
Effects of Sublethal Exposure to the Herbicide Atrazine and Insecticide Profenofos on Queen Performance and Colony Strength in Apiary Settings
Abstract
Honey bees (Apis mellifera) serve as primary pollinators for plants but are frequently exposed to pesticides. While numerous studies have investigated the impact of pesticides on honey bees in laboratory settings, research conducted in apiaries remains insufficient to fully grasp the true effects of these pesticides on colonies and queens. This study delves into the consequences of sublethal doses of atrazine (a herbicide), profenofos (an insecticide), and their combination on queen honey bees. The following concentrations were tested: For atrazine, 6.25 mg/100 g of candy (abbreviated as A0.05), 12.5 mg/100 g of candy (abbreviated as A0.1); for Profenofos, 2.5 μL/100g of candy (abbreviated as P0.02), 3.125 μL/100 g of candy (abbreviated as P0.025); and a combination of atrazine 0.05 and profenofos 0.02 in a 1 : 1 ratio (abbreviated as A+P). The research scrutinized aspects such as egg-laying and various parameters linked to colony development and activities, including measuring sealed brood areas, honey reserves, and stored pollen areas. Although neither pesticide affected queen mating potential, both influenced egg-laying capabilities. By day 24 after introduction, only 80% of queens treated with A0.05, A0.1, or P0.025 were capable of laying eggs. Nonetheless, both pesticides had minimal impact on food storage activities and colony vigor. Colonies from the P0.025 group exhibited the lowest brood-rearing activity, while the A0.1 group showed the lowest honey and pollen storage activities. When combined, the two pesticides did not show synergistic effects; instead, intermediate negative effects between profenofos and atrazine were observed. The study indicates short-term effects of these pesticides on colony dynamics.
Keywords:
Pesticides, Pollution, Queens, Honey bees, ApiaryINTRODUCTION
The worldwide decline in pollinator populations is a pressing issue (Durant, 2019; Panziera et al., 2022). Honey bees, Apis mellifera, are globally recognized as the primary crop pollinators, making a substantial annual contribution exceeding $230 billion USD to the global agricultural economy (Calderone, 2012; Fei et al., 2021; Ferenczi et al., 2023). Nevertheless, the exposure of honey bees to pesticides, commonly used by farmers for crop protection, presents a significant challenge (Johnson et al., 2010; Mullin et al., 2010). For instance, herbicide use can lead to adverse effects on honey bees, resulting in disruptions to their behavior (Vázquez et al., 2024). Over the past 15 years, there has been a notable increase in pesticide studies focusing on honey bees of the Apis genus (Abati et al., 2021). Further investigation is imperative to grasp the cumulative risks posed by interactions among multiple pesticides (Graham et al., 2022; Siviter et al., 2023), specifically in relation to their impact on bee behaviors (Capela et al., 2022; Ohlinger et al., 2022).
Pesticide contamination can manifest in bee bodies, flowers, and soil (Ward et al., 2022), resulting in the presence of pesticides in bee products (Calatayud-Vernich et al., 2018; Lozano et al., 2019; Nassar et al., 2024). Moreover, forager bees can serve as vital agents in transferring contaminants from the environment to the colonies (Abou-Shaara, 2014). The vitality and well-being of bee colonies are intricately tied to the quality of their queens (Tarpy et al., 2011; Mattila et al., 2012; Rangel et al., 2013). Nevertheless, numerous factors, such as rearing conditions, environmental variables, and exposure to environmental pollutants, can influence the performance of queens (Rangel and Tarpy, 2016; De Souza et al., 2019; Abou-Shaara et al., 2021). Queens may encounter pesticides during rearing via tainted wax cups and during feeding by worker bees (Forfert et al., 2017; McAfee et al., 2021). Pesticides may contribute to queen failures within colonies, yet our comprehension of the impacts of pesticides on queens and subsequent activities remains limited.
Pesticides can exert detrimental impacts on colony activities and performance at the collective level (Johnson et al., 2010; da Costa Domingues et al., 2022; Christen, 2023). Understanding the effects of pesticides at sublethal concentrations on queens and colonies is paramount, given that queens or colonies frequently encounter such doses. This study focuses on investigating the impacts of pesticides containing atrazine (a chlorinated herbicide with the chemical formula C8H14ClN5, which enters the weed through its roots and then affects photosynthesis in the shoots and leaves) and profenofos (an organophosphate insecticide with the chemical formula C11H15BrClO3PS, which inhibits the acetylcholinesterase enzyme in insects) as active components on both queen and colony levels. These two pesticides are widely used by local farmers and have been detected at residue levels in bee products (Nassar et al., 2024). However, their potential effects on honey bee queens and colonies under field conditions are not yet fully understood. Therefore, the primary objective of this research is to provide valuable insights into the adverse outcomes of these pesticides on honey bee colonies and their queens.
MATERIALS AND METHODS
1. Pesticides
The research was conducted at an apiary located in the Hosh Essa district (latitude: 30°55ʹ13ʺN, longitude: 030°18ʹ33ʺE), El-Beheira Governorate, Egypt. Two pesticides were employed in the study: atrazine (Gesaprim granules (GR) herbicide 80% atrazine) and profenofos (Ictacron 72% Emulsifiable Concentrate (EC), an organophosphate insecticide containing profenofos as the active ingredient) sourced from Alnasar chemical company, Cairo, Egypt. Due to the potential impact of sublethal pesticide concentrations on honey bee queens, initial laboratory experiments were conducted using various concentrations on queens to determine the appropriate sublethal concentrations for application. Based on the results of these initial experiments, the following concentrations were selected for further investigation: 6.25 mg 100 g-1 candy (corresponding to atrazine at 0.05 of the field concentration, abbreviated as A0.05), 12.5 mg 100 g-1 candy (equivalent to atrazine at 0.1 of the field concentration, abbreviated as A0.1), 2.5 μL 100 g-1 candy (indicating profenofos at 0.02 of the field concentration, abbreviated as P0.02), 3.125 μL 100 g-1 candy (representing profenofos at 0.025 of the field concentration, abbreviated as P0.025), and a combination of atrazine 0.05 and profenofos 0.02 in a 1 : 1 ratio (abbreviated as A+P).
2. Exposing queens to pesticides
In Egypt, hybrid Carniolan honey bees are prevalent (Hussein, 2000; Abou-Shaara and Ahmed, 2015). Queens of these hybrid bees were exposed to pesticides through candy (a mixture of powdered sugar and 60% sugar syrup). Sister newly emerged queens, raised from a robust queenless colony using the standard grafting technique (Büchler et al., 2024), were placed in queen cages. Five cages were utilized for each treatment, and an additional five cages with untreated queens were included as a control group in the experiment. Each cage contained contaminated candy and five attendant workers. Each queen was exposed to the treatments for 24 hours at room temperature, with only 0.5 grams of candy provided to each one. Within the 24-hour period, the queens were exposed to the liquids from the candies. Virgin queens can consume various types of sugary food by themselves, regardless of the presence of worker bees (Abou-Shaara and Elbanoby, 2017). The study was conducted from May to July 2022 when limited sources of nectar and pollen were available.
3. Effects on honey bee colonies
Both treated and untreated queens, housed in cages, were introduced into 30 queen-less colonies of equal strength, each comprising four combs densely populated with bees (2 brood combs + 2 food combs). Consequently, each colony was assigned one of the tested queens (5 colonies per group). Subsequently, the queens were permitted to mate in a natural manner, and their egg-laying activities were observed. After the successful acceptance and mating of the introduced queens, the time required for queens to lay eggs was compared among treatments.
Colony strength was evaluated approximately two months after the successful integration of queens into the colonies, which is a sufficient duration to assess colony progress post-queen mating (Abou-Shaara, 2023). This assessment involved measuring sealed brood areas, honey reserves, and stored pollen areas using a wire-framed grid (Jeffree, 1958).
4. Statistical analysis
To examine the impact of atrazine and profenofos on honey bees, the data’s normality was assessed using the Shapiro-Wilk test. Following this, parametric tests were conducted, utilizing ANOVA followed by Tukey’s test for comparing means. The analysis was carried out using SPSS v. 16 (Chicago, USA, 2007), with a significance level set at p≤0.05.
RESULTS
1. Egg laying
All treated and untreated queens were successfully integrated by the queen-less colonies. Following acceptance, the queens underwent natural mating, with some demonstrating the ability to lay eggs within a specific timeframe. At day 24 after introduction, all the untreated queens and those from P0.02 and A+P were capable of laying eggs, but only 80% of queens treated with A0.05, A0.1, or P0.025. This suggests a delay in egg laying in some treatments.
2. Colony strength
Significant differences among the treatments were observed (ANOVA: F=8.32, df=5, p<0.05), as illustrated in Fig. 1. Colonies led by queens treated with A0.05 displayed the highest brood-rearing activity, with the control group following closely behind. The control group exhibited similarities but no significant variance compared to colonies treated with A0.1, P0.02, and A+P. Conversely, colonies from the P0.025 group exhibited the lowest brood-rearing activity.
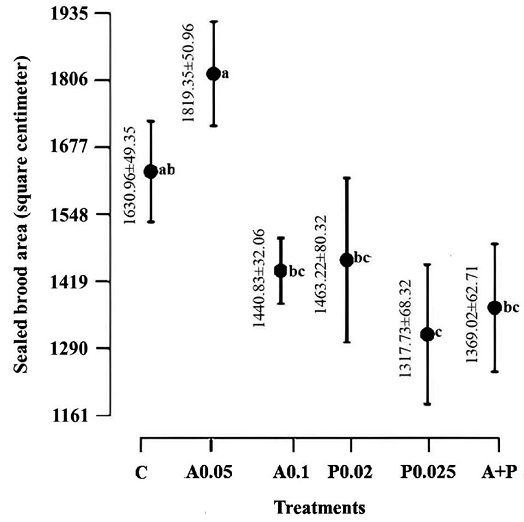
Effects of the tested pesticides on brood-rearing activity. The figure illustrates the confidence intervals for the mean sealed brood areas across the treatments: control (C), atrazine 0.05 (A0.05), atrazine 0.1 (A0.1), profenofos 0.02 (P0.02), profenofos 0.025 (P0.025), and atrazine 0.05+profenofos 0.02 (A+P). Significant variances are denoted by letters following the Tukey Test at a significance level of 0.05 (N=5 colonies per treatment group).
Significant differences were detected among the treatments in their honey storage activity (ANOVA: F=4.87, df=5, p<0.05), as depicted in Fig. 2A. There were no significant variations noted between treatments with elevated means (A0.05, control, P0.02, A+P, and P0.025). The lowest mean was observed for A0.1, with no significant difference from P0.025 exclusively. Furthermore, significant differences were observed among the treatments in their pollen storage activity (ANOVA: F= 7.47, df=5, p<0.05), as shown in Fig. 2B. The A0.05 group exhibited the highest mean area, followed by the A+P group, and finally the control group, with no significant discrepancies among them. The A0.1 group showed the lowest mean, yet no significant variances were noted compared to P0.025 and P0.02.
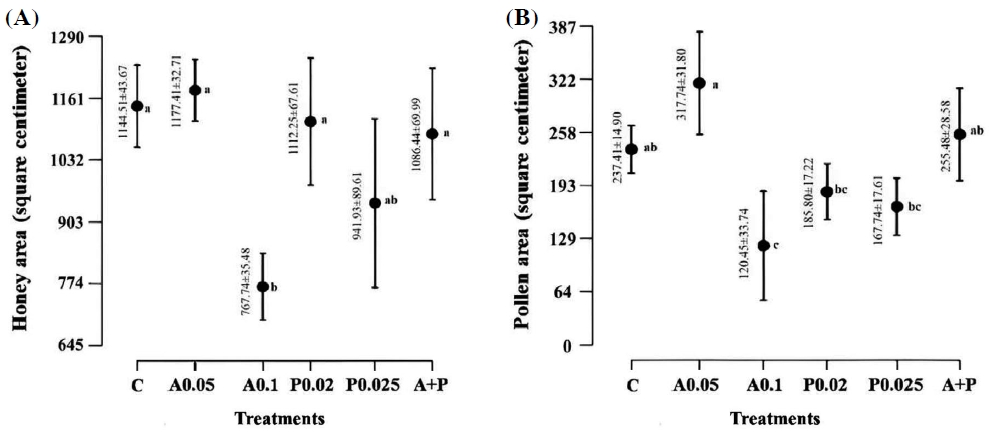
Effects of the tested pesticides on honey and pollen storing activities. The figure displays confidence intervals for the mean areas of honey (A) and pollen (B) across the following treatments: control (C), atrazine 0.05 (A0.05), atrazine 0.1 (A0.1), profenofos 0.02 (P0.02), profenofos 0.025 (P0.025), and atrazine 0.05+profenofos 0.02 (A+P). Significant distinctions are indicated by letters according to the Tukey Test at a significance level of 0.05 (N=5 colonies per treatment group).
DISCUSSION
It could be argued that the sublethal doses of the tested pesticides did not influence the initial acceptance of queens by worker bees but rather affected subsequent egg laying. These subsequent impacts might stem from the pesticides’ effects on the mating quality of queens. Bee queens are recognized for their tendency to mate with a large number of drones (Cobey, 2007; Abou-Shaara et al., 2021), with at least seven mates needed for successful mating (Tarpy et al., 2012, 2013). Queens exposed to the neonicotinoid pesticides thiamethoxam and clothianidin during their developmental stage exhibited fewer matings (Forfert et al., 2017). In support of this, pesticides can affect the pheromone and reproductive behavior of honey bee queens when exposure occurs during development (Walsh et al., 2020). The viability of stored sperm in queens’ spermatheca decreases when exposed to neonicotinoid insecticides (Chaimanee et al., 2016; Kozii et al., 2021). Similarly, thiamethoxam negatively impacts queen quality and the sperm count in the spermathecae (Gajger et al., 2017). These studies support the idea that pesticides can impact the mating and subsequent egg-laying of queens. Particularly, on day 24 after introduction, only 80% of queens treated with A0.05, A0.1, or P0.025 were capable of laying eggs.
The subsequent development of bee colonies can be affected by pesticides. For example, imidacloprid, in particular, has been associated with detrimental impacts on colony progression (Wu-Smart and Spivak, 2016; Fine et al., 2021). However, certain colonies overseen by treated queens exhibited either increased or comparable brood rearing activity compared to the control group, indicating transient effects on colony advancement. Thus, A0.05 exhibited higher brood-rearing activity compared to the control group, while A0.1, P0.02, and A+P demonstrated activity insignificantly similar to the control. In contrast, the P0.025 group showed lower activity than the control.
As indicated in the results section, A0.1 displayed the lowest honey storing activity. Additionally, P0.02, P0.025, and A0.1 exhibited the lowest pollen storing activity. Consequently, food storing activity was notably more adversely affected in atrazine-treated colonies, particularly A0.1, when compared to those treated with profenofos. It is established that certain pesticides can have detrimental effects on the foraging activity of honey bees (Karahan et al., 2015; Gashout et al., 2020), thereby impacting the food storing activity within their colonies. However, some treatments, such as A0.05, showed higher food-storing activity compared to the control colonies. This suggests short-term effects of the pesticides without causing significant disruption to honey bee behaviors. While studies on queens are limited compared to those on workers, exposing adult queens to pesticides-as done in this study-can be considered as complementary to other studies exposing queens during their developmental stages. As an example, during the development of queens, exposure to pesticides like coumaphos, amitraz, and tau-fluvalinate has been found to have detrimental effects on the health and quality of queens in adulthood (Rangel and Tarpy, 2015; Walsh et al., 2020). This study enhances our understanding of the effects of pesticides on bee queens.
CONCLUSION
The current research underscores the impacts of the insecticide profenofos and the herbicide atrazine on honey bee colonies. These effects exhibited variability across different parameters measured. At times, the effects of profenofos were more or less pronounced, or even similar to those of atrazine and the control group. Moreover, not all effects persisted over an extended period, as colony strength was not greatly impacted by the applied pesticides. Furthermore, there were no observable synergistic effects from the pesticide combination. The most prominent adverse effects were associated with egg laying, with higher occurrences observed in atrazine-exposed colonies compared to those exposed to profenofos. This study advances our comprehension of pesticide impacts on queens. In light of these findings, queen breeders are advised to regularly monitor their surroundings to prevent pesticide exposure during queen production and mating phases.
References
-
Abati, R., A. R. Sampaio, A. M. A. Maciel, F. C. Colombo, G. Libardoni, L. Battisti, E. R. Lozano, N. D. Ghisi, F. M. Costa-Maia and M. Potrich. 2021. Bees and pesticides: The research impact and scientometrics relations. Env. Sci. Poll. Res. 28(25): 32282-32298.
[https://doi.org/10.1007/s11356-021-14224-7]

-
Abou-Shaara, H. F. 2023. A comparative study on the quality of honey bee (Apis mellifera) queens developed from larvae after the collection of royal jelly. U. Bee J. 23(1): 14-22.
[https://doi.org/10.31467/uluaricilik.1190100]

-
Abou-Shaara, H. F. 2014. The foraging behaviour of honey bees, Apis mellifera: a review. Vet. Med. 59(1): 1-10.
[https://doi.org/10.17221/7240-vetmed]

-
Abou-Shaara, H. F. and M. E. Ahmed. 2015. Characterisation and tracking changes of morphological characteristics in honey bee, Apis mellifera, colonies. J. Entomol. Acarol. Res. 47(3): 103-108.
[https://doi.org/10.4081/jear.2015.5120]

-
Abou-Shaara, H. F. and M. I. Elbanoby. 2017. Using new cage type for keeping virgin honey bee queens with investigating impacts of feeding type and number of attendant workers. Egy. Acad. J. Biol. Sci. A. Entomol. 10(2): 81-87.
[https://doi.org/10.21608/eajbsa.2017.12677]

-
Abou-Shaara, H. F., N. Adgaba and A. A. Al-Ghamdi. 2021. Current knowledge about behaviors of honey bee queens with highlighting of the importance future studies. J. Basic Appl. Zool. 82: 1-7.
[https://doi.org/10.1186/s41936-021-00234-x]

-
Büchler, R., S. Andonov, R. Bernstein, K. Bienefeld, C. Costa, M. Du, ... and J. Wilde. 2024. Standard methods for rearing and selection of Apis mellifera queens 2.0. J. Apic. Res. 1-57.
[https://doi.org/10.3896/ibra.1.52.1.07]

-
Calatayud-Vernich, P., F. Calatayud, E. Simó and Y. Picó. 2018. Pesticide residues in honey bees, pollen and beeswax: Assessing beehive exposure. Env. Pollu. 241: 106-114.
[https://doi.org/10.1016/j.envpol.2018.05.062]

-
Calderone, N.W. 2012. Insect pollinated crops, insect pollinators and US agriculture: trend analysis of aggregate data for the period 1992-2009. PLoS One 7: e37235.
[https://doi.org/10.1371/journal.pone.0037235]

-
Capela, N., A. Sarmento, S. Simões, H. M. Azevedo-Pereira and J. P. Sousa. 2022. Sub-lethal doses of sulfoxaflor impair honey bee homing ability. Sci. Total Env. 837: 155710.
[https://doi.org/10.1016/j.scitotenv.2022.155710]

-
Chaimanee, V., J. D. Evans, Y. Chen, C. Jackson and J. S. Pettis. 2016. Sperm viability and gene expression in honey bee queens (Apis mellifera) following exposure to the neonicotinoid insecticide imidacloprid and the organophosphate acaricide coumaphos. J. Insect Physiol. 89: 1-8.
[https://doi.org/10.1016/j.jinsphys.2016.03.004]

-
Christen, V. 2023. Different effects of pesticides on transcripts of the endocrine regulation and energy metabolism in honeybee foragers from different colonies. Sci. Rep. 13: 1985.
[https://doi.org/10.1038/s41598-023-29257-w]

-
Cobey, S. W. 2007. Comparison studies of instrumentally inseminated and naturally mated honey bee queens and factors affecting their performance. Apidologie 38: 390-410.
[https://doi.org/10.1051/apido:2007029]

-
da Costa Domingues, C. E., A. M. P. Sarmento, N. X. J. Capela, J. M. Costa, R. M. R. Mina, A. A. da Silva, A. R. Reis, C. Valente, O. Malaspina, H. M. V. S. Azevedo-Pereira and J. P. Sousa. 2022. Monitoring the effects of field exposure of acetamiprid to honey bee colonies in Eucalyptus monoculture plantations. Sci. Total Env. 844: 157030.
[https://doi.org/10.1016/j.scitotenv.2022.157030]

-
De Souza, D. A., M. H. Huang and D. R. Tarpy. 2019. Experimental improvement of honey bee (Apis mellifera) queen quality through nutritional and hormonal supplementation. Apidologie 50: 14-27.
[https://doi.org/10.1007/s13592-018-0614-y]

-
Durant, J. L. 2019. Where have all the flowers gone? Honey bee declines and exclusions from floral resources. J. Rural Stud. 65: 161-171.
[https://doi.org/10.1016/j.jrurstud.2018.10.007]

-
Fei, C. J., K. M. Williamson, R. T. Woodward, B. A. McCarl and J. Rangel. 2021. Honey bees, almonds, and colony mortality: An economic simulation of the US pollination market. Land Econ. 97: 688-703.
[https://doi.org/10.3368/le.97.3.688]

-
Ferenczi, A. F., I. Szűcs and A. B. Gáthy. 2023. Evaluation of the pollination ecosystem service of the honey bee (Apis mellifera) based on a beekeeping model in Hungary. Sustainabil. 15: 9906.
[https://doi.org/10.3390/su15139906]

-
Fine, J. D., K. M. Torres, J. Martin and G. E. Robinson. 2021. Assessing agrochemical risk to mated honey bee queens. J. Visual. Exp. (169): e62316.
[https://doi.org/10.3791/62316-v]

-
Forfert, N., A. Troxler, G. Retschnig, L. Gauthier, L. Straub, R. F. Moritz, P. Neumann and G. G. Williams. 2017. Neonicotinoid pesticides can reduce honeybee colony genetic diversity. PLoS One 12: e0186109.
[https://doi.org/10.1371/journal.pone.0186109]

-
Gajger, I. T., M. Sakač and A. Gregorc. 2017. Impact of thiamethoxam on honey bee queen (Apis mellifera carnica) reproductive morphology and physiology. Bull. Env. Contamin. Toxicol. 99: 297-302.
[https://doi.org/10.1007/s00128-017-2144-0]

-
Gashout, H. A., E. Guzman-Novoa and P. H. Goodwin. 2020. Synthetic and natural acaricides impair hygienic and foraging behaviors of honey bees. Apidologie 51: 1155-1165.
[https://doi.org/10.1007/s13592-020-00793-y]

-
Graham, K. K., M. O. Milbrath, Y. Zhang, N. Baert, S. McArt and R. Isaacs. 2022. Pesticide risk to managed bees during blueberry pollination is primarily driven by off-farm exposures. Sci. Rep. 12: 7189.
[https://doi.org/10.1038/s41598-022-11156-1]

-
Hussein, M. H. 2000. A review of beekeeping in Arab countries. Bee World 81: 56-71.
[https://doi.org/10.1080/0005772x.2000.11099473]

-
Jeffree, E. P. 1958. A shaped wire grid for estimating quantities of brood and pollen in combs. Bee World 39: 115-118.
[https://doi.org/10.1080/0005772x.1958.11095048]

-
Johnson, R. M., M. D. Ellis, C. A. Mullin and M. Frazier. 2010. Pesticides and honey bee toxicity-USA. Apidologie 41: 312-331.
[https://doi.org/10.1051/apido/2010018]

-
Karahan, A., I. Çakmak, J. M. Hranitz, I. Karaca and H. Wells. 2015. Sublethal imidacloprid effects on honey bee flower choices when foraging. Ecotoxicology 24: 2017-2025.
[https://doi.org/10.1007/s10646-015-1537-2]

-
Kozii, I. V., S. Barnsley, M. C. B. D. Silva, S. C. Wood, C. D. Klein, I. M. de Mattos, M. W. Zabrodski, R. C. M. Silva, C. I. O. Fabela, L. Guillemin, I. Dvylyuk, M. C. O. Ferrari and E. Simko. 2021. Reproductive fitness of honey bee queens exposed to thiamethoxam during development. Vet. Pathol. 58: 1107-1118.
[https://doi.org/10.1177/03009858211031845]

-
Lozano, A., M. D. Hernando, S. Uclés, E. Hakme and A. R. Fernández-Alba. 2019. Identification and measurement of veterinary drug residues in beehive products. Food Chem. 274: 61-70.
[https://doi.org/10.1016/j.foodchem.2018.08.055]

-
Mattila, H. R., H. K. Reeve and M. L. Smith. 2012. Promiscuous honey bee queens increase colony productivity by suppressing worker selfishness. Curr. Biol. 22: 2027-2031.
[https://doi.org/10.1016/j.cub.2012.08.021]

-
McAfee, A., J. P. Milone, B. Metz, E. McDermott, L. J. Foster and D. R. Tarpy. 2021. Honey bee queen health is unaffected by contact exposure to pesticides commonly found in beeswax. Sci. Rep. 11: 1-12.
[https://doi.org/10.1038/s41598-021-94554-1]

-
Mullin, C. A., M. Frazier, J. L. Frazier, S. Ashcraft, R. Simonds and J. S. Pettis. 2010. High levels of miticides and agrochemicals in North American apiaries: implications for honey bee health. PLoS One 5: e9754.
[https://doi.org/10.1371/journal.pone.0009754]

-
Nassar, M. K. A., Y. M. Salim, E. Nour-Eldeen, M. S. Younis, M. M. Kelany, M. A. Shebl, A. S. Shafey and H. F. Abou-Shaara. 2024. Seasonal screening of pesticide residues in beehive products collected from different districts in Egypt. Env. Monit. Assess. 196 (3): 297.
[https://doi.org/10.1007/s10661-024-12451-2]

-
Ohlinger, B. D., R. Schürch, S. Durzi, P. M. Kietzman, M. R. Silliman and M. J. Couvillon. 2022. Honey bees (hymenoptera: Apidae) decrease foraging but not recruitment after neonicotinoid exposure. J. Insect Sci. 22 (1): 16.
[https://doi.org/10.1093/jisesa/ieab095]

-
Panziera, D., F. Requier, P. Chantawannakul, C. W. Pirk and T. Blacquière. 2022. The diversity decline in wild and managed honey bee populations urges for an integrated conservation approach. Front. Ecol. Evol. 10: 767950.
[https://doi.org/10.3389/fevo.2022.767950]

-
Rangel, J., J J. Keller and D. R. Tarpy. 2013. The effects of honey bee (Apis mellifera L.) queen reproductive potential on colony growth. Ins. Soci. 60: 65-73.
[https://doi.org/10.1007/s00040-012-0267-1]

-
Rangel, J. and D. R. Tarpy. 2015. The combined effects of miticides on the mating health of honey bee (Apis mellifera L.) queens. J. Apic. Res. 54(3): 275-283.
[https://doi.org/10.1080/00218839.2016.1147218]

-
Rangel, J. and D. R. Tarpy. 2016. In-hive miticides and their effect on queen supersedure and colony growth in the honey bee (Apis mellifera). J. Env. Analyt. Toxicol. 6: 2161-0525.
[https://doi.org/10.4172/2161-0525.1000377]

-
Siviter, H., A. Linguadoca, A. Ippolito and F. Muth. 2023. Pesticide licensing in the EU and protecting pollinators. Curr. Biol. 33: R44-R48.
[https://doi.org/10.1016/j.cub.2022.12.002]

-
Tarpy, D. R., J. J. Keller, J. R. Caren and D. A. Delaney. 2011. Experimentally induced variation in the physical reproductive potential and mating success in honey bee queens. Ins. Soc. 58: 569-574.
[https://doi.org/10.1007/s00040-011-0180-z]

-
Tarpy, D. R., J. J. Keller, J. R. Caren and D. A. Delaney. 2012. Assessing the mating ‘health’ of commercial honey bee queens. J. Econ. Entomol. 105: 20-25.
[https://doi.org/10.1603/ec11276]

-
Tarpy, D. R., D. Vanengelsdorp and J. S. Pettis. 2013. Genetic diversity affects colony survivorship in commercial honey bee colonies. Naturwissenschaften 100: 723-728.
[https://doi.org/10.1007/s00114-013-1065-y]

-
Traynor, K. S. and Z. S. Lamas. 2021. Social disruption: Sublethal pesticides in pollen lead to Apis mellifera queen events and brood loss. Ecotoxicol. Env. Saf. 214: 112105.
[https://doi.org/10.1016/j.ecoenv.2021.112105]

-
Vázquez, D. E., F. Verellen and W. M. Farina. 2024. Early exposure to glyphosate during larval development induces late behavioural effects on adult honey bees. Env. Poll. 360: 124674.
[https://doi.org/10.1016/j.envpol.2024.124674]

-
Walsh, E. M., S. Sweet, A. Knap, N. Ing and J. Rangel. 2020. Queen honey bee (Apis mellifera) pheromone and reproductive behavior are affected by pesticide exposure during development. Behav. Ecol. Sociobiol. 74: 1-14.
[https://doi.org/10.1007/s00265-020-2810-9]

-
Ward, L. T., M. L. Hladik, A. Guzman, S. Winsemius, A. Bautista, C. Kremen and N. J. Mills. 2022. Pesticide exposure of wild bees and honey bees foraging from field border flowers in intensively managed agriculture areas. Sci. Total Env. 831: 154697.
[https://doi.org/10.1016/j.scitotenv.2022.154697]

-
Wu-Smart, J. and M. Spivak. 2016. Sub-lethal effects of dietary neonicotinoid insecticide exposure on honey bee queen fecundity and colony development. Sci. Rep. 6: 1-11.
[https://doi.org/10.1038/srep32108]
