. Ecology and behaviour of three bee species pollinating loquat (Eriobotrya japonica Lindley). Proc. Indian Natn. Sci. Acad. B 54(2): 161-163.
-
Abrol, D. P. and R. P. Kapil. 1986. Factors affecting pollination activity of Megachile lanata Lepel. Proc. Indian Acad. Sci. (Anim Sci.) 95: 757-769.
[https://doi.org/10.1007/bf03179493]

- Abrol, D. P. 2012. Pollination biology- biodiversity conservation and agricultural productivity. Springer Dordrecht Heidelberg London New York, 787 pp.
- Appanah, S. 1981. Pollination in Malaysian primary forest. Malay. For. 44: 37-42.
-
Ashman, T. L., T. M. Knight, J. Steets, P. Amarasekare, M. Burd, D. R. Campbell, M. R. Dudash, M. O. Johnston, S. J. Mazer, R. J. Mitchell, M. T. Morgan and W. Wilson. 2004. Pollen limitation of plant reproduction: ecological and evolutionary causes and consequences. Ecology 85: 2408-2421.
[https://doi.org/10.1890/03-8024]

-
Bawa, K. S., S. H. Bullock, D. R. Perry, R. E. Coville and M. H. Grayum. 1985. Reproductive biology of tropical lowland rainforest trees. Part 2. Pollination systems. Am. J. Bot. 72: 346-356.
[https://doi.org/10.1002/j.1537-2197.1985.tb05358.x]

- Bhatia, R., D. Gupta, J. S. Chandel and N. K. Sharma. 1995. Relative abundance of insect visitors on flowers of major subtropical fruits in Himachal Pradesh and their effect on fruit set. Indian J. Agric. Sci. 65(12): 907-912.
-
Brunet, J. 2009. Pollinators of the Rocky Mountain columbine: temporal variation, functional groups and associations with floral traits. Ann. Bot. 103: 1567-1578.
[https://doi.org/10.1093/aob/mcp096]

- Castro, M. S. 2002. Bee fauna of some tropical and exotic fruits: potential pollinators and their conservation. In: Kevan, P. and Imperatriz Fonseca, V.L. (eds.). The Pollinating Bees: The Conservation link between Agriculture and Nature. Brasília, DF. Ministério Meio Ambiente: 275-288.
- Chacoff, N. P. and M. A. Aizen. 2006. Edge effects on flower-visiting insects in grapefruit plantations bordering premontane subtropical forest. J. Appl. Ecol. 43: 18-27.
-
Chacoff, N. P. and M. A. Aizen, 2007. Pollination requirements of pigmented grapefruit (Citrus paradisi Macf.) from Northwestern Argentina. Crop Sci. 47: 1143-1150.
[https://doi.org/10.2135/cropsci2006.09.0586]

- Chao, C. C. T. 2005. Pollinations study of mandarins and the effect on seediness and fruit set: implications for seedless mandarins production. HortScience 40: 362-365.
- Chauhan, P. S. 1985. Studies on pollinating ecology of apple orchards by hymenopterous insects in Narkanda and Matina areas of Himachal Pradesh. M. Phil. Dissertation, Himachal Pradesh University Shimla 101 pp.
-
Collevatti, R. G. 1998. Pollination ecology of the tropical weed Triumfetta semitriloba Jacq. (Tiliaceae), in south-eastern Brazil. Rev. Bras. Biol. 58: 383-392.
[https://doi.org/10.1590/s0034-71081998000300004]

-
Cuenca, J., A. Garcia-Lor, L. Navarro and P. Aleza. 2016. Citrus genetics and breeding, In Al-Khayri, J. M., S. M. Jain, and D. V. Johnson (Eds.): Advances in plant breeding strategies: fruits (pp. 403-436). Cham: Springer International Publishing.
[https://doi.org/10.1007/978-3-319-91944-7_11]

-
da Santos, R. S., L. T. Carneiro, J. P. de Oliveira Santos, M. M. da Silva, M. de Oliveira Milfont and C. C. Castro. 2021. Bee pollination services and the enhancement of fruit yield associated with seed number in self-incompatible tangelos. Scientia Horticulturae 276: 10974.
[https://doi.org/10.1016/j.scienta.2020.109743]

- Dafni, A. 1992. Pollination Ecology: A Practical Approach. Oxford University Press, Oxford.
-
Dag, A. and D. Eisikowitch, 1995. The influence of hive location on honeybee foraging activity and fruit set in melons grown in plastic greenhouses. Apidologie 26: 511-519.
[https://doi.org/10.1051/apido:19950608]

-
Espíndola, A., L. Pellissier and N. Alvarez. 2011. Variation in the proportion of flower visitors of Arum maculatum along its distributional range in relation with community-based climatic niche analyses. Oikos 120(5): 728-734.
[https://doi.org/10.1111/j.1600-0706.2010.18937.x]

-
Faulkner, G. 1976. Honeybee behaviour as affected by plant height and flower colour in brussels sprouts. J. Apic. Res. 15(1): 15-18.
[https://doi.org/10.1080/00218839.1976.11099827]

- Frankie, G. W. and R. Coville. 1979. An experimental study on the foraging behavior of selected solitary bee species in the Costa Rican dry forest (Hymenoptera, Apoidea). J. Kans. Entomol. Soc. 52: 591-602.
- Frankie, G. W. and W. A. Haber. 1983. Why bees move among mass flowering Neotropical trees. In Jones, C. E. and R. J. Little (Eds.). Handbook of experimental pollination biology. Scientific and Academic Editions, New York, USA.
- Free, J. B. 1962. Studies on the pollination of fruit trees by honey bees. J. Royal Hortic. Soc. 87: 302-309.
- Free, J. B. 1993. Insect Pollination of Crops. New York Academic Press, London, UK.
-
Gaines-Day, H. R. and C. Gratton. 2015. Biotic and abiotic factors contribute to cranberry pollination. J. Pollin. Ecol. 15: 15-22.
[https://doi.org/10.26786/1920-7603(2015)2]

- Ghosh, S. P. 1990. Citrus, in: Fruits, Tropical and Subtropical, pp. 63-131.
-
Ginsberg, H. S. 1983. Foraging ecology of bees in an old field. Ecology 64: 165-175.
[https://doi.org/10.2307/1937338]

-
Gumbert, A. and J. Kunze. 1999. Inflorescence Height Affects Visitation Behavior of Bees-A Case Study of an Aquatic Plant Community in Bolivia. Biotropica 31(3): 466-477.
[https://doi.org/10.1111/j.1744-7429.1999.tb00389.x]

-
Gurung, S. and A. Chettri. 2021. Threat to citrus in a global pollinator decline scenario: current understanding of its pollination requirements and future directions. Plant Reprod. Ecol. Recent. Adv. 134: 23-38.
[https://doi.org/10.5772/intechopen.101159]

-
Handel, S. N. 1983. Contrasting gene flow patterns and genetic subdivision in adjacent populations of Cucumis sativus (Cucurbitaceae). Evolution 37: 760-771.
[https://doi.org/10.1111/j.1558-5646.1983.tb05598.x]

- Hassanein, M. H. and M. M. Ibrahim. 1959. Studies on the importance of insects specially the honey bee in pollination of citrus in Egypt. The Agricultural Research Review 37(3): 390-409.
- Hearn, C. J., P. C. Reece and R. Fenton. 1968. Effects of pollen source on fruit characteristics and set of four citrus hybrids. Proc. Florida State Hortic. Soc. 81: 94-98.
- Heinrich, B. 1975. Energetics of pollination. Ann. Rev. Ecol. Syst. 6: 139-170.
-
Hoehn, P., T. Tscharntke, J. M. Tylianakis and I. Steffan-Dewenter. 2008. Functional group diversity of bee pollinators increases crop yield. Proc. Biol. Sci. 275(1648): 2283-2291.
[https://doi.org/10.1098/rspb.2008.0405]

-
Jiang, X. F. and Q. J. Li 2017. Self- and intra-morph incompatibility and selection analysis of an inconspicuous distylous herb growing on the Tibetan plateau (Primula tibetica). Ecol. Evol. 7: 5746-5753.
[https://doi.org/10.1002/ece3.3151]

- Joshi, N. C. and P. C. Joshi. 2015. Foraging Behaviour of Apis Spp. on Apple Flowers in a Subtropical Environment. New York Sci. J. 3(3): 71-76.
-
Junker, R. R., N. Blüthgen, T. Brehm, J. Binkenstein, J. Paulus, H. M. Schaefer and M. Stang. 2013. Specialization on traits as basis for the niche-breadth of flower visitors and as structuring mechanism of ecological networks. Functional Ecology 27: 329-341.
[https://doi.org/10.1111/1365-2435.12005]

- Kapil, R. P. and H. S. Brar. 1971. Foraging behaviour of Apis florea F. in relation to Brassica campestris var. toria. Proc. 12th Inl. Apic. Cong. Moscow 335-340.
- Kapil, R. P. and S. Kumar. 1974. Foraging behaviour of Apis dorsata F. workers on Brassica I Hook. J. Bomb. Nat. Hist. Soc. 72: 327-332.
- Karmakar, P. 2013. Pollination biology of Citrus aurantifolia (Christm Swingle), a medicinally important fruit plant. Int. J. Innov. Res. Dev. 2(7): 138-142.
-
Klecka, J., J. Hadrava and P. Koloušková. 2018. Vertical stratification of plant-pollinator interactions in a temperate grassland. PeerJ 22:6: e4998.
[https://doi.org/10.7717/peerj.4998]

-
Lerer, H., W. G. Bailey, P. F. Mills and M. Pankiw. 1982. Pollination activity of Megachile roundata (Hymenoptera: Apoidea). Environ. Ent. 1: 997-1000.
[https://doi.org/10.1093/ee/11.5.997]

-
Levin, D. A. and H. W. Kerster. 1973. Assortative pollination for stature in Lythrum, salicaria. Evolution 27(1): 144-152.
[https://doi.org/10.2307/2407128]

-
Maasse, R. 1992. Ecological risks of transgenic plants: Effects of spatial dispersion on gene flow. Ecol. Appl. 2: 431-438.
[https://doi.org/10.2307/1941878]

-
Malerbo-Souza, D. T., R. H. Nogueira-Couto and L. A. Couto. 2004. Honey bee attractants and pollination in sweet orange, Citrus sinensis (L.) Osbeck, var. Pera-Rio. J. Venom. Anim. Toxins Incl. Trop. Dis. 1: 144-153.
[https://doi.org/10.1590/s1678-91992004000200004]

-
Manzoor-ul-Haq, M. Rafie-ul-Din and A. Ghaffar. 1978. Effect of insect pollination on fruit bearing in Kinnow Mandarin (Citrus reticulata), and physical and chemical properties of the fruit. J. Apic. Res. 17: 47-49.
[https://doi.org/10.1080/00218839.1978.11099900]

- Mehmood, K., H. Shahid, M. Naveed, B. Imran and A. Munir. 2015. Insect pollinators visiting citrus (Citrus limon) and Avocardo (Persea americana) fruit trees. Asian J. Agri. Biol. 3(1): 23-27.
- Michener, C. D. 2000. The bees of the world. The John Hopkins University Press, Baltimore/ London, 913 p.
-
Monzon, V. H., J. Bosch and J. Retana. 2004. Foraging behaviour and pollinating effectiveness of Osmia cornuta (Hymenoptera: Megachilidae) and Apis mellifera (Hymenoptera: Apidae) on “Comice” pear. Apidologie 35: 575-585.
[https://doi.org/10.1051/apido:2004055]

-
Morris, W., M. Price, N. Waser, J. Thomson, B. Thomson and D. Stratton. 1994. Systematic increase in pollen carryover and its consequences for geitonogamy in plant populations. Oikos 71(3): 431-440.
[https://doi.org/10.2307/3545831]

-
Newman, E., J. Manning and B. Anderson. 2015. Local adaptation: Mechanical fit between floral ecotypes of Nerine humilis (Amaryllidaceae) and pollinator communities. Evolution 69(9): 2262-2275.
[https://doi.org/10.1111/evo.12736]

-
Olesen, J. M., J. Bascompte, H. Elberling and P. Jordano. 2008. Temporal dynamics in a pollination network. Ecology 89(6): 1573-1582.
[https://doi.org/10.1890/07-0451.1]

- Oliver, I. and A. J. Beattie. 1993. A Possible Method for the Rapid Assessment of Biodiversity. Conserv. Biol. 7: 562-568.
-
Opler, P. A., H. G. Baker and G. W. Frankie 1980. Plant reproductive characteristics during secondary succession in Neotropical lowland forest ecosystems. Biotropica 12: 40-46.
[https://doi.org/10.2307/2388155]

-
Papadakis, I. E., E. E. Protopapadakis and I. N. Therios. 2009. Yield and fruit quality of ‘Nova’ hybrid [Citrus clementina Hort. ex Tanaka (C. reticulata Blanco×C. paradisi Macfad)] and two Clementine varieties (C. clementina Hort. ex Tana Ka) as affected by self- and cross-pollination. Sci. Hortic. 121: 38-41.
[https://doi.org/10.1016/j.scienta.2009.01.011]

-
Peakall, R. and S. N. Handel. 1993. Pollinators discriminate among floral heights of a sexually deceptive orchid: implications for selection. Evolution 47(6): 1681-1687.
[https://doi.org/10.1111/j.1558-5646.1993.tb01260.x]

-
Perry, D. R. and A. Starrett. 1980. The pollination ecology and blooming strategy of a neotropical emergent tree, Dipteryx pamamensis. Biotropica 12: 307-313.
[https://doi.org/10.2307/2387702]

-
Pyke, G. H. 1978. Optimal foraging: movement patterns of bumblebees between inflorescences. Theo. Pop. Biol. 13(1):72-98.
[https://doi.org/10.1016/0040-5809(78)90036-9]

-
Roubik, D. W., D. J. Ackerman, C. Christine and H. S. Brian. 1982. Stratum, Tree, and Flower Selection by Tropical Bees: Implications for the Reproductive Biology of Outcrossing Cochlospermum Vitifolium in Panama. Ecology 63(3): 712-720.
[https://doi.org/10.2307/1936792]

-
Roussos, P. A. 2015. Orange (Citrus sinensis Osbeck). In Nutritional Composition of Fruit Cultivars (pp. 469-496). Elsevier Inc.
[https://doi.org/10.1016/b978-0-12-408117-8.00020-9]

- Sanford, M. T. 1992. Pollination of Citrus by Honey Bees (University of Florida Cooperative Extension Service, Institute of Food and Agriculture Sciences, EDIS, 1992).
- Sanford, M. T. 2003. Pollination of citrus by honeybees. University of Florida IFAS Extension http://edis.ifas.ufl.edu/pdffiles/AA/AA09200.pdf
-
Schulze, C. H., K. E. Linsenmair and K. Fiedler. 2001. Understorey Versus Canopy: Patterns of Vertical Stratification Diversity among Lepidoptera in a Bornean Rain Forest. Plant Ecol. 153: 133-152.
[https://doi.org/10.1023/a:1017589711553]

- Singh, A. K. 2016. Insect pollinators of sweet orange and their attributes. Indian J. Entomol. 78(1): 46-50.
-
Sletvold, N., J. M. Grindeland and J. A. Gren. 2010. Pollinator-mediated selection on floral display, spur length and flowering phenology in the deceptive orchid Dactylorhiza lapponica. New Phytol. 188(2): 385-392.
[https://doi.org/10.1111/j.1469-8137.2010.03296.x]

- Snedecor, G. W. and W. G Cochran. 1989. Statistical methods (8th edition). Iowa University Press, Ames, IA.
-
Southwick, E. E., G. M. Loper and S. E. Sadwick. 1981a. Nectar, Energy, Bees, and Microclimate. BioScience 31(4): 324-326.
[https://doi.org/10.2307/1308152]

-
Southwick, E. E., G. M. Loper and S. E. Sadwick. 1981b. Nectar Production, Composition, Energetics and Pollinator Attractiveness in Spring Flowers of Western New York. Amer. J. Bot. 68(7): 994-1002.
[https://doi.org/10.2307/2443231]

-
Stanghellini, M. S., J. R. Schultheis and J. T. Ambrose. 2002. Pollen mobilization in selected Cucurbitaceae and the putative effects on pollinator abundance on pollen depletion rates. J. Amer. Soc. Hort. Sci. 127: 729-736.
[https://doi.org/10.21273/jashs.127.5.729]

-
Stout, J. C. 2007. Pollination of invasive Rhododendron ponticum (Ericaceae) in Ireland. Apidologie 38: 198-206.
[https://doi.org/10.1051/apido:2006071]

-
Trunschke, J., N. Sletvold and A. J. Gren. 2017. Interaction intensity and pollinator-mediated selection. New Phytologist 214(3): 1381-1389.
[https://doi.org/10.1111/nph.14479]

-
Vanlalhmangaiha, R., H. K. Singh, T. Boopathi, S. Lalhruaitluangi and T. T. Sangma. 2023. Impact of insect pollination on the quantitative and qualitative characteristics of sweet orange, Citrus sinensis (L.) Osbeck. J. Apic. Res. 62: 767-776.
[https://doi.org/10.1080/00218839.2021.2013401]

-
Vázquez, D. P., W. F. Morris and P. Jordano. 2005. Interaction frequency as a surrogate for the total effect of animal mutualists on plant. Ecol. Lett. 8: 1088-1094.
[https://doi.org/10.1111/j.1461-0248.2005.00810.x]

-
Vicens, N. and J. Bosch. 2000. Pollinating efficacy of Osmia cornuta and Apis mellifera (Hymenoptera: Megachilidae, Apidae) on ‘Red Delicious’ apple. Environ. Entomol. 29(2): 235-240.
[https://doi.org/10.1093/ee/29.2.235]

-
Vithanage, V. 1991. Effect of different pollen parents on seediness and quality of ‘Ellendale’ tangor. Sci. Hortic. 48: 253-260.
[https://doi.org/10.1016/0304-4238(91)90133-j]

-
Wallace, H. M. and L. S. Lee. 1999. Pollen source, fruit set and xenia in mandarins. J. Hortic. Sci. Biotechnol. 74: 82-86.
[https://doi.org/10.1080/14620316.1999.11511077]

-
Wallace, H. M. 2004. Pollination effects on quality in ‘oroval clementine’ mandarin in Australia. Acta Hortic. 632: 99-103.
[https://doi.org/10.17660/actahortic.2004.632.12]

-
Walther, B. A. 2002. Vertical Stratification and Use of Vegetation and Light Habitats by Neotropical Forest Birds. J. Ornithol. 143: 64-81.
[https://doi.org/10.1007/bf02465460]

-
Wu, G. A., J. Terol, V. Ibanez, A. López-García, E. Pérez-Román, C. Borredá, C. Domingo, E. R. Tadeo, J. Carbonell-Caballero, R. Alonso, F. Curk, D. Du, P. Ollitrault, M. L. Roose, J. Dopazo, F. G. Gmitter, D. S. Rokhsar and M. Talon. 2018. Genomics of the origin and evolution of Citrus. Nature 554(7692): 311-316.
[https://doi.org/10.1038/nature25447]

- Yildiz, E. and M. Kaplankiran. 2017. The effect of cross-pollination on fruit set and quality in ‘Robinson’ and ‘Fremont’ mandarins. Ege Üniv Ziraat Fak Derg 54: 107-112.