Botanical Origin and Diversity of Pollen in Honey Samples Collected from Uzbekistan Determined by the Melissopalynological Analysis
Abstract
Beekeeping and honey production is important agricultural component in Uzbekistan. However, scientific verification of honey origin remains limited. Given increasing concerns about honey authenticity and labeling accuracy, this study aimed to evaluate the botanical origin of Uzbek honey using melissopalynological analysis. A total of 26 honey samples collected from supermarkets, markets, apiaries, and universities in the Tashkent region, examined to determine the correspondence between pollen composition and the floral source stated on product labels. Of the total, 20 samples were classified as monofloral honey based on the dominant pollen (>45%), while the remaining six were considered multifloral. Notably, 12 samples labeled as multifloral were reclassified as monofloral based on pollen dominance, revealing inconsistencies between actual content and labeling. Only 5 samples (1 Tilia, 3 Alhagi, 1 Helianthus) showed agreement between dominant pollen and the label claim. A total of 17 pollen taxa were identified, and hierarchical clustering analysis revealed distinct groupings based on dominant pollen types. These findings underscore the need for standardized labeling practices and demonstrate the value of melissopalynology as a reliable tool for verifying the botanical origin of honey and supporting the development of quality standards for Uzbekistan honey.
Keywords:
Apis mellifera, Palynology, Uzbekistan, Melliferous flower, HoneyINTRODUCTION
Uzbekistan, located in Central Asia, offers a unique environment for honey research due to its diverse topography and climate. The majority of the country’s territory consists of desert landscapes (85%), while mountainous areas and foothills occupy only about 15% of the land. This distinct ecological setting contributes to the diversity of both cultivated and wild melliferous plants in Uzbekistan, which in turn reflects the environmental specificity of local apiculture (Mamadalieva et al., 2017; Atamuratova et al., 2021). Despite the ecological and apicultural importance of these plants, studies on their taxonomy and the characteristics of the honey produced from them remain limited (Atamuratova et al., 2021).
Honey is defined as a natural sweet substance produced by honey bees from the nectar of plants or from honeydew, which they collect and store within the hive (EC, 2001). After foraging, bees bring nectar back to the colony, where it is processed into honey primarily through the activity of the enzyme invertase. Honey is generally classified into monofloral and multifloral types: monofloral honey is derived predominantly from the nectar of a single plant species, while multifloral honey is produced from the nectar of multiple floral sources (Mureşan et al., 2022). The botanical origin of honey significantly affects its biochemical composition, including amino acid profiles, carbon isotope ratios, and organoleptic properties such as taste and aroma (Khan et al., 2024).
Honey naturally contains pollen, and the quantity and composition of pollen present can vary depending on the plant species and the foraging behavior of honey bees (Louveaux et al., 1978). The scientific analysis of pollen grains found in honey to determine its botanical origin is known as mellisopalynology. This method enables the identification of the geographical and floral origin of honey and helps elucidate the floral preferences of honey bees (Dórea et al., 2010). Based on the relative abundance of pollen types in a honey sample, dominance categories are established: a pollen taxon comprising more than 45% of the total pollen count is classified as predominant, 16-45% as secondary, and 3-15% as minor (Forcone, 2008). When a single pollen type exceeds 45% of the total, the honey is generally considered monofloral. This classification provides valuable insight into the surrounding vegetation and plays a critical role in identifying melliferous plants favored by honey bees (Von Der Ohe et al., 2004).
Uzbekistan is known for its high levels of honey production and consumption. However, scientific investigations into the botanical origins of Uzbek honey remain scarce. Several previous studies have already proposed standards regarding pollen content in honey (El-Sofany et al., 2020). Moreover, in some European countries, regulations require the indication of pollen composition on honey labels (Thrasyvoulou et al., 2018; Greek Decision 127, 2004). Given the importance of accurate labeling and botanical traceability in ensuring honey quality, there is a pressing need to evaluate the floral composition of locally produced honey.
To address this gap, the present study employs melissopalynological techniques to analyze the pollen content of honey samples produced in Uzbekistan. The primary objective is to assess the consistency between the identified botanical origins and the floral information provided on consumer labels. By doing so, this research aims to establish a foundational reference for future studies and standardization efforts related to Uzbek honey.
MATERIALS AND METHODS
1. Honey samples
A total of 26 honey samples were used in this study. The samples were collected from four different sources in Tashkent: supermarkets (n=5), local markets (n=7), apiaries (n=9), and Tashkent State Agrarian University (n=5). All honey was produced in Tashkent in 2022. Detailed information on each sample is provided in Table 1.
2. Pollen extract
The experimental procedure was conducted with slight modifications to the method described by Von Der Ohe et al. (2004), as outlined below. Crystallized, spreadable honey samples were first liquefied by placing them in a water bath at 60°C until fully dissolved and homogenized. A 15 g aliquot of honey was then mixed with 30 mL of distilled water at room temperature, and the mixture was vortexed to ensure uniform dispersion. The resulting solution was centrifuged at 1080×g for 10 minutes, after which the supernatant was discarded. This washing step was repeated at least twice to ensure complete removal of sugar crystals from the mixture.
To enhance the visibility of pollen grains under the microscope, 10 mL of a 10% Safranin-O staining solution was added to the residue and vortexed thoroughly. The stained mixture was then centrifuged at 1080×g for 10 minutes, and the supernatant was carefully removed.
Subsequently, 10 mL of absolute ethanol (100%) was added to the pellet to remove excess stain and moisture, followed by a third centrifugation under the same conditions. After discarding the ethanol supernatant, 1 mL of the resulting concentrate was transferred to a 2 mL microcentrifuge tube using a micropipette.
The concentrate was mixed with glycerol and left to stand at room temperature for 24 hours to allow ethanol evaporation. This process yielded the final pollen preparation for microscopic analysis.
3. Identification
Pollen grains were counted in three randomly selected fields within a single cover glass area (18 mm×18 mm), and the average was calculated. For each replicate, a minimum of 300 pollen grains were identified. Pollen identification was carried out by referencing previously published studies and internationally recognized palynological databases, including PalDat (https://www.paldat.org/) and The Global Pollen Project (https://globalpollenproject.org/).
Morphological characterization of pollen was conducted using a light microscope (BX53, OLYMPUS, Tokyo, Japan), and images were captured with the IMT i-Solution Lite software (IMT i-Solution Inc., Burnaby, British Columbia, Canada).
4. Data analysis
All analyses were conducted using R Studio. To explore the similarity among honey samples based on their pollen composition, hierarchical cluster analysis was performed using the identified pollen taxa. Additionally, to assess the diversity, dominance, richness, and evenness of pollen taxa in Uzbek honey samples, the Kruskal-Wallis test was applied. Subsequently, Dunn’s test was conducted as a post hoc analysis to identify significant pairwise differences between groups.
RESULTS
1. Melissopalynological analysis
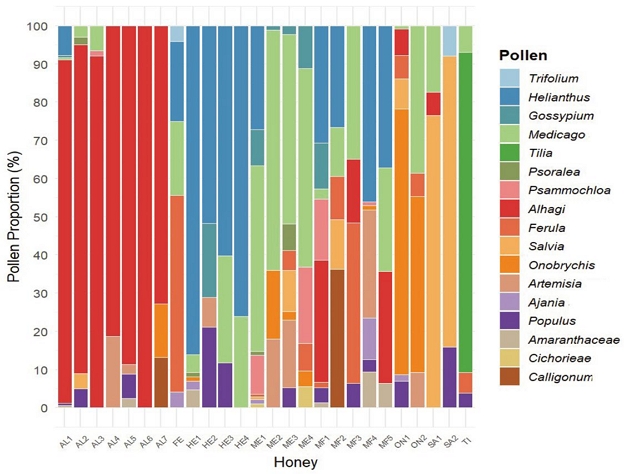
17 types of pollen were identified at the genus level in the 26 honey samples analyzed. The types and proportions of pollen varied among the samples. The lowest proportion was observed for Amaranthaceae (0.56%), while the highest was for Tilia (95.61%), indicating considerable variation in pollen content among the honey samples. Based on the melissopalynological analysis, 20 honey samples were classified as monofloral, each containing more than 45% of a single dominant pollen type. The remaining 6 samples were classified as multifloral, with the dominant pollen type ranging from 16% to 45%, which was still sufficient for them to be categorized separately. Finally, 7 types of mono floral honey were identified: Alhagi (n=7), Helianthus (n=4), Medicago (n=4), Salvia (n=2), Ferula (n=1), Onobrychis (n=1), Tilia (n=1), as well as five types of multi flower honey with dominant species: Multifloral (Helianthus) (n=2), Multifloral (Alhagi) (n=1), Multifloral (Calligonum) (n=1), Multifloral (Ferula) (n=1), and Multifloral (Onobrychis) (n=1). The content of dominant and subordinate pollen is detailed in Table 1.
The diverse plant origins of honey produced in Uzbekistan are illustrated in Fig. 1. The pollen content of the 11 honey samples classified by nectar source was analyzed. Medicago was commonly found in all samples, followed by Populus, Helianthus, and Ferula, which were present in 9, 8, and 8 samples, respectively. Additionally, Tilia and Cichorieae were found in only one sample each.
2. Ecological index analysis of pollen in honey produced in Uzbekistan
We analyzed four ecological indicesdiversity, dominance, evenness, and abundance-based on the botanical origins of pollen identified in 26 honey samples produced in Uzbekistan. The indices were grouped according to nectar source, and samples without biological replicates were represented as individual dots in the visualizations (Fig. 2). Kruskal-Wallis test results indicated significant differences in dominance (p=0.02007) and diversity (p=0.02046), whereas abundance and evenness did not show statistically significant differences (p=0.268 and p=0.1542, respectively). Post-hoc analysis using Dunn’s test revealed that, in terms of dominance, Alhagi honey significantly differed from Medicago, Multifloral (Alhagi), Multifloral (Calligonum), and MultifloralHelianthus honeys. Similarly, for diversity, Alhagi honey was significantly different from the same four groups.
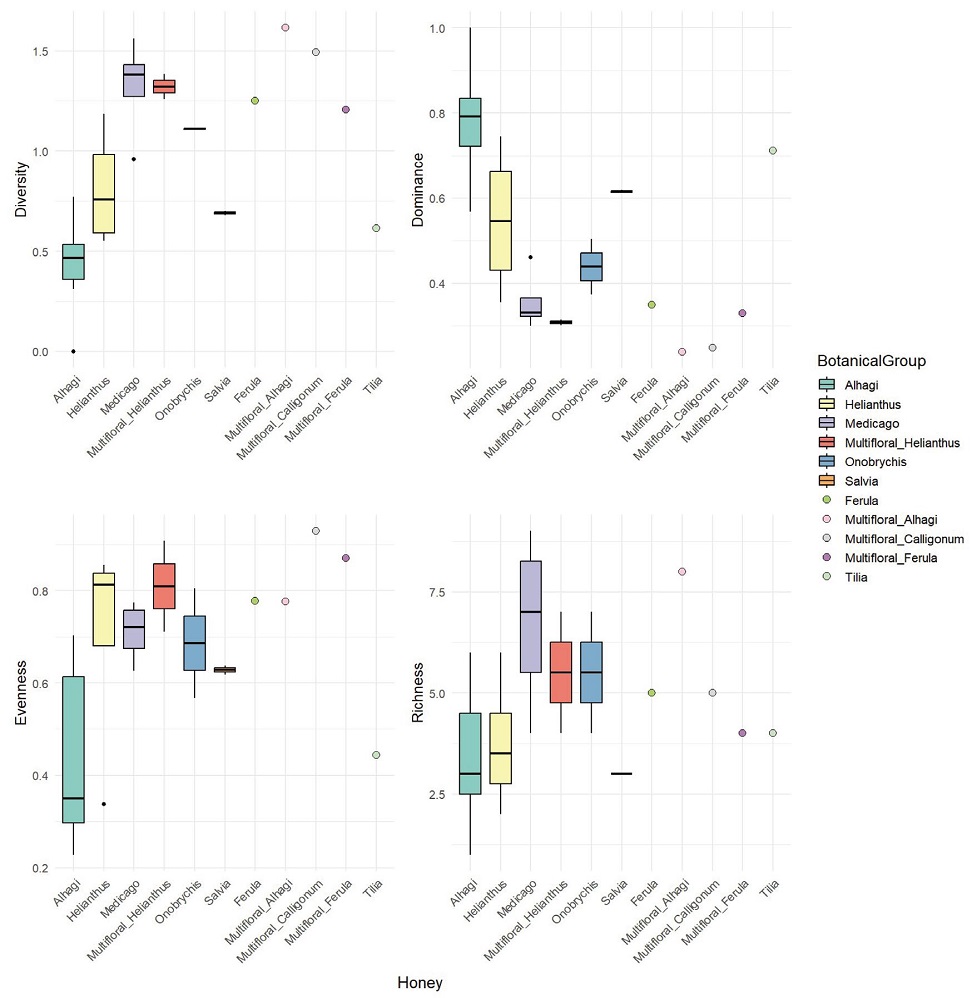
Comparison of ecological indices (diversity, dominance, evenness, and richness) among Uzbekistan honey samples grouped by the dominant pollen type determined by melissopalynological analysis. The x-axis shows honey sample with dominant pollen type. Dots indicate samples without biological replicates (n=1).
DISCUSSION
Melissopalynology is a valuable method for determining the authenticity, potential adulteration, and botanical distinctiveness of honey (Shakoori et al., 2023). In this study, we analyzed honey samples produced in Uzbekistan using melissopalynological analysis to assess their pollen composition and the consistency between the identified botanical origins and the labeled claims. Prior to the evaluation, we confirmed in Fig. 3 that all identified plant taxa are native to Central Asia, based on previous studies (Lu et al., 2018a, 2018b). According to Abdiniyazova et al. (2016), Artemisia, Ferula, and Alhagi are plants of commercial importance in Uzbekistan apiculture, and among these, Ferula and Alhagi were identified as sources of monofloral honeys in this study.
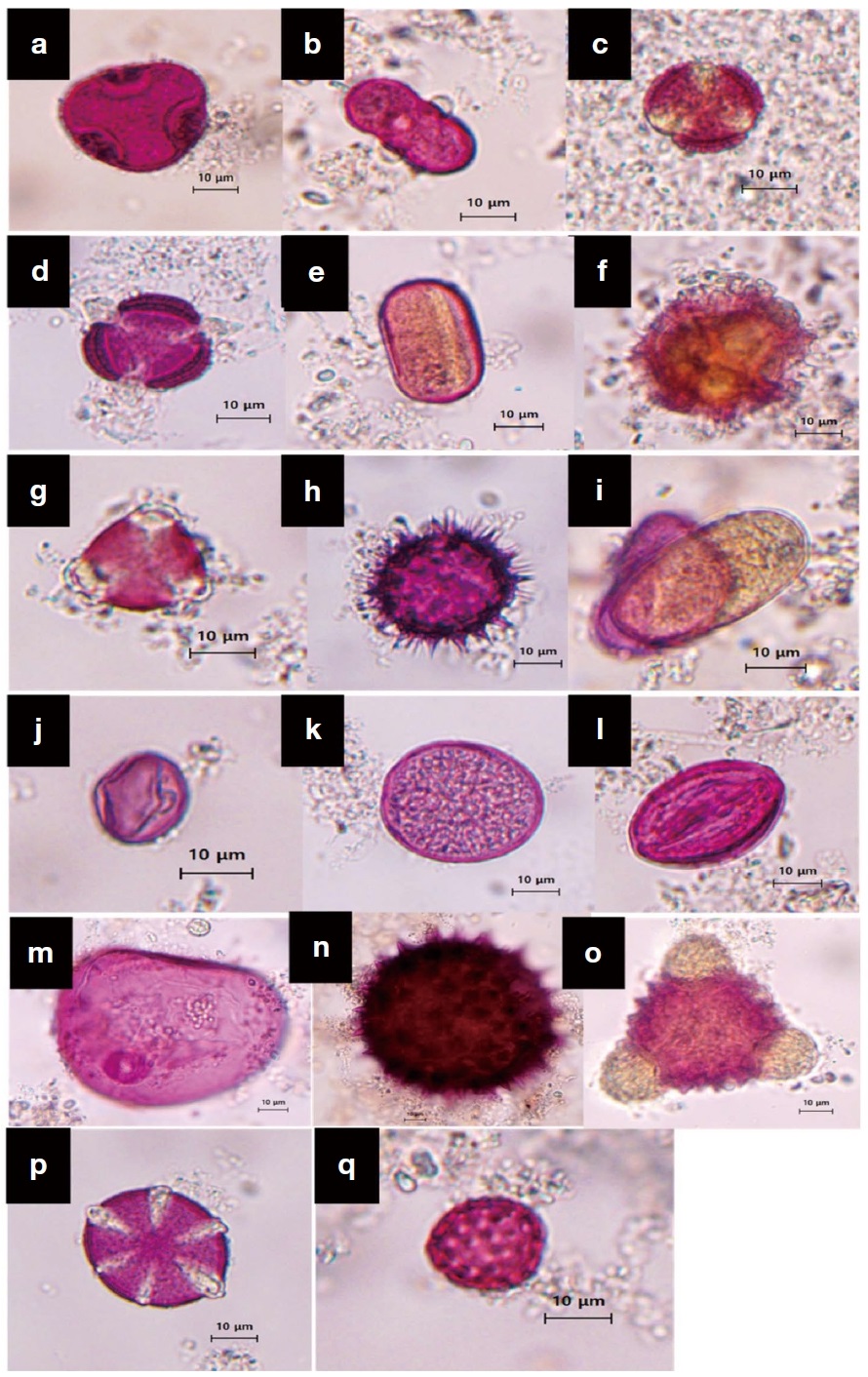
Pollen morphology of major nectar plants photographed under the light microscope (400×). (a) Tilia, (b) Ferula, (c) Ajania, (d) Artemisia, (e) Trifolium, (f) Cichorieae, (g) Medicago, (h) Helianthus, (i) Onobrychis, (j) Alhagi, (k) Populus, (l) Calligonum, (m) Psammochloa, (n) Gossypium, (o) Psoralea, (p) Salvia, (q) Amaranthaceae.
Fig. 1 visually contrasts typical pollen compositions of monofloral and multifloral honeys. Monofloral honeys are typically dominated by a single pollen type, whereas multifloral honeys exhibit more diverse floral origins. As shown in Table 1, 20 samples were classified as monofloral honeys based on the criterion of dominant pollen exceeding 45%, while the remaining 6 samples were classified as multifloral. For the multifloral honeys, although the dominant pollen types did not exceed 45%, they still showed relatively high dominance values ranging from 26.23% to 38.81% and were accordingly highlighted.
We also examined the consistency between the labeled botanical origins and the actual pollen profiles. Among all the honey samples, only 5 had labels that matched the melissopalynological findings, while the remaining 11 were inconsistent. This discrepancy suggests that Uzbekistan’s apiculture industry currently lacks a fully standardized system to ensure honey quality and accurate labeling.
One notable exception was Medicago honey. Although the proportion of Medicago pollen did not exceed the conventional 45% threshold for monofloral classification, it was still regarded as monofloral due to the clear dominance of Medicago pollen. This aligns with earlier studies that recognized Medicago as a low pollen-producing genus. As such, prior research proposed lower thresholds for certain species: ≥20% for Medicago (Forcone, 2008; Ciappini et al., 2016), ≥15% for Salvia (Kenjerić et al., 2006), and ≥20% for Tilia (PN-88/A-77626, 1988). These species-specific thresholds reflect the need to consider differential pollen productivity when conducting melissopalynological evaluations.
This species-specific variation is also evident in Fig. 2 compared to other monofloral honeys, Medicago honey displayed a lower dominance index and a higher diversity index, in stark contrast to Alhagi honey, which showed high dominance and low diversity. Another remarkable observation was the Tilia honey, which, despite prior assumptions that a 20% pollen threshold suffices for its monofloral status, showed an unusually high pollen proportion of 95.61%. However, it is important to note that many of these thresholds are based on outdated data, and floral resource availability can fluctuate significantly due to alternating flowering patterns (Szklanowska and Teper, 1999; Dmitruk, 2019; Sultanova et al., 2022), climate, and other environmental factors (Timberlake et al., 2019; Baldock, 2020).
Such variability may account for the deviations in pollen proportions observed in the current study. Relying solely on pollen morphology has inherent limitations, especially when identifying species within morphologically similar taxa (Brodschneider et al., 2019). This is particularly true for a migration beekeeping context like Uzbekistan, where accurate botanical identification requires knowledge of flowering phenology and geographic distribution (Williams and Kremen, 2007). Therefore, to more precisely determine the botanical origin of honey, integrating genetic analysis with morphological identification is essential (Bruni et al., 2012).
Melissopalynology offers valuable insights into the distinctiveness and authenticity of honey, and it can play a critical role in enhancing the credibility of honey (Escriche et al., 2023; Hussein et al., 2024). To comprehensively evaluate honey quality, however, it is essential to integrate melissopalynological findings with complementary physicochemical analyses. This combined approach provides a robust foundation for identifying functional honeys in Uzbekistan, comparable to antioxidant-rich chestnut honey in Korea (Kim et al., 2023) and pharmacologically active manuka honey in New Zealand (Patel and Cichello, 2013).
Overall, the results confirm that melissopalynology is a powerful tool for determining the botanical origin of honey and mapping regionally dominant nectar sources. This method provides a scientific basis for establishing honey certification standards and developing regional branding strategies for the Uzbekistan honey industry.
CONCLUSION
This study examined the botanical origin of honey produced in Uzbekistan by applying melissopalynological analysis and evaluating the consistency between labeled floral claims and actual pollen composition. Among the 26 samples analyzed, 20 met the criteria for monofloral honey based on the dominant pollen content (≥45%). However, 12 of these were mislabeled as multifloral, and only three samples exhibited complete consistency between the label and the dominant pollen type. These discrepancies highlight the lack of standardized labeling practices and underscore the urgent need for a reliable certification system to enhance product transparency and consumer confidence.
A total of 17 pollen taxa were identified, indicating that Uzbekistan honeybees forage in a diverse and florally rich environment. Multifloral honeys demonstrated higher species richness and diversity compared to monofloral honeys, reflecting broader foraging behavior and more complex floral resources. However, exceptions such as Medicago and Tilia honeys illustrate that pollen content can vary significantly due to intrinsic factors like low pollen production and external factors such as climate. Therefore, field-based surveys of nectar plant distribution and studies on environment-dependent pollen productivity are necessary to contextualize and complement melissopalynological data.
These findings suggest that pollen profiles in honey can serve not only as indicators of floral origin but also as proxies for local biodiversity and landscape structure. While melissopalynology provides critical insight into the botanical authenticity of honey, it is insufficient on its own for comprehensive quality evaluation. Hence, a holistic approach integrating melissopalynological and physicochemical analyses is required (Jang et al., 2025, in submission).
The present results reaffirm the utility of melissopalynology as both a diagnostic and ecological tool. It offers a scientific basis for developing standardized labeling protocols, supporting floral origin authentication, and informing region-specific branding strategies. These measures are essential for improving quality control, market competitiveness, and sustainable growth within Uzbekistan’s beekeeping industry.
Acknowledgments
This study was supported by the National Research Foundation of Korea (NRF) on Smart AI UZ project (NRF-2022K1A3A9A05036394).
References
-
Abdiniyazova, G. J., O. K. Khojimatov and V. V. Pak. 2016. Honey in traditional cuisine of Uzbekistan and analysis of melliferous flora of Karakalpakstan. J. Ethnic Foods 3(3): 222-227.
[https://doi.org/10.1016/j.jef.2016.07.002]

-
Atamuratova, N. T., R. Mukhamatzanova and C. K. Briev. 2021. Honey significance of forest lands in south Uzbekistan. IOP Conference Series: Earth and Environmental Science 775: 012013.
[https://doi.org/10.1088/1755-1315/775/1/012013]

-
Baldock, K. C. 2020. Opportunities and threats for pollinator conservation in global towns and cities. Curr. Opin. Insect Sci. 38: 63-71.
[https://doi.org/10.1016/j.cois.2020.01.006]

-
Bruni, I., F. De Mattia, S. Martellos, A. Galimberti, P. Savadori, M. Casiraghi, P. L. Nimis and M. Labra. 2012. DNA barcoding as an effective tool in improving a digital plant identification system: A case study for the area of Mt. Valerio, Trieste (NE Italy). PLoS One 7(9): e43256.
[https://doi.org/10.1371/journal.pone.0043256]

-
Brodschneider, R., K. Gratzer, E. Kalcher-Sommersguter, H. Heigl, W. Auer, R. Moosbeckhofer and K. Crailsheim. 2019. A citizen science supported study on seasonal diversity and monoflorality of pollen collected by honey bees in Austria. Sci. Rep. 9: 16633.
[https://doi.org/10.1038/s41598-019-53016-5]

-
Ciappini, M., M. Vitelleschi and A. Calviño. 2016. Chemometrics classification of Argentine clover and eucalyptus honeys according to palynological, physicochemical, and sensory properties. Int. J. Food Properties 19(1): 111-123.
[https://doi.org/10.1080/10942912.2015.1020436]

-
Dmitruk, M. 2019. Flowering, nectar secretion, and structure of the nectary in the flowers of Acer pseudoplatanus L. Acta Agrobotanica 72(3).
[https://doi.org/10.5586/aa.1787]

-
Dórea, M. D. C., J. S. D. Novais and F. D. A. R. D. Santos. 2010. Botanical profile of bee pollen from the southern coastal region of Bahia, Brazil. Acta Botanica Brasilica, 24(3): 862-867.
[https://doi.org/10.1590/S0102-33062010000300028]

- EC Council Directive. 2001. EC Council Directive 2001/110/EC of 20 December 2001 relating to honey. Official Journal of the European Communities, L10, 47-52.
-
El-Sofany, A., Y. A. Naggar, E. Naiem, J. P. Giesy and A. Seif. 2020. Authentication of the botanical and geographic origin of Egyptian honey using pollen analysis methods. J. Apic. Res. 59(5): 946-955.
[https://doi.org/10.1080/00218839.2020.1764333]

-
Escriche, I., M. Juan-Borrás, M. Visquert and J. M. Valiente. 2023. An overview of the challenges when analysing pollen for monofloral honey classification. Food Control 143: 109305.
[https://doi.org/10.1016/j.foodcont.2022.109305]

-
Forcone, A. 2008. Pollen analysis of honey from Chubut (Argentinean Patagonia). Grana 47(2): 147-158.
[https://doi.org/10.1080/10652460802106340]

- Greek Decision 127/2004. 2005. Identification of Greek monofloral pine, fir, chestnut, erica, thyme, citrus, cotton and helianthus honeys. Codex Alimentarius, FEK 239/B/23.2. Athens: Greek Ministry of Agricultural and Food Development.
-
Hussein, O. and A. Seid. 2024. Botanical origins of honeys from pollen analysis during the main honey flow across agro-ecologies in Kelala District, South Wollo, Ethiopia. Heliyon 10(21): e40101.
[https://doi.org/10.1016/j.heliyon.2024.e40101]

- Kenjerić, D., L. J. Primorac, M. L. Mandić, D. Bubalo, A. Perl Pirički and I. Flanjak. 2006. Dalmatian sage (Salvia officinalis L.) honey characterization. Deuts. Lebens.-Rund. 102(10): 479-484.
-
Khan, K., K. Malik, M. Ahmad, M. N. I. Raja, M. Nazish, A. Kamal and M. T. Naseem. 2024. Exploring the nutritional composition and quality parameters of natural honey from diverse melliferous flora. Sci. Rep. 14(1): 31626.
[https://doi.org/10.1038/s41598-024-79672-w]

-
Kim, J., N. P. Uy, D. Kim and S. Lee. 2023. Analysis of phenolic acid content and antioxidant activity of chestnut honey from different regions of Korea. Nat. Prod. Sci. 29(3): 127-131.
[https://doi.org/10.20307/nps.2023.29.3.127]

-
Louveaux, J., A. Maurizio and G. Vorwohl. 1978. Methods of melissopalynology. Bee World 59(4): 139-157.
[https://doi.org/10.1080/0005772x.1978.11097714]

-
Lu, K. Q., G. Xie, M. Li, J. F. Li, A. Trivedi, D. K. Ferguson and Y. F. Wang. 2018a. Dataset of pollen morphological traits of 56 dominant species among desert vegetation in the eastern arid central Asia. Data Brief 18: 1022-1046.
[https://doi.org/10.1016/j.dib.2018.03.122]

-
Lu, K. Q., G. Xie, M. Li, J. F. Li, A. Trivedi, D. K. Ferguson and Y. F. Wang. 2018b. Pollen spectrum, a cornerstone for tracing the evolution of the eastern central Asian desert. Quat. Sci. Rev. 186: 111-122.
[https://doi.org/10.1016/j.quascirev.2018.02.009]

-
Mamadalieva, N. Z., D. K. Akramov, E. Ovidi, A. Tiezzi, L. Nahar, S. S. Azimova and S. D. Sarker. 2017. Aromatic medicinal plants of the Lamiaceae family from Uzbekistan: Ethnopharmacology, essential oils composition, and biological activities. Medicines 4(1): 8.
[https://doi.org/10.3390/medicines4010008]

-
Mureşan, C. I., M. Cornea-Cipcigan, R. Suharoschi, S. Erler and R. Mărgăoan. 2022. Honey botanical origin and honey-specific protein pattern: Characterization of some European honeys. LWT 154: 112883.
[https://doi.org/10.1016/j.lwt.2021.112883]

-
Patel, S. and S. Cichello. 2013. Manuka honey: An emerging natural food with medicinal use. Nat. Prod. Biopros. 3: 121-128.
[https://doi.org/10.1007/s13659-013-0018-7]

- Polski Komitet Normalizacyjny (PKN). 1988. PN-88/A-77626: Honey - Requirements and tests. Warsaw: Polish Committee for Standardization.
-
Shakoori, Z., A. Mehrabian, D. Minai, F. Salmanpour and F. Khajoei Nasab. 2023. Assessing the quality of bee honey on the basis of melissopalynology as well as chemical analysis. PLoS One 18(8): e0289702.
[https://doi.org/10.1371/journal.pone.0289702]

-
Sultanova, R., M. Martynova and R. Sazgutdinova. 2022. Honey-bearing potential of Tilia cordata Mill. forests in the Southern Urals. Front. Ecol. Evol. 10.
[https://doi.org/10.3389/fevo.2022.832442]

- Szklanowska, K. and D. Teper. 1999. Wydajnosc pylkowa roznych gatunkow i mieszancow lipy [Tilia L.]. Pszczelnicze Zeszyty Naukowe 43: 291-302.
-
Thrasyvoulou, A., C. Tananaki, G. Goras, E. Karazafiris, M. Dimou, V. Liolios and D. Kanelis. 2018. Legislation of honey criteria and standards. J. Apic. Res. 57(1): 88-96.
[https://doi.org/10.1080/00218839.2017.1411181]

-
Timberlake, T. P., I. P. Vaughan and J. Memmott. 2019. Phenology of farmland floral resources reveals seasonal gaps in nectar availability for bumblebees. J. Applied Ecol. 56: 1585-1596.
[https://doi.org/10.1111/1365-2664.13403]

-
Von Der Ohe, W., L. P. Oddo, M. L. Piana, M. Morlot and P. Martin. 2004. Harmonized methods of melissopalynology. Apidol. 35(Suppl. 1): S18-S25.
[https://doi.org/10.1051/apido:2004050]

-
Williams, N. M. and C. Kremen. 2007. Resource distributions among habitats determine solitary bee offspring production in a mosaic landscape. Ecol. App. 17(3): 910-921.
[https://doi.org/10.1890/06-0269]