
Antibacterial Activity of Honey Bee Venom against Multidrug-resistant S. aureus and E. coli Isolated from Bovine Mastitis
Abstract
Bee venom (BV) consists of various bio-actives, including proteins, peptides, enzymes, and volatile metabolites. Over the last decade, the antimicrobial effect of BV against bacterial pathogens has been demonstrated through various in vitro and in vivo experiments. Because mastitis has a serious impact on the dairy industry, the present study aimed to evaluate the antibacterial activity of BV against multidrug-resistant S. aureus and E. coli, which are difficult to treat using antibiotics. In this study, 25 E. coli and 15 S. aureus strains from bovine mastitis were used for BV MIC testing. Based on the antibiotic resistance pattern, all isolates were categorized into three groups: susceptible, resistant, and multidrug-resistant. BV MICs for the E. coli and S. aureus isolates ranged from 16-128 μg/mL and 16-32 μg/mL, respectively. There were no significant differences between the antibiotic resistance and BV MICs of the isolates. However, two multidrug resistance (AMP-CEF-OXA-PEN) S. aureus isolates showed 2-fold higher BV MIC values than the other S. aureus isolates. Therefore, BV can be used as an adjunct treatment for mastitis caused by antibiotic-resistant bacteria. Further studies are needed to understand the resistance mechanisms of multidrug-resistant bacteria, and in vivo studies are required to evaluate the efficacy and safety of BV.
Keywords:
Bee venom, Mastitis, Multidrug-resistant bacteriaINTRODUCTION
Bovine mastitis (BM) is one of the most economically significant diseases that decreases milk production in dairy cattle. BM is an inflammatory disease of the mammary glands associated with the infection by several bacterial pathogens (Gomes et al., 2016). Many studies have analyzed to identify the primary cause and the major microbial pathogens causing mastitis (Ma et al., 2020; Sokolov et al., 2021). It was reported that mastitis is usually caused by Escherichia coli, Streptococcus spp., and Staphylococcus aureus (Dalanezi et al., 2020).
E. coli is the most common Gram-negative bacterium among the pathogens that cause acute mastitis during early lactation. E. coli-related mastitis, the common symptoms of the udder include redness, swelling, tenderness, and an abnormal texture of milk (Burvenich et al., 2003; Zhang et al., 2018).
S. aureus, one of the primary mastitis pathogens worldwide, causes chronic intramammary infections that respond poorly to antibiotics, disseminate within the herd, and lead to high economic losses. Classic mastitis control programs are based on hygiene and antibiotic therapy (Günther et al., 2017; Cheng and Han, 2020; Duse et al., 2021).
Antibiotics are the most commonly used control measures owing to their remarkable effectiveness (Duse et al., 2021). Several antibiotics, including penicillin, ampicillin, tetracycline, and gentamicin, have been used to treat mastitis, and antibiotic treatment largely relies on the primary pathogen/s of mastitis (Gomes and Henriques, 2016). However, the misuse of antibiotics has led to the development of multidrug-resistant bacteria. In addition, the use of antibiotics in the treatment of mastitis directly affects consumers through the presence of antibiotic residues in milk (Gomes and Henriques, 2016; Cheng and Han, 2020). Antimicrobial drug resistance in pathogenic bacteria has become a present danger for many countries (WHO), and only a few alternatives are currently available (Cheng and Han, 2020). Therefore, the search for new candidates with novel modes of action is required.
Natural products, including plant- and animal-based products, are rich in bioactive compounds that exhibit diverse activities against various diseases (Cheng and Han, 2020). Among them, bee venom (BV) is a venomous cocktail of proteins, peptides, enzymes, and volatile metabolites secreted by the poison glands of honeybee as a protective mechanism. BV also contains many biochemically and pharmacologically active substances. Melittin is a compound that represents 40-60% of the dry BV weight (Abd El-Wahed et al., 2019). Owing to its constituents, BV has been proven to be an effective anti-inflammatory and antibacterial agent against several gram-positive (Staphylococcus aureus and Streptococcus) and gram-negative (Escherichia coli, Klebsiella and Pseudomonas) bacterial strains (Wehbe et al., 2019; Srichok et al., 2022). AL-Ani et al. (2015) reported that BV and its constituents, together with chemotherapy agents (vancomycin, oxacillin, and amikacin), have a synergistic effect against multidrug resistance bacteria.
Several studies have demonstrated the effectiveness of BV against mastitis pathogens, including E. coli and S. aureus (Han et al., 2007; Park et al., 2013). However, these studies have primarily focused on evaluating the antibacterial activity of BV. Therefore, this study aimed to compare the antibacterial activity of BV against antibiotic-susceptible, antibiotic-resistant, and multidrug-resistant E. coli and S. aureus isolated from cows with mastitis in Korea. In this study, we confirmed the applicability of bee venom to mastitis caused by multidrug-resistant bacteria for which antibiotics are difficult to use.
MATERIALS AND METHODS
1. Collection and preparation of bee venom (BV)
BV was supplied by the Department of Agricultural Biology of the National Institute of Agricultural Science, Rural Development Administration, Korea. BV was collected from honeybees, Apis mellifera, using electric BV-collection devices (Chungjin Biotech, Ansan, Korea) and purified under sterile laboratory conditions. Briefly, the devices were placed on the hive, and the honeybees were given weak electric stimulation to cause them to sting a glass plate. After drying the BV for sufficient time, it was collected by scraping. The collected BV was washed in sterile water and centrifuged at 10,000×g at 4℃ for 5 min to remove the residues. BV was lyophilized by freeze-drying and refrigerated at 4℃ for later use. Purified BV was diluted in sterile distilled water and aseptically filtered through a 0.45 μm syringe filter (Advantec, Tokyo, Japan). BV was prepared in two-fold concentration series ranging from 0.5-512 μg/mL for the antibacterial experiment.
2. Bacteria and culture conditions
E. coli (n=25) and S. aureus (n=15) were isolated from milk samples of cows diagnosed with mastitis at dairy farms in Jeollabuk-do and Chungcheongbuk-do from 2019-2020. The strains were cultured on Blood agar (Kisan Biotech, Seoul, Korea) medium at 37℃, aerobic conditions for 24 h.
3. Minimum inhibitory concentration (MIC)
A broth microdilution assay was performed to evaluate the antibacterial effects of BV against the isolates. Two-dilution series of BV was prepared by adding 90 μL of Muller-Hinton (MH) broth and 10 μL of BV samples and then sequentially adding it to a 96-well plate to obtain final concentrations of 0, 0.5, 1, 2, 4, 8, 16, 32, 64, 128, 256 and 512 μg/mL. Each bacterial strain was cultured in MH broth, diluted, and adjusted to 0.5 McFarland standard by Sensititre reader (Thermo Fisher Scientific, US). Each 100 μL of bacterial suspension was added to a 96-well plate containing the two-diluted BV and incubated at 37℃ for 18 h. Bacterial growth was visually confirmed and the concentration at which proliferation did not occur was expressed as the minimum growth-inhibitory concentration. If there was a difference in the minimum growth inhibition concentration among strains of the same species, the minimum (Min) and maximum (Max) values were confirmed.
4. Antimicrobial susceptibility
MICs of E. coli were determined by the standard micro broth dilution method using the Sensititre system (TREK Diagnostic System, East Grinstead, UK) with antimicrobial testing plates containing the following 16 antimicrobials: Amoxicillin/Clavulanic Acid (AMC), Ampicillin (AMP), Cefepime (CEF), Cefoxitin (FOX), Ceftazidime (CAZ), Ceftiofur (CTF), Chloramphenicol (CHL), Ciprofloxacin (CIP), Colistin (CL), Gentamicin (GEN), Meropenem (MER), Nalidixic Acid (NAL), Streptomycin (STR), Sulfisoxazole (SXZ), Tetracycline (TET), and Trimethoprim/Sulphamethoxazole (TMP-SXT). Similarly, the MICs of S. aureus were determined using antimicrobial testing plates containing the following 10 antimicrobials: ampicillin (AMP), ceftiofur (CTF), cephalothin (CEP), erythromycin (EM), oxacillin (OXA), penicillin (PCN), penicillin/novobiocin (NB), pirlimycin (PIR), sulfadimethoxine (SDM), and tetracycline (TET). Resistance profiles (resistant, intermediate, or susceptible) were assigned according to Clinical and Laboratory Standards Institute (CLSI) guidelines (CLSI, 2015).
RESULTS AND DISCUSSION
1. Antibiotic resistance and of isolates
All E. coli isolates were susceptible to ampicillin, cefoxitin, ceftazidime, ceftiofur, ciprofloxacin, meropenem, sulfisoxazole, whereas 40% and 36% of isolates were resistant to tetracycline and streptomycin, respectively. Moreover, below 12% of isolates were resistant to amoxicillin/clavulanic Acid, cefepime, chloramphenicol, colistin, gentamicin, nalidixic acid, and trimethoprim/sulfamethoxazole. All S. aureus isolates were susceptible to cephalothin, erythromycin, penicillin/novobiocin, pirlimycin, and tetracycline, whereas 47% and 33% of isolates were resistant to the penicillin and ampicillin, respectively. Additionally, 20% of the isolates were resistant to the ceftiofur and oxacillin, and sulfadimethoxine resistance was shown by 15% of S. aureus isolates (Fig. 1).
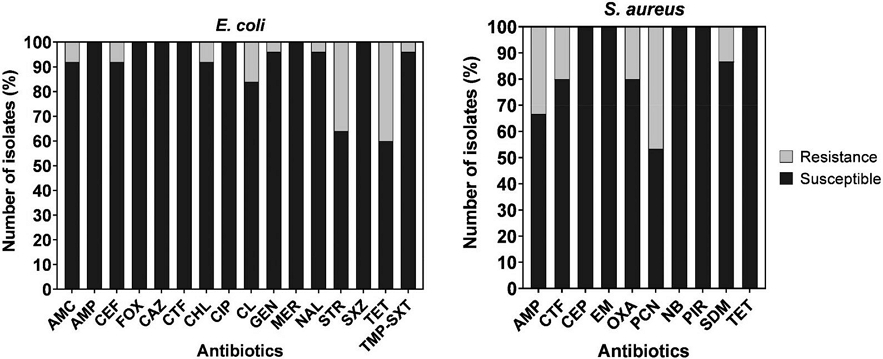
Antibiotic susceptibility of the E. coli (n=25) and S. aureus (n=15) isolates. AMC, Amoxicillin/ Clavulanic Acid; AMP, Ampicillin; CEF, Cefepim; FOX, Cefoxitin; CAZ, Ceftazidime; CTF, Ceftiofur; CHL, Chloramphenicol; CIP, Ciprofloxacin; CL, Colistin; GEN, Gentamicin; MER, Meropenem; NAL, Nalidixic Acid; STR, Streptomycin; SXZ, Sulfisoxazole; TET, Tetracycline; TMP-SXT, Trimethoprim/ Sulphamethoxazole; CEP, Cephalothin; EM, Erythromycin; OXA, Oxacillin; PCN, Penicillin; NB, Penicillin/ Novobiocin; PIR, Pirlimycin; SDM, Sulphadimethoxime, and TET, Tetracycline.
This study used 25 E. coli and 15 isolates of S. aureus obtained from dairy cows with mastitis (Jung et al., 2021). Both these pathogens are the most common causative agents of mastitis worldwide (Gilbert et al., 2013; Gomes et al., 2016; Rana et al., 2022). These two bacteria also show a high percentage of multi-resistance. Therefore, effective control measures against these pathogens are important.
2. Antibiotic resistance patterns of isolates
Based on the pattern of antibiotic resistance, all isolates were categorized into three groups: susceptible, resistant, and multidrug-resistant. Thirteen of the 25 E. coli isolates showed susceptibility to all tested antibiotics. The STR-TET resistance pattern was observed in five E. coli isolates, whereas the remaining resistance patterns were observed in each isolate. Five multidrug resistance patterns (CAZ-GEN-NAL/CAZ-STR-TET/ AMP-STR-TET/ CHL-STR-TET-TMP/SMX/ AMP-CHL-STR-TET) were observed for each isolate (Table 1). Seven of the 15 S. aureus isolates were susceptible to all the tested antibiotics. Among the five resistance patterns, three isolates exhibited the AMP-CEF-OXA-PEN multidrug-resistance pattern (Table 2).
The present study selected different sets of antibiotics to evaluate the antibiotic susceptibility of E. coli and S. aureus depending on the clinical use and CLSI guidelines. E. coli and S. aureus isolates showed different antibiotic resistance patterns. Most of the tested antibiotics were effective against most E. coli and S. aureus isolates. Over 30% of each E. coli and S. aureus isolates were resistant to the two antibiotics. In contrast, E. coli was resistant to tetracycline and streptomycin, whereas S. aureus was resistant to ampicillin and penicillin. Previous studies have reported different antibiotic resistance patterns in E. coli and S. aureus isolates (Oliver and Murinda, 2012; Yu et al., 2020; Mbindyo et al., 2021). Common antibiotics used for the treatment of mastitis include streptomycin, ampicillin, cloxacillin, penicillin, and tetracycline (Bhosale et al., 2014). However, different antibiotics are used for treatment depending on the primary cause of mastitis. In the present study, tetracycline, streptomycin, ampicillin, and penicillin selective pressures due to their overuse in the cattle industry might be the reason for the dissemination of resistant isolates among cows with mastitis.
Furthermore, three S. aureus isolates were resistant to penicillin, amoxicillin, oxacillin, and cefepime, which are potentially risky strains. Similarly, Park et al. (2012) reported that mastitis origin Staphycocci strains showed significantly less resistance to β-lactam antibiotics. Therefore, alternative treatments are required to control these multidrug-resistant strains.
3. Minimum Inhibitory Concentrations (MICs) of bee venom (BV) against E. coli and S. aureus isolates
The MIC values of bee venom (BV) against E. coli and S. aureus isolates showed different trends (Fig. 2). The mean MIC values were 81.3±38.1 μg/mL for E. coli (n=25) and 18.1±5.6 μg/mL for S. aureus (n=15). The MIC values ranged from 16-128 μg/mL for E. coli and 16-32 μg/mL for S. aureus, respectively. There was no significant relationship between BV MICs of E. coli isolates and antibiotic resistance patterns. However, BV MICs of the multidrug resistance isolates were shown to have significantly elevated compared to that in the resistant and susceptible groups.
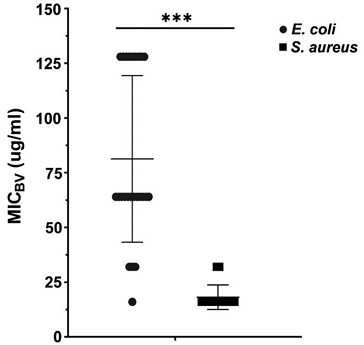
Minimum inhibitory concentrations (MICs) of Bee venom (BV) against E. coli (n=25) and S. aureus (n=15) isolates. *p≤0.05; **p≤0.01; *** p≤0.001, statistical differences referred to BV MICs of E. coli and S. aureus isolates by t-test. Vertical bars show SD.
During the last decade, the antimicrobial action of BV against bacterial pathogens has been demonstrated through various in vitro and in vivo experiments. In the present study, there was a significant difference in the MIC values of BV between E. coli and S. aureus isolates. As opposed to the present study, Han et al. (2007) reported more or less similar BV MICs against mastitis origin E. coli (17.4 μg/mL) and S. aureus (21.4 μg/mL) isolates. Furthermore, another study reported comparatively high BV MICs against E. coli (62.5-250 μg/mL) and S. aureus (62.5-125 μg/mL) isolated from cattle with mastitis in Korea. Similar to the present study, several previous studies have reported a comparatively higher efficacy of BV against Gram-positive bacteria than against gram-negative bacteria, and major component of BV, mellittin, is more effective against Gram-positive than against Gram-negative bacteria (Čujová et al., 2014). Diverse mechanisms may be involved in disrupting the membrane structure, which has a lethal impact on bacterial pathogens.
In this study, isolates were categorized into three groups (susceptible, resistant, and multidrug resistance) depending on their antibiotic susceptibility results, and the MICs of BV against each isolate were compared with the antibiotic resistance patterns. There were no significant differences in the antibiotic resistance patterns and BV MICs of E. coli isolates. However, the two multidrug resistance S. aureus isolates showed significantly higher BV MIC values than the susceptible and resistant groups. Furthermore, both resistant and multidrug resistant isolates showed AMP and PEN resistance, and only CEF and OXA resistance were the excesses of the multidrug nature of the three S. aureus isolates. Common resistance mechanisms might be associated with the use of antibiotics (CEF and OXA) and BV. However, further studies with a higher number of CEF and OXA resistance isolates are needed to confirm this hypothesis.
Mastitis is a major challenge faced by dairy herds worldwide. Many bacterial species can cause infections in the udder, but most infections are caused by specific pathogens, including S. aureus, E. coli, E. faecalis, and Streptococcus spp. (Gomes et al., 2016).
In conclusion, the present E. coli and S. aureus isolates originated from cows with mastitis, and some isolates showed multidrug resistance. Therefore, there is a potential risk of disseminating antibiotic-resistant strains among herds. There were no significant differences between the antibiotic resistance and BV MICs of the isolates. Thus, BV could be used as a substitute for antibiotics against antibiotic-resistant E. coli. However, two multidrug resistance (AMP-CEF-OXA-PEN) S. aureus isolates showed 2-fold high BV MIC values, indicating that common antibacterial mechanisms drive resistance against antibiotics and BV. Further studies are needed to gain deeper insight into these mechanisms. Furthermore, BV can be used effectively to control mastitis causing pathogens, including multidrug-resistant bacteria; however, in vivo studies are required to evaluate its efficacy and safety.
Acknowledgments
This study was supported by 2022 RDA (Rural Development Administration) fellowship program of NIAS (National Institute of Animal Science) and by the “Cooperative Research Program for Agriculture Science & Technology Development (Project title: Study of immunological responses by the application of bee venom and vaccines, Project No. PJ0142982021)” of RDA, Korea.
DISCLOSURE STATEMENT
The authors reported no potential conflict of interest.
References
-
Abd El-Wahed, A. A., S. A. M. Khalifa, B. Y. Sheikh, M. A. Farag, A. Saeed, F. A. Larik and H. R. El-Seedi. 2019. Bee venom composition: from chemistry to biological activity. Stud. Nat. Prod. Chem. 60: 459-484.
[https://doi.org/10.1016/b978-0-444-64181-6.00013-9]

-
Al-Ani, I., S. Zimmermann, J. Reichling and M. Wink. 2015. Pharmacological synergism of bee venom and melittin with antibiotics and plant secondary metabolites against multi-drug resistant microbial pathogens. Phytomedicine 22: 245-255.
[https://doi.org/10.1016/j.phymed.2014.11.019]

- Bhosale, R., R. A. Osmani, P. P. Ghodake, S. M. Shaikh and S. R. Chavan. 2014. Mastitis: An intensive crisis in veterinary science. Int. J. Pharma Res. Health Sci. 2: 96-103.
-
Burvenich, C., V. Van Merris, J. Mehrzad, A. Diez-Fraile and L. Duchateau. 2003. Severity of E. coli mastitis is mainly determined by cow factors. Vet. Res. 34(5): 521-564.
[https://doi.org/10.1051/vetres:2003023]

-
Cheng, W. N. and S. G. Han. 2020. Bovine mastitis: risk factors, therapeutic strategies, and alternative treatments - a review. Asian-Aust. J. Anim. Sci. 33(11): 1699-1713.
[https://doi.org/10.5713/ajas.20.0156]

- CLSI. Performance Standards for Antimicrobial Susceptibility Testing; Twenty-Fifth Informational Supplement. CLSI document M100-S25. Wayne, PA: Clinical and Laboratory Standards Institute; 2015.
-
Čujová, S., L. Bednárová, J. Slaninová, J. Straka and V. Čeřovský. 2014. Interaction of a novel antimicrobial peptide isolated from the venom of solitary bee Colletes daviesanus with phospholipid vesicles and Escherichia coli cells. J. Pept. Sci. 20: 885-895.
[https://doi.org/10.1002/psc.2681]

-
Dalanezi, F. M., S. F. Joaquim, F. F. Guimaraes, S. T. Guerra, B. C. Lopes, E. M. S. Schmidt and H. Langoni. 2020. Influence of pathogens causing clinical mastitis on reproductive variables of dairy cows. J. Dairy Sci. 103(4): 3648-3655.
[https://doi.org/10.3168/jds.2019-16841]

-
Duse, A., K. Persson-Waller and K. Pedersen. 2021. Microbial aetiology, antibiotic susceptibility and pathogen-specific risk factors for udder pathogens from clinical mastitis in dairy cows. Animals 11(7): 2113.
[https://doi.org/10.3390/ani11072113]

-
Gilbert, F. B., P. Cunha, K. Jensen, E. J. Glass, G. Foucras, C. Robert-Granié and P. Rainard. 2013. Differential response of bovine mammary epithelial cells to Staphylococcus aureus or Escherichia coli agonists of the innate immune system. Vet. Res. 44: 40.
[https://doi.org/10.1186/1297-9716-44-40]

-
Gomes, F. and M. Henriques. 2016. Control of bovine mastitis: old and recent therapeutic approaches. Curr. Microbiol. 72: 377-382.
[https://doi.org/10.1007/s00284-015-0958-8]

-
Gomes, F., M. J. Saavedra and M. Henriques. 2016. Bovine mastitis disease/pathogenicity: evidence of the potential role of microbial biofilms. Pathog. Dis. 74(3): ftw006.
[https://doi.org/10.1093/femspd/ftw006]

-
Günther, J., W. Petzl, I. Bauer, S. Ponsuksili, H. Zerbe, H. J. Schuberth and H. M. Seyfert. 2017. Differentiating Staphylococcus aureus from Escherichia coli mastitis: S. aureus triggers unbalanced immune-dampening and host cell invasion immediately after udder infection. Sci. Rep. 7: 4811.
[https://doi.org/10.1038/s41598-017-05107-4]

- Han, S. M., K. G. Lee, J. H. Yeo, H. Y. Kweon, B. S. Kim, J. M. Kim and S. T. Kim. 2007. Antibacterial activity of the honey bee venom against bacterial mastitis pathogens infecting dairy cows. Int. J. Ind. Entomol. 14(2): 137-142.
- Jung, S. K., S. I. Oh, H. G. Lee, Y. H. Jung, T. Y. Hur, S. M. Han and A. Cho. 2021. Antibacterial effect of bee venom against Gram-positive and negative bacteria isolated from mastitis in dairy cattle. Korean J. Vet. Serv. 44(3): 169-174.
-
Ma, T., L. Shen, Q. Wen, R. Lv, Q. Hou, L. Y. Kwok and H. Zhang. 2020. Microbiota in milk from healthy and mastitis cows varies greatly in diversity, species richness and composition, as revealed by PacBio sequencing. Microbiology 167(7).
[https://doi.org/10.1101/2020.08.13.249524]

-
Mbindyo, C. M., G. C. Gitao, P. J. Plummer, B. W. Kulohoma, C. M. Mulei and R. Bett. 2021. Antimicrobial resistance profiles and genes of staphylococci isolated from mastitic cow’s milk in Kenya. Antibiotics 10: 772.
[https://doi.org/10.3390/antibiotics10070772]

-
Oliver, S. P. and S. E. Murinda. 2012. Antimicrobial resistance of mastitis pathogens. Vet. Clin. North Am. Food Anim. Pract. 28: 165-185.
[https://doi.org/10.1016/j.cvfa.2012.03.005]

-
Park, S. W., B. R. Park, S. M. Yun, H. G. Kang, B. J. So, S. J. Yun and G. J. Lee. 2013. Antimicrobial activities of honey bee venom against pathogens isolated from clinical bovine mastitis in Korea. Planta Med. 79.
[https://doi.org/10.1055/s-0033-1352324]

-
Park, Y. K., L. K. Fox, D. D. Hancock, W. McMahan and Y. H. Park. 2012. Prevalence and antibiotic resistance of mastitis pathogens isolated from dairy herds transitioning to organic management. J. Vet. Sci. 13: 103.
[https://doi.org/10.4142/jvs.2012.13.1.103]

-
Rana, E. A., M. A. Fazal and M. A. Alim. 2022. Frequently used therapeutic antimicrobials and their resistance patterns on Staphylococcus aureus and Escherichia coli in mastitis affected lactating cows. Int. J. Vet. Sci. Med. 10: 1-10.
[https://doi.org/10.1080/23144599.2022.2038494]

-
Sokolov, S., K. Fursova, I. Shulcheva, D. Nikanova, O. Artyemieva, E. Kolodina and F. Brovko. 2021. Comparative analysis of milk microbiomes and their association with bovine mastitis in two farms in central Russia. Animals 11: 1401.
[https://doi.org/10.3390/ani11051401]

-
Srichok, J., N. Yingbun, T. Kowawisetsut, S. Kornmatitsuk, U. Suttisansanee, P. Temviriyanukul and B. Chantong. 2022. Synergistic antibacterial and anti-inflammatory activities of Ocimum tenuiflorum ethanolic extract against major bacterial mastitis pathogens. Antibiotics 11(4): 510.
[https://doi.org/10.3390/antibiotics11040510]

-
Wehbe, R., J. Frangieh, M. Rima, D. El Obeid, J. M. Sabatier and Z. Fajloun. 2019. Bee venom: Overview of main compounds and bioactivities for therapeutic interests. Molecules 24(16): 2997.
[https://doi.org/10.3390/molecules24162997]

-
Yu, Z. N., J. Wang, H. Ho, Y. T. Wang, S. N. Huang and R. W. Han. 2020. Prevalence and antimicrobial-resistance phenotypes and genotypes of Escherichia coli isolated from raw milk samples from mastitis cases in four regions of China. J. Glob. Antimicrob. Resist. 22: 94-101.
[https://doi.org/10.1016/j.jgar.2019.12.016]

-
Zhang, D., Z. Zhang, C. Huang, X. Gao, Z. Wang, Y. Liu and M. Liu. 2018. The phylogenetic group, antimicrobial susceptibility, and virulence genes of Escherichia coli from clinical bovine mastitis. J. Dairy Sci. 101(1): 572-580.
[https://doi.org/10.3168/jds.2017-13159]

