
The Neuromodulatory Landscape of Honey Bee Cognition: Roles of Acetylcholine, Glutamate, GABA, and Biogenic Amines
Abstract
Honey bees (Apis mellifera) have emerged as a powerful model organism for studying the neurobiology of learning and memory. Research in the past decade highlights that multiple neurotransmitter systems contribute to different phases and forms of memory in the honey bee brain. Acetylcholine (ACh) is the primary excitatory transmitter in insect neural circuits, playing key roles in the acquisition and retrieval of learned associations. Glutamatergic signaling, although less prevalent than in vertebrates, is essential for certain memory processes, involving both ionotropic (e.g. NMDA-type) and metabotropic pathways. Inhibitory neurotransmission via γ-aminobutyric acid (GABA) shapes olfactory learning by gating and refining neural activity, enabling cognitive flexibility. Biogenic amines act as critical neuromodulators: dopamine and octopamine provide aversive and appetitive reinforcement signals, respectively, during associative learning, while recent findings also implicate dopamine in appetitive motivation (“wanting”). This review synthesizes current knowledge on these neurotransmitter systems at both molecular and systems levels in the honey bee, illustrating how a complex interplay of signaling molecules underlies learning and memory. We conclude with an overview of how these neuromodulatory pathways integrate to support the sophisticated cognitive capacities of honey bees, and point to open questions for future research.
Keywords:
Honey bees, Learning and memory, Neuromodulation, NeurotransmittersINTRODUCTION
Honey bees possess remarkable learning and memory capabilities, which they use to associate floral cues with food rewards, navigate complex environments, and communicate resource locations to nest mates. Classic conditioning paradigms such as the olfactory proboscis extension reflex (PER) have been instrumental in dissecting the neural substrates of associative learning in bees (Jarriault et al., 2018; Ai and Farina, 2023). These studies reveal that honey bee memory formation is subserved by discrete phases (short-term, mid-term, long-term memory) and involves multiple brain regions, notably the antennal lobes and mushroom bodies (MBs), which are centers for sensory integration and memory storage. Underpinning these processes is a rich tapestry of neurotransmitter systems. The insect brain employs many of the same chemical messengers found in vertebrates-including acetylcholine, amino acid transmitters, and biogenic amines-but often with distinct roles or modes of action due to differences in neural architecture (Schatton et al., 2018).
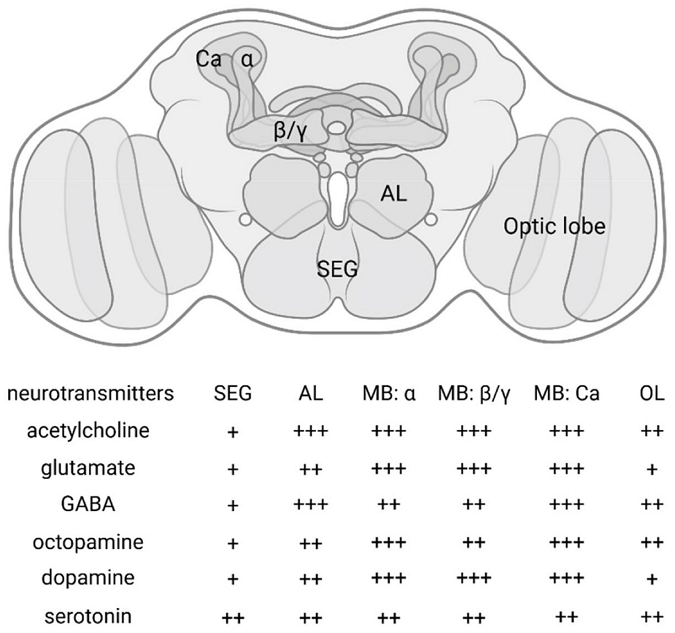
Schematic and relative neurotransmitter distribution across honey bee brain regions. (A) Schematic diagram of the honey bee brain (dorsal view) illustrating the major neuropils: Mushroom Body (MB) with its calyx (Ca), alpha (α) lobe, and beta/gamma (β/γ) lobes; Antennal Lobe (AL); Optic Lobe (OL); and Suboesophageal Ganglion (SEG). (B) Qualitative summary of the relative expression levels of key neurotransmitters in the indicated brain regions. The expression levels are represented as: + (Low/Present), ++ (Medium), and +++ (High/Abundant), based on current literature.
In the honey bee, acetylcholine (ACh) functions as the major fast excitatory neurotransmitter, whereas glutamate assumes both excitatory and inhibitory roles in specific circuits (Dupuis et al., 2011; Grunewald and Siefert, 2019). Inhibitory signaling is largely mediated by γ-aminobutyric acid (GABA) and, in certain sensory neuropils, by histamine (Sachse et al., 2006). Beyond these fast transmitters, several neuromodulators critically influence learning and memory. Dopamine (DA), octopamine (OA), serotonin (5-HT), and tyramine (TA) are biogenic amines that modulate neural plasticity and behavior in accordance with reinforcement, arousal, and motivational states (Scheiner, 2004; Sinakevitch and Strausfeld, 2006; Vergoz et al., 2007). Understanding the honey bee’s neurotransmitter systems thus requires integrating molecular insights-such as receptor subtypes and signaling pathways-with systems-level perspectives on how these chemicals regulate circuit function during learning tasks. Given the critical role of honey bees as pollinators and their alarming global decline, understanding their cognitive abilities is more urgent than ever. Current research faces challenges in bridging laboratory findings with real-world environmental stressors, and fully unraveling the complex neural mechanisms underlying their sophisticated behaviors. Our work aims to address these gaps by elucidating the precise neurotransmitter roles in learning and memory. This knowledge is not merely academic; it’s practically applicable. By identifying how various factors disrupt bee cognition, we can develop targeted interventions, such as nutritional supplements or protective measures against specific pesticides, to enhance colony health and resilience, thereby safeguarding agricultural productivity and biodiversity.
This review summarizes recent findings on how key neurotransmitters in honey bees, specifically ACh, glutamate, GABA, dopamine, octopamine, serotonin, and tyramine, influence learning and memory (Table 1). For each, we’ll cover their receptor types, signaling mechanisms, and their role in memory formation or retrieval within neural circuits and behavior. We’ll present a concise overview of current knowledge from peer-reviewed studies, excluding detailed neuroanatomy or developmental effects unless directly relevant. Our goal is to provide researchers with an accessible synthesis of how these neurotransmitter systems collectively contribute to the honey bee’s complex learning and memory capabilities.
ACETYLCHOLINE: CHOLINERGIC MECHANISMS OF MEMORY
Acetylcholine is a cornerstone of neurotransmission in the honey bee brain. In insects, ACh is the principal excitatory neurotransmitter at central synapses, fulfilling a role analogous to that of glutamate in vertebrates (Grunewald and Siefert, 2019). Cholinergic neurons are pervasive in honey bee brain circuits involved in olfaction and memory; for example, projection neurons from the antennal lobe (the insect analog of the olfactory bulb) release ACh onto mushroom body Kenyon cells, initiating the encoding of odor cues. Pharmacological and genetic studies have demonstrated that disrupting cholinergic signaling impairs learning and memory in bees. Classic work showed that blocking nicotinic ACh receptors (nAChRs) with antagonists like mecamylamine or α-bungarotoxin hampers both the acquisition and retrieval of olfactory memories (Gauthier et al., 2006). Consistent with this, recent molecular analyses confirm that ACh-driven excitatory currents are crucial for activating neural circuits during conditioning (Barbara et al., 2008; Dupuis et al., 2011). This functional specialization was suggested by differential effects of subtype-specific toxins and by subunit-specific expression patterns in the mushroom bodies (Gauthier, 2010; Dupuis et al., 2011).
At the molecular level, honey bees possess genes encoding both nicotinic and muscarinic ACh receptors, though nAChRs have been studied far more extensively. Honey bee nAChRs are pentameric cation channels that, when activated by ACh, depolarize neurons to propagate excitatory signals. Homologs of vertebrate nicotinic subunits (α and β types) are present; for instance, the AChRβ1 subunit is highly expressed in the bee brain and contributes to receptor assemblies in Kenyon cells (Gauthier, 2010; Cartereau et al., 2022). By contrast, muscarinic ACh receptors (mAChRs), which are G-protein-coupled receptors, have been identified in the bee genome but their neurophysiological roles remain less clear. It is suspected that mAChRs modulate aspects of cognition and network excitability (as they do in Drosophila), but direct evidence in honey bees is still lacking.
Behavioral studies underscore ACh’s importance for bee memory. Local infusion of cholinergic antagonists into the mushroom bodies impairs memory formation and recall, indicating that cholinergic synapses in this region are necessary for associative memory storage (Lozano et al., 2001). Conversely, enhancing cholinergic signaling can improve learning performance: low doses of ACh esterase inhibitors (which elevate synaptic ACh levels) have been reported to facilitate olfactory learning in bees. However, excessive cholinergic stimulation can be detrimental, causing neural desensitization or excitotoxic effects (Williamson and Wright, 2013). The impact of environmental cholinergic toxins has also been a subject of recent research. Neonicotinoid insecticides, which are nAChR agonists, provide an unintended experiment in cholinergic hyperactivation. Field-realistic doses of neonicotinoids like imidacloprid and clothianidin have been shown to disrupt honey bee learning and memory performance (Grunewald and Siefert, 2019). For example, chronic exposure to imidacloprid impairs olfactory learning acquisition and memory retention in bees, even at sublethal doses (Wright et al., 2015; Zhang and Nieh, 2015; Grunewald and Siefert, 2019). These findings reinforce that normal ACh signaling is finely tuned for optimal cognitive function, and perturbations can lead to significant deficits.
In summary, acetylcholine acts at the nexus of sensory input and memory encoding in the honey bee brain (Lozano et al., 2001). It drives the synaptic plasticity underlying associative learning, with nicotinic receptors on mushroom body neurons playing decisive roles in memory acquisition and retrieval (Goldberg et al., 1999). The cholinergic system thus provides the fast excitatory drive and initial neural representations of learned stimuli, upon which neuromodulators then act to assign value and salience (Jones et al., 2006). Ongoing research continues to explore cholinergic modulation (e.g. via muscarinic pathways or plastic changes in nAChR expression) in complex forms of learning and in age- or caste-related differences in cognitive ability (Ismail et al., 2006).
GLUTAMATE: EXCITATORY AND INHIBITORY ROLES
Although acetylcholine predominates in fast excitatory signaling in the honey bee brain, glutamate is also a significant neurotransmitter with multifaceted roles in learning and memory (El Hassani et al., 2008). Glutamate in insects can activate both ionotropic receptors (iGluRs) and metabotropic receptors (mGluRs), similar to vertebrates, but insects also possess a unique class of glutamate-gated chloride channels (GluCl) that mediate inhibition (Kucharski et al., 2007; El Hassani et al., 2012). Honey bees have been shown to express homologs of AMPA/kainate-type and NMDA-type iGluRs, as well as multiple metabotropic glutamate receptors (Schatton et al., 2018). These receptors are not as ubiquitously employed for fast synaptic transmission as in mammalian brains, yet they are crucial for specific memory processes in bees.
Pharmacological experiments indicate that blocking glutamatergic signaling can impair long-term memory (LTM) formation in honey bees. Antagonists of NMDA receptors, such as MK-801, were found to interfere with the formation of 24-hour olfactory memory without affecting initial learning or short-term memory (Kucharski et al., 2007). In one study, RNA interference knockdown of the NR1 subunit of the NMDA receptor in the bee brain selectively abolished long-term associative memory while leaving short-term memory intact (Mussig et al., 2010). This aligns with the notion that NMDA receptor-dependent synaptic plasticity (like a Ca2+ influx trigger for downstream cascades) is required for consolidating enduring memories, analogous to mechanisms in vertebrates. Similarly, disruption of a honey bee group II metabotropic glutamate receptor (AmGluRA) by selective agonists or antagonists impairs 24-hour memory when administered before training (Kucharski et al., 2007). This suggests that glutamate’s actions on metabotropic receptors also contribute to memory formation, possibly through modulation of intracellular signaling pathways that stabilize synaptic changes.
Interestingly, glutamate in honey bees can also exert inhibitory effects via GluCl receptors. These chloride channels hyperpolarize neurons when activated by glutamate (El Hassani et al., 2012). Honey bees have genes for GluCl channel subunits, and electrophysiological studies have described glutamate-induced inhibitory currents in bee neurons (Demares et al., 2014). Such dual functionality means glutamate can both excite and inhibit, depending on receptor context. In the antennal lobes (AL), for example, some local interneurons might use glutamate as an inhibitory transmitter, as seen in other insects (Barbara et al., 2005). The presence of GluCl receptors in the bee AL has been implicated in shaping odor processing and possibly in non-associative learning like habituation. A notable finding is that both GluCl channels and GABA_A receptors are involved in honey bee long-term memory formation (Raccuglia and Mueller, 2013). Blocking either type of inhibitory receptor can impair the formation of stable memory, highlighting that a proper balance of excitation and inhibition-in which glutamatergic signaling plays both parts-is essential for memory consolidation (Schatton et al., 2018).
On the excitatory side, evidence is mounting that glutamatergic neurotransmission in honey bees supports certain forms of learning beyond the cholinergic domain. For instance, one line of research implicates an AMPA/kainate-like receptor in appetitive learning: when bees were treated with a glutamate analog or uptake inhibitor, their long-term memory of an odor-sugar association was enhanced, suggesting glutamate availability can be a limiting factor in memory strength (Maleszka et al., 2000; Locatelli et al., 2005). Additionally, a metabotropic glutamate receptor (likely Group I or II) was found to be required in the early stages of memory formation; its pharmacological activation or blockade around the time of training altered the bees’ memory retention 1 day later (Lopatina et al., 2004; Kucharski et al., 2007). These findings point to glutamate acting as a neuromodulator that fine-tunes the induction of memory-related plasticity in tandem with ACh.
In summary, while not the primary excitatory transmitter in honey bees, glutamate plays vital roles in memory through a variety of receptors. It contributes to synaptic plasticity underlying long-term memory via NMDA and mGluR activation, and it participates in inhibitory circuits via GluCl channels that ensure neural network stability during learning. The requirement for glutamatergic signaling in long-lasting memory suggests a conserved biochemical mechanism (e.g. Ca2+-dependent cascades) shared with vertebrates. Ongoing studies are investigating how glutamate interacts with other neurotransmitter systems in the bee brain-for example, whether dopaminergic or octopaminergic neurons modulate glutamate release or receptor sensitivity as part of the reinforcement process. Understanding glutamate’s dual nature in the honey bee nervous system is key to a complete picture of the molecular bases of memory in insects.
GABA: INHIBITION AND MEMORY MODULATION
GABA (γ-aminobutyric acid) is the primary inhibitory neurotransmitter in the honey bee brain, as in other animals (Mustard et al., 2020). GABAergic neurons are widespread in regions involved in sensory processing and learning, including the antennal lobes, mushroom bodies, and protocerebral complexes (Mustard et al., 2020). By activating ionotropic GABA_A receptors (chloride channels) or metabotropic GABA_B receptors on neurons, GABA serves to restrain and shape neural activity (Raccuglia and Mueller, 2013; Boitard et al., 2015). This inhibitory modulation is crucial for processes such as odor discrimination, attention, and memory updating. In the context of olfactory learning, GABAergic circuits help ensure that only the relevant neural signals are strengthened while irrelevant or background activity is suppressed.
One well-studied role of GABA in honey bees is in the antennal lobes-the first relay for odor information. The AL contains many GABAergic local interneurons that provide lateral inhibition between olfactory glomeruli, refining the spatial patterns of odor-evoked activity (Sachse and Galizia, 2002; Krofczik et al., 2008). When bees are presented with an odor, these interneurons can inhibit neighboring glomeruli, enhancing the contrast of the odor representation (Krofczik et al., 2008). This mechanism likely improves the bees’ ability to discriminate similar odor cues and thus can influence learning specificity. Indeed, pharmacological blocking of GABA_A receptors in the AL can lead to broader generalization of learned odors or deficits in odor discrimination learning, consistent with a loss of pattern separation (Krofczik et al., 2008). Histamine, another inhibitory transmitter in the AL, appears to play a complementary role; however, GABA is considered the dominant fast inhibitor in most of the bee brain (Sachse et al., 2006).
Beyond the AL, GABA’s influence on memory is prominently seen in the mushroom bodies (MBs). The MB calyces (input regions) receive olfactory input and are innervated by a notable GABAergic neuron called the anterior paired lateral (APL) neuron (or a set of feedback neurons) that broadly connects MB output back to input (Raccuglia and Mueller, 2013; Boitard et al., 2015). Recent research has illuminated the function of this GABAergic feedback in cognitive flexibility. Reversal learning, wherein a bee must learn that a previously rewarded odor is now unrewarded (and vice versa), requires the bee to suppress or “unlearn” the first association and acquire a new one. Localized drug injections in vivo showed that if GABA_A receptors in the MB calyces are blocked (preventing GABA’s action), bees become impaired in reversal learning while still being able to learn initial odor discriminations (Boitard et al., 2015). Specifically, inhibiting ionotropic GABAergic signaling in the calyx caused bees to persist in responding to the formerly rewarded odor, unable to adjust to the reversed contingency (Boitard et al., 2015). In contrast, blocking GABA_B (metabotropic) receptors had no such effect, and blocking GABA_A in the MB lobes (output region) did not affect reversal learning. These results indicate that fast GABAergic feedback onto MB input neurons is necessary for clearing or suppressing the old memory trace, thereby allowing a new memory to form-essentially facilitating contextual behavioral flexibility. In the absence of this inhibition, the original memory exerts a proactive interference that the bee cannot overcome.
GABA also appears to modulate the general arousal and threshold for learning. Honey bees under GABA_A antagonists sometimes show signs of hyperexcitability or motor side effects (e.g., extended wing posture or tremors) (Mustard et al., 2020), suggesting GABA normally keeps neural excitation in check. In learning experiments, slight disinhibition (via low doses of antagonist) can transiently increase responsiveness, but stronger disinhibition impairs performance, presumably by causing excessive, uncoordinated neural activity (El Hassani et al., 2008). This delicate balance confirms that an optimal level of inhibition is needed for memory formation-too little and the signal-to-noise ratio may worsen, too much and necessary signal propagation is curtailed.
At the receptor level, the honey bee genome encodes multiple subunits of GABA_A receptors (including the insect-specific RDL subunit known for resistance to certain insecticides) and GABA_B receptors (Henry et al., 2020). Expression studies show GABA_A subunits are present in the MB calyx and lobes, aligning with sites of GABAergic innervation. One study found evidence for tonic GABA_A receptor activity regulating Kenyon cell excitability, meaning Kenyon cells (the intrinsic MB neurons) are under constant inhibitory tone that can be lifted or enhanced as needed (Palmer and Harvey, 2014). Such tonic inhibition might set the threshold for these neurons to respond to input, thereby affecting whether a given stimulus can induce plasticity. In Drosophila, RDL-mediated inhibition in MBs has been shown to constrain memory formation (Liu et al., 2007); something similar might occur in honey bees.
In summary, GABAergic systems provide essential inhibitory control that shapes honey bee learning and memory. They sharpen sensory representations in the antennal lobes and enable flexible updating of associations in the mushroom bodies (Boitard et al., 2015). GABA’s role in the MBs, particularly via feedback neurons, underscores that forgetting or suppressing outdated information is an active process needed for efficient new learning. Thus, GABA acts as a gatekeeper, preventing runaway excitation and ensuring that only meaningful patterns of neural activity are consolidated into memory. As new techniques like optical imaging and genetic manipulation are applied to honey bees, we can expect even deeper insights into how spatiotemporal patterns of GABA release during training modulate memory strength and specificity.
DOPAMINE: AVERSIVE REINFORCEMENT AND BEYOND
Dopamine (DA) is a biogenic amine with prominent roles in the reward and punishment systems of insects (Liu et al., 2012; Jiang and Litwin-Kumar, 2021). In honey bees, dopamine has long been associated primarily with aversive reinforcement-signaling negative or punishing events-in the context of associative learning (Vergoz et al., 2007). Early pharmacological studies demonstrated that blocking dopamine receptors in bees prevented the formation of aversive memories (for example, learning to associate an odor with an electric shock), whereas blocking octopamine (the primary “reward” neurotransmitter in insects) did not affect aversive learning (Unoki et al., 2005; Agarwal et al., 2011). Conversely, direct activation of dopamine pathways could substitute for a punishment in conditioning. These findings mirror the division of labor between dopamine and octopamine observed in other insects like crickets and wasps, and stand in intriguing contrast to Drosophila melanogaster, where different dopaminergic neurons mediate both punishment and reward (Klappenbach et al., 2013; Matsumoto et al., 2015). In honey bees, the prevailing model has been that dopaminergic neurons convey the unconditioned stimulus (US) information for aversive events, such as an electric shock or bitter taste, to the mushroom bodies, thereby reinforcing an odor as something to avoid (Jarriault et al., 2018).
A key piece of direct evidence for dopamine’s role in aversive learning came from real-time measurements of DA release in the bee brain (Jarriault et al., 2018). Using fast-scan cyclic voltammetry, researchers recorded dopamine transients in the mushroom bodies of live bees experiencing aversive stimuli. They found that an electric shock to a bee triggers a rapid, transient surge of dopamine in the mushroom body lobes (Jarriault et al., 2018). The magnitude of this dopamine release correlates with the intensity of the shock and the bee’s behavioral reaction (e.g., a vigorous sting extension response). Notably, when an odor was paired with the shock (as in an aversive conditioning paradigm), the presence of the odor enhanced dopamine release beyond what shock alone produced. This suggests a mechanism whereby an odorant, once it gains predictive value for a shock, can itself elicit or amplify dopaminergic activity via associative processes. Such activity-dependent DA release likely underpins synaptic plasticity in the mushroom bodies leading to the formation of an odor-aversive memory. These findings solidify that dopamine is the biochemical reinforcement signal for punishment in bees, analogous to how an appetitive US (like sugar) triggers octopamine release.
Honey bees have multiple dopamine receptor subtypes (classified into D1-like and D2-like families) expressed in their brains. For instance, genes AmDOP1, AmDOP2 (both D1-class GPCRs) and AmDOP3 (D2-class) have been identified (Blenau et al., 1998; Humphries et al., 2003). There is evidence that these receptors have distinct distributions and functions: D1-like receptors (which stimulate cAMP signaling) are abundant in mushroom bodies and are thought to mediate the reinforcing effects of dopamine on memory formation (Blenau et al., 1998). Indeed, an antagonist like fluphenazine that broadly blocks dopamine receptors can inhibit aversive learning in bees, as well as certain motivational aspects of behavior (Vergoz et al., 2007; Matsumoto et al., 2015). One study found that pharmacologically elevating dopamine levels (or stimulating D1 receptors) in bees can interfere with appetitive memory formation (Klappenbach et al., 2013), presumably by introducing a “punishment” signal or otherwise disrupting the normal balance of amines during reward learning. This highlights that dopamine’s effects on memory depend on context: in an appetitive context, excess dopamine may be misinterpreted as a negative prediction error, thus impairing reward learning.
Intriguingly, recent research has expanded our view of dopamine in honey bees beyond aversive reinforcement (Huang et al., 2022). It appears that dopamine also plays a role in positive motivation-essentially a “wanting” system for rewards-although this function is separable from the classic octopamine-mediated reward pathway. Huang et al. (2022) reported that when honey bees are starved (increasing their appetite), brain dopamine levels rise and this is linked to heightened sucrose responsiveness and more vigorous foraging behavior (Huang et al., 2022). In that study, transient surges of dopamine were observed in forager bees precisely at the moments when they discovered a rich food source and initiated a waggle dance to communicate it. Blocking dopamine receptors in such starved bees reduced their propensity to forage and dance, suggesting that dopamine is required to translate an internal hunger state into the motivation to seek and communicate food. This dopaminergic “appetitive motivation” system in bees is somewhat analogous to the role of mesolimbic dopamine in mammalian reward-seeking, though it likely interacts with octopamine-dependent circuits (octopamine still being the immediate reward signal for sucrose reception). Notably, this finding does not contradict the aversive reinforcement role of dopamine; rather, it shows that dopamine has dual facets-driving goal-directed behavior under hunger and encoding aversive outcomes in learning. In both cases, dopamine may be modulating the overarching behavioral state (high motivation or strong aversion) that then biases learning and memory formation.
On a systems level, dopaminergic neurons in the honey bee brain innervate not only the mushroom bodies but also other regions like the antennal lobes and the central complex. They form part of neural circuits for more complex behaviors. For example, in honey bee navigation and dance communication, differences in dopamine signaling have been observed between bees that follow waggle dances and those that rely on personal foraging experience (Kennedy et al., 2021). Additionally, dopamine levels in the antennal lobes can be modulated by sensory input and may affect how salient an odor is perceived (e.g., food odors might induce dopamine release that enhances odor memory retrieval in the appropriate context) (Perk and Mercer, 2006; Finkelstein et al., 2019).
In conclusion, dopamine in honey bees is a versatile neuromodulator central to learning and motivated behavior. It classically mediates aversive learning by signaling punishment to the neural circuits that store memories. Beyond this, it also contributes to positive motivation, particularly under conditions of high need, by activating neural pathways that energize and reinforce foraging efforts (Huang et al., 2022). These roles are mediated by distinct populations of dopaminergic neurons and receptor pathways, ensuring that bees appropriately learn from negative experiences and vigorously pursue valuable resources when necessary. Understanding dopamine’s full spectrum of action in bees provides insight into how internal states and reinforcement signals are integrated to shape adaptive behavior.
OCTOPAMINE: REWARD SIGNALING AND AROUSAL
Octopamine (OA) is often termed the invertebrate analog of norepinephrine, and in honey bees it functions as the primary neurotransmitter mediating positive reinforcement and reward (Hammer, 1993). A wealth of studies over the past decades have established that octopamine signals the presence of rewarding stimuli-most notably the taste of sucrose-during learning (Hammer and Menzel, 1998; Farooqui et al., 2003; Scheiner, 2004). In the honey bee brain, a subset of octopaminergic neurons are activated by sucrose on the bee’s tongue (the unconditioned stimulus in appetitive conditioning) and release octopamine in regions such as the antennal lobes and mushroom bodies (Barron et al., 2002). The most famous of these is the neuron called VUMmx1, which has its cell body in the subesophageal zone and projections to the primary olfactory areas (Schroter et al., 2007); this single neuron can mediate the reinforcing effect of a sugar reward by pairing an odor representation with octopamine release (Hammer and Menzel, 1995). The seminal experiment by Hammer and Menzel (1998) showed that artificial stimulation of VUMmx1 or exogenous octopamine application could substitute for a sucrose reward in conditioning-causing bees to learn an odor as if it were associated with sugar (Hammer and Menzel, 1998). This firmly identified octopamine as the reward neurotransmitter in honey bee associative learning.
Honey bees have several octopamine receptor subtypes, all of which are G-protein-coupled receptors (GPCRs) akin to adrenergic receptors (Grohmann et al., 2003; Beggs et al., 2011). These include Octβ receptors (octopamine beta-adrenergic-like receptors) and Octα/Octopamine-Tyramine receptors. Activation of octopamine receptors generally leads to increased cAMP or Ca2+ signaling in target neurons, resulting in modulatory effects such as increased excitability or enhanced synaptic plasticity (Grohmann et al., 2003). In the mushroom bodies, octopamine receptors on Kenyon cells modulate the strength of sensory inputs (Farooqui et al., 2003). When octopamine is released coincidentally with an odor signal (as occurs during reward conditioning), it induces intracellular cascades that cause the Kenyon cells to become more responsive to that odor or to form memory traces. Blocking octopamine signaling-for instance with the antagonist mianserin-prevents bees from forming normal appetitive memories (Matsumoto et al., 2015). Mianserin-treated bees can detect the odor and sugar, but fail to associate the two, underscoring that octopamine is required to assign reward value to the conditioned stimulus.
Beyond its direct role in associative memory, octopamine broadly influences honey bee behavior in ways that support foraging and responsiveness to rewards. Octopamine elevates the general arousal level of bees: it increases locomotor activity, enhances responsiveness to sucrose, and can stimulate metabolic processes. Laboratory assays have shown that injecting octopamine into a honey bee increases its sucrose sensitivity, meaning the bee will exhibit proboscis extension to lower concentrations of sugar solution than it normally would without octopamine stimulation (Tsvetkov et al., 2019). This effect, also produced by tyramine (a closely related amine, see next section) (Scheiner, 2004), effectively puts the bee in a “hungry”, more reward-seeking state. High levels of octopamine can actually decrease responsiveness if they go beyond an optimal range, suggesting a complex dose-response relationship. Additionally, octopamine plays a role in how honey bees divide labor; foragers tend to have more octopamine in their brains than nurse bees, and increasing octopamine experimentally can make younger bees start foraging earlier (Schulz et al., 2002). Octopamine also modulates the honey bee dance communication-studies found that feeding bees with octopamine or increasing its levels made them more likely to perform recruitment dances when they find food, whereas blocking octopamine could suppress dancing (Barron et al., 2002). This indicates that octopamine not only helps a bee learn that a flower is rewarding, but also encourages the honey bee to seek out and signal those rewards.
In the neural circuitry, octopamine acts on multiple levels. In the antennal lobes, octopamine can modulate the odor processing by acting on both projection neurons and local interneurons, effectively heightening the salience of food-related odors (Rein et al., 2013). A study in 2019 showed that foraging experiences that elevate octopamine (and dopamine) levels in the AL can durably increase a bee’s sensitivity to nectar scents (Finkelstein et al., 2019). In the mushroom bodies, octopamine’s effect is classically to coincide with the CS-US pairing, but it may also affect memory consolidation, as octopamine is known to activate cAMP/PKA signaling pathways which are involved in memory stabilization (similar to the role of adrenaline/noradrenaline in vertebrates) (Farooqui, 2007). Some evidence suggests that octopamine treatment after learning can enhance memory retention under certain conditions, possibly by reinforcing the memory trace during the early consolidation period. However, results are mixed, indicating timing and concentration are critical.
In summary, octopamine in honey bees is the quintessential neuromodulator of reward. It transforms sensory experiences of sweetness into neural signals that promote memory formation and positive reinforcement (Ai and Farina, 2023). By adjusting sensory responsiveness and motor patterns, it gears the honey bee toward efficient foraging and exploitation of resources. At the molecular level, octopamine receptors trigger signaling cascades that likely converge on memory-associated proteins and gene expression changes needed for long-term memory. Through its interplay with dopamine (for negative reinforcement) and other modulators, octopamine helps maintain the balance between seeking rewards and heeding risks that is vital for a foraging insect’s survival.
CONCLUSION
Honey bees demonstrate remarkable learning and memory abilities, supported by an intricate network of neurotransmitters and neuromodulators-including acetylcholine, glutamate, GABA, dopamine, octopamine, serotonin, and tyramine. Rather than acting in isolation, these chemical signals interact dynamically to encode, stabilize, and adapt memories across various behavioral contexts.
Acetylcholine drives fast excitatory transmission for associative learning, while glutamate contributes to memory consolidation through both excitatory and inhibitory signaling. GABA ensures neural precision and flexibility, preventing runaway excitation. Biogenic amines provide reinforcement cues: dopamine encodes punishment, octopamine conveys reward, and serotonin and tyramine shape behavioral states like hunger or stress. Additional modulators such as histamine and sNPF finetune learning under specific conditions. Importantly, many of these mechanisms mirror principles found in vertebrate systems, pointing to evolutionary convergence in how brains learn. Understanding these neuromodulatory systems in honey bees also has practical implications-especially as pesticides often target these very receptors-impacting honey bee cognition and pollination efficiency. Finally, insights from the honey bee brain are already inspiring artificial intelligence systems, particularly in reinforcement learning.
Ongoing research into molecular cascades and real-time neuromodulator dynamics promises to deepen our understanding of how even small brains achieve complex, adaptable behavior. This knowledge not only informs pollinator health but enriches the broader field of comparative neuroscience.
Acknowledgments
This work was supported by a grant from the National Institute of Agricultural Sciences, Rural Development Administration, Republic of Korea (Project No.: RS-2023-00232749).
References
-
Agarwal, M., M. Giannoni Guzman, C. Morales-Matos, R. A. Del Valle Diaz, C. I. Abramson and T. Giray. 2011. Dopamine and octopamine influence avoidance learning of honey bees in a place preference assay. PLoS One 6(9): e25371.
[https://doi.org/10.1371/journal.pone.0025371]

-
Ai, H. and W. M. Farina. 2023. In search of behavioral and brain processes involved in honey bee dance communication. Front. Behav. Neurosci. 17: 1140657.
[https://doi.org/10.3389/fnbeh.2023.1140657]

-
Barbara, G. S., B. Grunewald, S. Paute, M. Gauthier and V. Raymond-Delpech. 2008. Study of nicotinic acetylcholine receptors on cultured antennal lobe neurones from adult honeybee brains. Invert. Neurosci. 8(1): 19-29.
[https://doi.org/10.1007/s10158-007-0062-2]

-
Barbara, G. S., C. Zube, J. Rybak, M. Gauthier and B. Grunewald. 2005. Acetylcholine, GABA and glutamate induce ionic currents in cultured antennal lobe neurons of the honeybee, Apis mellifera. J. Comp. Physiol. A Neuroethol. Sens. Neural. Behav. Physiol. 191(9): 823-836.
[https://doi.org/10.1007/s00359-005-0007-3]

-
Barron, A. B., D. J. Schulz and G. E. Robinson. 2002. Octopamine modulates responsiveness to foraging-related stimuli in honey bees (Apis mellifera). J. Comp. Physiol. A Neuroethol. Sens. Neural. Behav. Physiol. 188(8): 603-610.
[https://doi.org/10.1007/s00359-002-0335-5]

-
Beggs, K. T., J. D. Tyndall and A. R. Mercer. 2011. Honey bee dopamine and octopamine receptors linked to intracellular calcium signaling have a close phylogenetic and pharmacological relationship. PLoS One 6(11): e26809.
[https://doi.org/10.1371/journal.pone.0026809]

-
Bestea, L., M. Paoli, P. Arrufat, B. Ronsin, J. Carcaud, J. C. Sandoz, R. Velarde, M. Giurfa and M. G. de Brito Sanchez. 2022. The short neuropeptide F regulates appetitive but not aversive responsiveness in a social insect. iScience 25(1): 103619.
[https://doi.org/10.1016/j.isci.2021.103619]

-
Blenau, W. and M. Thamm. 2011. Distribution of serotonin (5-HT) and its receptors in the insect brain with focus on the mushroom bodies: lessons from Drosophila melanogaster and Apis mellifera. Arthropod Struct. Dev. 40(5): 381-394.
[https://doi.org/10.1016/j.asd.2011.01.004]

-
Blenau, W., J. Erber and A. Baumann. 1998. Characterization of a dopamine D1 receptor from Apis mellifera: cloning, functional expression, pharmacology, and mRNA localization in the brain. J. Neurochem. 70(1): 15-23.
[https://doi.org/10.1046/j.1471-4159.1998.70010015.x]

-
Boitard, C., J. M. Devaud, G. Isabel and M. Giurfa. 2015. GABAergic feedback signaling into the calyces of the mushroom bodies enables olfactory reversal learning in honey bees. Front. Behav. Neurosci. 9: 198.
[https://doi.org/10.3389/fnbeh.2015.00198]

-
Bornhauser, B. C. and E. P. Meyer. 1997. Histamine-like immunoreactivity in the visual system and brain of an orthopteran and a hymenopteran insect. Cell Tissue Res. 287(1): 211-221.
[https://doi.org/10.1007/s004410050747]

-
Cartereau, A., X. Pineau, J. Lebreton, M. Mathe-Allainmat, E. Taillebois and S. H. Thany. 2022. Impairments in learning and memory performances associated with nicotinic receptor expression in the honeybee Apis mellifera after exposure to a sublethal dose of sulfoxaflor. PLoS One 17(8): e0272514.
[https://doi.org/10.1371/journal.pone.0272514]

-
Dacks, A. M., T. A. Christensen and J. G. Hildebrand. 2006. Phylogeny of a serotonin-immunoreactive neuron in the primary olfactory center of the insect brain. J. Comp. Neurol. 498(6): 727-746.
[https://doi.org/10.1002/cne.21076]

-
Demares, F., F. Drouard, I. Massou, C. Crattelet, A. Loeuillet, C. Bettiol, V. Raymond and C. Armengaud. 2014. Differential involvement of glutamate-gated chloride channel splice variants in the olfactory memory processes of the honeybee Apis mellifera. Pharmacol. Biochem. Behav. 124: 137-144.
[https://doi.org/10.1016/j.pbb.2014.05.025]

-
Dupuis, J. P., M. Gauthier and V. Raymond-Delpech. 2011. Expression patterns of nicotinic subunits alpha2, alpha7, alpha8, and beta1 affect the kinetics and pharmacology of ACh-induced currents in adult bee olfactory neuropiles. J. Neurophysiol. 106(4): 1604-1613.
[https://doi.org/10.1152/jn.00126.2011]

-
Eiri, D. M. and J. C. Nieh. 2012. A nicotinic acetylcholine receptor agonist affects honey bee sucrose responsiveness and decreases waggle dancing. J. Exp. Biol. 215(Pt 12): 2022-2029.
[https://doi.org/10.1242/jeb.068718]

-
El Hassani, A. K., M. Giurfa, M. Gauthier and C. Armengaud. 2008. Inhibitory neurotransmission and olfactory memory in honeybees. Neurobiol. Learn. Mem. 90(4): 589-595.
[https://doi.org/10.1016/j.nlm.2008.07.018]

-
El Hassani, A. K., S. Schuster, Y. Dyck, F. Demares, G. Leboulle and C. Armengaud. 2012. Identification, localization and function of glutamate-gated chloride channel receptors in the honeybee brain. Eur. J. Neurosci. 36(4): 2409-2420.
[https://doi.org/10.1111/j.1460-9568.2012.08144.x]

-
Farooqui, T. 2007. Octopamine-mediated neuronal plasticity in honeybees: implications for olfactory dysfunction in humans. Neuroscientist 13(4): 304-322.
[https://doi.org/10.1177/10738584070130040501]

-
Farooqui, T., K. Robinson, H. Vaessin and B. H. Smith. 2003. Modulation of early olfactory processing by an octopaminergic reinforcement pathway in the honeybee. J. Neurosci. 23(12): 5370-5380.
[https://doi.org/10.1523/JNEUROSCI.23-12-05370.2003]

-
Finkelstein, A. B., C. S. Brent, M. Giurfa and G. V. Amdam. 2019. Foraging Experiences Durably Modulate Honey Bees’ Sucrose Responsiveness and Antennal Lobe Biogenic Amine Levels. Sci. Rep. 9(1): 5393.
[https://doi.org/10.1038/s41598-019-41624-0]

-
Gauthier, M. 2010. State of the art on insect nicotinic acetylcholine receptor function in learning and memory. Adv. Exp. Med. Biol. 683: 97-115.
[https://doi.org/10.1007/978-1-4419-6445-8_9]

-
Gauthier, M., M. Dacher, S. H. Thany, C. Niggebrugge, P. Deglise, P. Kljucevic, C. Armengaud and B. Grunewald. 2006. Involvement of alpha-bungarotoxin-sensitive nicotinic receptors in long-term memory formation in the honeybee (Apis mellifera). Neurobiol. Learn. Mem. 86(2): 164-174.
[https://doi.org/10.1016/j.nlm.2006.02.003]

-
Goldberg, F., B. Grunewald, H. Rosenboom and R. Menzel. 1999. Nicotinic acetylcholine currents of cultured Kkenyon cells from the mushroom bodies of the honey bee Apis mellifera. J. Physiol. 514(3): 759-768.
[https://doi.org/10.1111/j.1469-7793.1999.759ad.x]

-
Grohmann, L., W. Blenau, J. Erber, P. R. Ebert, T. Strunker and A. Baumann. 2003. Molecular and functional characterization of an octopamine receptor from honeybee (Apis mellifera) brain. J. Neurochem. 86(3): 725-735.
[https://doi.org/10.1046/j.1471-4159.2003.01876.x]

-
Grunewald, B. and P. Siefert. 2019. Acetylcholine and Its Receptors in Honeybees: Involvement in Development and Impairments by Neonicotinoids. Insects 10(12): 420.
[https://doi.org/10.3390/insects10120420]

-
Guez, D., H. Zhu, S. W. Zhang and M. V. Srinivasan. 2010. Enhanced cholinergic transmission promotes recall in honeybees. J. Insect Physiol. 56(9): 1341-1348.
[https://doi.org/10.1016/j.jinsphys.2010.04.022]

-
Hammer, M. 1993. An identified neuron mediates the unconditioned stimulus in associative olfactory learning in honeybees. Nature 366: 59-63.
[https://doi.org/10.1038/366059a0]

-
Hammer, M. and R. Menzel. 1995. Learning and memory in the honeybee. J. Neurosci. 15(3): 1617-1630.
[https://doi.org/10.1523/JNEUROSCI.15-03-01617.1995]

- Hammer, M. and R. Menzel. 1998. Multiple sites of associative odor learning as revealed by local brain microinjections of octopamine in honeybees. Learn. Mem. 5(1-2): 146-156. https://www.ncbi.nlm.nih.gov/pubmed/10454379, .
-
Han, B., Q. Wei, F. Wu, H. Hu, C. Ma, L. Meng, X. Zhang, M. Feng, Y. Fang, O. Rueppell and J. Li. 2021. Tachykinin signaling inhibits task-specific behavioral responsiveness in honeybee workers. eLife 10: e64830.
[https://doi.org/10.7554/eLife.64830]

-
Henry, C., T. Cens, P. Charnet, C. Cohen-Solal, C. Collet, J. van-Dijk, J. Guiramand, M. C. de Jesus-Ferreira, C. Menard, N. Mokrane, J. Roussel, J. B. Thibault, M. Vignes and M. Rousset. 2020. Heterogeneous expression of GABA receptor-like subunits LCCH3 and GRD reveals functional diversity of GABA receptors in the honeybee Apis mellifera. Br. J. Pharmacol. 177(17): 3924-3940.
[https://doi.org/10.1111/bph.15135]

-
Huang, J., Z. Zhang, W. Feng, Y. Zhao, A. Aldanondo, M. G. de Brito Sanchez, M. Paoli, A. Rolland, Z. Li, H. Nie, Y. Lin, S. Zhang, M. Giurfa and S. Su. 2022. Food wanting is mediated by transient activation of dopaminergic signaling in the honey bee brain. Science 376(6592): 508-512.
[https://doi.org/10.1126/science.abn9920]

-
Humphries, M. A., J. A. Mustard, S. J. Hunter, A. Mercer, V. Ward and P. R. Ebert. 2003. Invertebrate D2 type dopamine receptor exhibits age-based plasticity of expression in the mushroom bodies of the honeybee brain. J. Neurobiol. 55(3): 315-330.
[https://doi.org/10.1002/neu.10209]

-
Ihle, K. E., N. A. Baker and G. V. Amdam. 2014. Insulin-like peptide response to nutritional input in honey bee workers. J. Insect Physiol. 69: 49-55.
[https://doi.org/10.1016/j.jinsphys.2014.05.026]

-
Ismail, N., G. E. Robinson and S. E. Fahrbach. 2006. Stimulation of muscarinic receptors mimics experience-dependent plasticity in the honey bee brain. Proc. Natl. Acad. Sci. USA. 103(1): 207-211.
[https://doi.org/10.1073/pnas.0508318102]

-
Jarriault, D., J. Fuller, B. I. Hyland and A. R. Mercer. 2018. Dopamine release in mushroom bodies of the honey bee (Apis mellifera L.) in response to aversive stimulation. Sci. Rep. 8(1): 16277.
[https://doi.org/10.1038/s41598-018-34460-1]

-
Jiang, L. and A. Litwin-Kumar. 2021. Models of heterogeneous dopamine signaling in an insect learning and memory center. PLoS Comput. Biol. 17(8): e1009205.
[https://doi.org/10.1371/journal.pcbi.1009205]

-
Jones, A. K., V. Raymond-Delpech, S. H. Thany, M. Gauthier and D. B. Sattelle. 2006. The nicotinic acetylcholine receptor gene family of the honey bee, Apis mellifera. Genome Res. 16(11): 1422-1430.
[https://doi.org/10.1101/gr.4549206]

-
Kennedy, A., T. Peng, S. M. Glaser, M. Linn, S. Foitzik and C. Gruter. 2021. Use of waggle dance information in honey bees is linked to gene expression in the antennae, but not in the brain. Mol. Ecol. 30(11): 2676-2688.
[https://doi.org/10.1111/mec.15893]

-
Klappenbach, M., L. Kaczer and F. Locatelli. 2013. Dopamine interferes with appetitive long-term memory formation in honey bees. Neurobiol. Learn. Mem. 106: 230-237.
[https://doi.org/10.1016/j.nlm.2013.09.011]

-
Krofczik, S., R. Menzel and M. P. Nawrot. 2008. Rapid odor processing in the honeybee antennal lobe network. Front. Comput. Neurosci. 2: 9.
[https://doi.org/10.3389/neuro.10.009.2008]

-
Kucharski, R., C. Mitri, Y. Grau and R. Maleszka. 2007. Characterization of a metabotropic glutamate receptor in the honeybee (Apis mellifera): implications for memory formation. Invert. Neurosci. 7(2): 99-108.
[https://doi.org/10.1007/s10158-007-0045-3]

-
Liu, C., P. Y. Placais, N. Yamagata, B. D. Pfeiffer, Y. Aso, A. B. Friedrich, I. Siwanowicz, G. M. Rubin, T. Preat and H. Tanimoto. 2012. A subset of dopamine neurons signals reward for odour memory in Drosophila. Nature 488(7412): 512-516.
[https://doi.org/10.1038/nature11304]

-
Liu, X., W. C. Krause and R. L. Davis. 2007. GABAA receptor RDL inhibits Drosophila olfactory associative learning. Neuron 56(6): 1090-1102.
[https://doi.org/10.1016/j.neuron.2007.10.036]

-
Locatelli, F., G. Bundrock and U. Muller. 2005. Focal and temporal release of glutamate in the mushroom bodies improves olfactory memory in Apis mellifera. J. Neurosci. 25(50): 11614-11618.
[https://doi.org/10.1523/JNEUROSCI.3180-05.2005]

- Lopatina, N. G., I. V. Ryzhova, T. G. Zachepilo, V. B. Smirnova and E. G. Chesnokova. 2004. L-glutamate and the formation of the long-term memory in the honey bee Apis mellifera. Zh. Evol Biokhim. Fiziol. 40(6): 539-545. https://www.ncbi.nlm.nih.gov/pubmed/15929416, .
-
Lozano, V. C., C. Armengaud and M. Gauthier. 2001. Memory impairment induced by cholinergic antagonists injected into the mushroom bodies of the honeybee. J. Comp. Physiol. A 187(4): 249-254.
[https://doi.org/10.1007/s003590100196]

-
Maleszka, R., P. Helliwell and R. Kucharski. 2000. Pharmacological interference with glutamate re-uptake impairs long-term memory in the honeybee, Apis mellifera. Behav. Brain Res. 115(1): 49-53.
[https://doi.org/10.1016/s0166-4328(00)00235-7]

-
Matsumoto, Y., C. S. Matsumoto, R. Wakuda, S. Ichihara and M. Mizunami. 2015. Roles of octopamine and dopamine in appetitive and aversive memory acquisition studied in olfactory conditioning of maxillary palpi extension response in crickets. Front. Behav. Neurosci. 9: 230.
[https://doi.org/10.3389/fnbeh.2015.00230]

-
Mussig, L., A. Richlitzki, R. Rossler, D. Eisenhardt, R. Menzel and G. Leboulle. 2010. Acute disruption of the NMDA receptor subunit NR1 in the honeybee brain selectively impairs memory formation. J. Neurosci. 30(23): 7817-7825.
[https://doi.org/10.1523/JNEUROSCI.5543-09.2010]

-
Mustard, J. A., L. Jones and G. A. Wright. 2020. GABA signaling affects motor function in the honey bee. J. Insect Physiol. 120: 103989.
[https://doi.org/10.1016/j.jinsphys.2019.103989]

-
Palmer, M. J. and J. Harvey. 2014. Honeybee Kenyon cells are regulated by a tonic GABA receptor conductance. J. Neurophysiol. 112(8): 2026-2035.
[https://doi.org/10.1152/jn.00180.2014]

-
Perk, C. G. and A. R. Mercer. 2006. Dopamine modulation of honey bee (Apis mellifera) antennal-lobe neurons. J. Neurophysiol. 95(2): 1147-1157.
[https://doi.org/10.1152/jn.01220.2004]

-
Raccuglia, D. and U. Mueller. 2013. Focal uncaging of GABA reveals a temporally defined role for GABAergic inhibition during appetitive associative olfactory conditioning in honeybees. Learn. Mem. 20(8): 410-416.
[https://doi.org/10.1101/lm.030205.112]

-
Rein, J., J. A. Mustard, M. Strauch, B. H. Smith and C. G. Galizia. 2013. Octopamine modulates activity of neural networks in the honey bee antennal lobe. J. Comp. Physiol. A Neuroethol. Sens. Neural Behav. Physiol. 199(11): 947-962.
[https://doi.org/10.1007/s00359-013-0805-y]

-
Sachse, S. and C. G. Galizia. 2002. Role of inhibition for temporal and spatial odor representation in olfactory output neurons: a calcium imaging study. J. Neurophysiol. 87(2): 1106-1117.
[https://doi.org/10.1152/jn.00325.2001]

-
Sachse, S., P. Peele, A. F. Silbering, M. Guhmann and C. G. Galizia. 2006. Role of histamine as a putative inhibitory transmitter in the honeybee antennal lobe. Front. Zool. 3: 22.
[https://doi.org/10.1186/1742-9994-3-22]

-
Schatton, A., J. Agoro, J. Mardink, G. Leboulle and C. Scharff. 2018. Identification of the neurotransmitter profile of AmFoxP expressing neurons in the honeybee brain using double-label in situ hybridization. BMC Neurosci. 19(1): 69.
[https://doi.org/10.1186/s12868-018-0469-1]

-
Scheiner, R. 2004. Responsiveness to sucrose and habituation of the proboscis extension response in honey bees. J. Comp. Physiol. A Neuroethol. Sens. Neural Behav. Physiol. 190(9): 727-733.
[https://doi.org/10.1007/s00359-004-0531-6]

-
Scheiner, R., S. Pluckhahn, B. Oney, W. Blenau and J. Erber. 2002. Behavioural pharmacology of octopamine, tyramine and dopamine in honey bees. Behav. Brain Res. 136(2): 545-553.
[https://doi.org/10.1016/s0166-4328(02)00205-x]

-
Schroter, U., D. Malun and R. Menzel. 2007. Innervation pattern of suboesophageal ventral unpaired median neurones in the honeybee brain. Cell Tissue Res. 327(3): 647-667.
[https://doi.org/10.1007/s00441-006-0197-1]

-
Schulz, D. J. and G. E. Robinson. 2001. Octopamine influences division of labor in honey bee colonies. J. Comp. Physiol. A 187(1): 53-61.
[https://doi.org/10.1007/s003590000177]

-
Schulz, D. J., A. B. Barron and G. E. Robinson. 2002. A role for octopamine in honey bee division of labor. Brain Behav. Evol. 60(6): 350-359.
[https://doi.org/10.1159/000067788]

-
Si, A., P. Helliwell and R. Maleszka. 2004. Effects of NMDA receptor antagonists on olfactory learning and memory in the honeybee (Apis mellifera). Pharmacol. Biochem. Behav. 77(2): 191-197.
[https://doi.org/10.1016/j.pbb.2003.09.023]

-
Sinakevitch, I. and N. J. Strausfeld. 2006. Comparison of octopamine-like immunoreactivity in the brains of the fruit fly and blow fly. J. Comp. Neurol. 494(3): 460-475.
[https://doi.org/10.1002/cne.20799]

-
Thamm, M., C. Scholl, T. Reim, K. Grubel, K. Moller, W. Rossler and R. Scheiner. 2017. Neuronal distribution of tyramine and the tyramine receptor AmTAR1 in the honeybee brain. J. Comp. Neurol. 525(12): 2615-2631.
[https://doi.org/10.1002/cne.24228]

-
Tsvetkov, N., C. N. Cook and A. Zayed. 2019. Effects of group size on learning and memory in the honey bee Apis mellifera. J. Exp. Biol. 222(9).
[https://doi.org/10.1242/jeb.193888]

-
Unoki, S., Y. Matsumoto and M. Mizunami. 2005. Participation of octopaminergic reward system and dopaminergic punishment system in insect olfactory learning revealed by pharmacological study. Eur. J. Neurosci. 22(6): 1409-1416.
[https://doi.org/10.1111/j.1460-9568.2005.04318.x]

-
Urlacher, E., L. Soustelle, M. L. Parmentier, H. Verlinden, M. J. Gherardi, D. Fourmy, A. R. Mercer, J. M. Devaud and I. Massou. 2016. Honey Bee Allatostatins Target Galanin/Somatostatin-Like Receptors and Modulate Learning: A Conserved Function? PLoS One 11(1): e0146248.
[https://doi.org/10.1371/journal.pone.0146248]

-
Vergoz, V., E. Roussel, J. C. Sandoz and M. Giurfa. 2007. Aversive learning in honeybees revealed by the olfactory conditioning of the sting extension reflex. PLoS One 2(3): e288.
[https://doi.org/10.1371/journal.pone.0000288]

-
Williamson, S. M. and G. A. Wright. 2013. Exposure to multiple cholinergic pesticides impairs olfactory learning and memory in honeybees. J. Exp. Biol. 216(10): 1799-1807.
[https://doi.org/10.1242/jeb.083931]

-
Wright, G. A., S. Softley and H. Earnshaw. 2015. Low doses of neonicotinoid pesticides in food rewards impair short-term olfactory memory in foraging-age honeybees. Sci. Rep. 5: 15322.
[https://doi.org/10.1038/srep15322]

-
Zannat, M. T., F. Locatelli, J. Rybak, R. Menzel and G. Leboulle. 2006. Identification and localisation of the NR1 sub-unit homologue of the NMDA glutamate receptor in the honeybee brain. Neurosci. Lett. 398(3): 274-279.
[https://doi.org/10.1016/j.neulet.2006.01.007]

-
Zhang, E. and J. C. Nieh. 2015. The neonicotinoid imidacloprid impairs honey bee aversive learning of simulated predation. J. Exp. Biol. 218(20): 3199-3205.
[https://doi.org/10.1242/jeb.127472]

