Establishment and Validation of an Analytical Method for Oleic Acid in Drone Pupae (Apis mellifera L.)
Abstract
This study established a rapid and simple liquid chromatography-tandem mass spectrometry (LC-MS/MS) method to quantify oleic acid in drone pupae (Apis mellifera L.). The method was validated for specificity, linearity, limit of detection and quantification, accuracy, and precision, and its applicability was evaluated using samples from beekeeping farms. The validated method showed a retention time of 5.22 min with an m/z value of 281.249, demonstrating high specificity. Linearity was confirmed over the concentration range of 0.889-17.780 μg/mL (R2≥0.99). The recovery ranged from 73.96 to 117.67%, with relative standard deviation of up to 10% for both the accuracy and precision. All validation parameters satisfied the acceptance criteria for residue and biological sample analysis and the SANTE (Health and Food Safety) guidelines. The validated LC-MS/MS method enables reliable quantification of oleic acid in drone pupae and can be applied to the development of functional food ingredients and the establishment of quality control standards. This approach is expected to contribute to value-added product development and increased income for beekeepers.
Keywords:
Drone pupae, Oleic acid, LC-MS/MS, Method validationINTRODUCTION
The domestic honey production value increased from approximately 425.6 billion KRW in 2013 to approximately 720.8 billion KRW in 2021, showing growth. However, the number of beekeeping farms has declined from approximately 34,102 in 2008 to approximately 26,890 in 2024, indicating a downward trend (Han, 2015; Lee, 2024; Park and Kim, 2024). This phenomenon can be interpreted as the combined result of several factors, including the expansion of production scale per farm and the growth of high value-added apicultural products. Beekeeping farms in Korea produce various apicultural products such as honey, propolis, pollen, and royal jelly. Currently, approximately 80% of the income of domestic beekeeping farms depends on honey sales (Kim et al., 2020c). Such heavy reliance on honey production has resulted in unstable farm income due to reduced honey yields and irregular flowering of nectar plants caused by climate change (Kim et al., 2021; RDA, 2022). Accordingly, the Beekeeping Industry Act was enacted, emphasizing the need to develop diverse apicultural products. Edible insects have attracted attention as potential alternative resource for promoting the branding of domestic apicultural products and securing stable income sources for beekeepers (Kim et al., 2019; Lee et al., 2019; RDA, 2022). Edible insects can serve as an alternative protein source to conventional livestock and have gained increasing interest as eco-friendly food resource because of their relatively low environmental impact (Oonincx et al., 2010; Choi et al., 2019). The market for edible insects, including drone pupae, is expected to grow from approximately 14,000 t in 2023 to 61,000 t in 2032 (Lee et al., 2025). Although the current production and sales volumes of alternative foods containing edible insects remain limited, the market is expected to expand steadily with increasing consumer demand.
According to the Food Code of the Ministry of Food and Drug Safety (MFDS) in Korea, nine insect species have been approved as edible insects: silkworms (Bombyx mori L.), grasshoppers (Oxya japonica Thungberg), white-spotted flower chafer larvae (Beauveria bassiana Vuill.), mealworm (Tenebrio molitor L.), white-spotted flower chafer (Protaetia brevitarsis L.), rhinoceros beetle (Allomyrina dichotoma L.), crickets (Gryllus bimaculatus), superworms (Zophobas atratus), and drone pupae (Apis mellifera L.) (Kim et al., 2021). Drone pupa are safe and nutritionally valuable food materials (Kim et al., 2020b; Zhou et al., 2011), which have recently been reclassified from temporarily approved food ingredients to general food ingredients. Additionally, its potential applications are expected to expand (Kim et al., 2023b). However, industrial utilization of drone pupae remains relatively low. To ensure sustainable industrial development, comprehensive research and institutional support are required to establish a stable supply system for raw materials, standardize quality control, verify functional properties, and develop products and distribution systems.
Drone pupae contain 22.33% carbohydrates, 48.52% crude protein, and 23.41% crude fat (Kim et al., 2020a). Among the lipid components, saturated and unsaturated fatty acids account for 13% and 10% of the total, respectively. Oleic acid constitutes approximately 93.8% of the total unsaturated fatty acids and is known to possess biofunctional properties such as anti-inflammatory and cholesterol-lowering effects (RDA, 2020; Kim et al., 2023b; Kim et al., 2024a). Drone pupae are considered a nutritionally valuable resource with physiological activity, and the establishment of a quality control system and development of functional food materials are required. Therefore, in this study, oleic acid, a major unsaturated fatty acid that reflects functionality and oxidative stability, was selected as the quality indicator. According to the U.S. FDA (2018), replacing fats high in saturated fatty acids with approximately 20 g per day of high-oleic oils in human intervention trials has been shown to potentially reduce the risk of cardiovascular diseases. In this study, the drone pupae powder and oil samples contained 7.92 g and 38.23 g of oleic acid per 100 g, respectively, corresponding to approximately 40% and 191% of the oleic acid intake reported in the literature. These results indicate that oleic acid possesses sufficient potential as a functional indicator compound for drone pupae. In addition, an analytical method for oleic acid determination was established and validated using LC-MS/MS.
MATERIALS AND METHODS
1. Preparation of reagents and standard solutions
The oleic acid standard was purchased from Sigma-Aldrich (Sigma-Aldrich, St. Louis, MO, USA). The reagents used in this study were high performance liquid chromatography (HPLC)-grade methanol (J.T.Baker, Phillipsburg, NJ, USA), LC-MS-grade formic acid (Li Chropur, MilliporeSigma, Billerica, MA, USA), and triple-distilled water (ELGA PureLab, ELGA LabWater, High Wycombe, UK). Oleic acid was dissolved in 100% methanol and serially diluted to final concentrations of 1.778, 4.445, 8.890, 17.780, and 26.670 μg/mL. Standard solutions were analyzed using LC-MS/MS and a calibration curve was constructed based on the peak area values obtained. Oleic acid content of the samples was quantified using a calibration curve.
2. Pre-treatment methods
Drone pupae of the western honeybee (Apis mellifera L.), aged 17-23 days after emergence, were collected from a beekeeping farm in Dangjin, Chungcheongnam-do, Korea, in 2024, and stored at -70℃ until use. Drone pupae oil was extracted following the method of Kim et al. (2023b), sealed in airtight containers to prevent oxidation, and stored at -20℃. For saponification of fatty acids, oil samples (2, 5, and 10 μL) were transferred into 1.5 mL microtubes, and 300 μL of 0.5 M KOH/MeOH solution was added. The mixtures were heated at 90℃ for 2 min, followed by ultrasonic extraction for 5 min using an ultrasonic cleaner (Branson 8510, Emerson Electric Co., Ltd., St. Louis, MO, USA). After sonication, 50 μL saturated NaCl and 500 μL hexane were added, vortexed, and extracted for an additional 5 min. The samples were centrifuged at 14,000 rpm (Hnail Science, Gimpo, Korea) and 400 μL of the hexane layer was collected. The solvent was completely evaporated using a speed vacuum concentrator(Caliper life science Inc, MA, USA) and the residue was dissolved in 300 μL of 100% methanol. The solution was centrifuged again (14,000 rpm, 10 min), and 100 μL of the supernatant was collected, diluted threefold with methanol, and transferred into vials for LC-MS analysis.
Drone pupae used for oil extraction were freeze-dried, ground into powder, and stored at -20℃ until analysis (Kim et al., 2019). For saponification of fatty acids, powdered samples (20, 40, and 80 mg) were placed into 1.5 mL microtubes, and 500 μL of 0.5 M KOH/MeOH solution was added. The mixtures was heated at 90℃ for 15 min, followed by ultrasonic treatment for 5 min. Subsequently, 50 μL of saturated NaCl and 500 μL of hexane were added, vortexed, and extracted for 5 min. After sonication, the samples were centrifuged at 14,000 rpm and 400 μL of the hexane layer was collected. The solvent was completely evaporated and the residue was dissolved in 300 μL of 100% methanol. The solution was centrifuged again (14,000 rpm, 10 min) and 100 μL of the supernatant was collected, diluted threefold with methanol, and transferred into vials for LC-MS analysis.
3. LC-MS/MS analytical conditions
Quantitative analysis of oleic acid was performed using ultra performance liquid chromatography-quadrupole time-of-flight mass spectrometry (UPLC-Q-TOF MS, Vion, Waters, Milford, MA, USA). The instrumental conditions are summarized in Table 1. The separation was carried out on a 2.1 mm×100 mm, 1.7 μm column (Waters). The mobile phase consisted of solvent A(0.1% formic acid in triple-distilled water) and B(0.1% formic acid in acetonitrile). The flow rate was set to 0.35 mL/min, column temperature was maintained at 40℃, and injection volume was 1 μL. The gradient elution was completed within 9 min. The analytes eluted from the column were detected using Q-TOF MS in negative electrospray ionization (ESI) mode.
4. Method validation
The validity of the LC-MS/MS analytical method for oleic acid detection was evaluated in terms of its specificity, linearity, accuracy, and precision. The specificity was confirmed by comparing the chromatograms of the standard and sample solutions to verify the coincidence of the major peaks. The peak areas were quantified by applying these to the calibration curve obtained from the standard solutions. Linearity was assessed using a five-point calibration curve and the coefficient of determination (R2) was calculated. The limit of detection (LOD) and limit of quantification (LOQ) were determined based on the slope (S) of the calibration curve and standard deviation (σ) of the response. The accuracy was evaluated by spiking the sample solution with a known amount of the standard solution and analyzing it in triplicate to calculate the recovery rate. Precision was assessed by measuring the samples at different concentrations five times, and repeatability and reproducibility were expressed as relative standard deviation (RSD).
RESULTS
1. Specificity
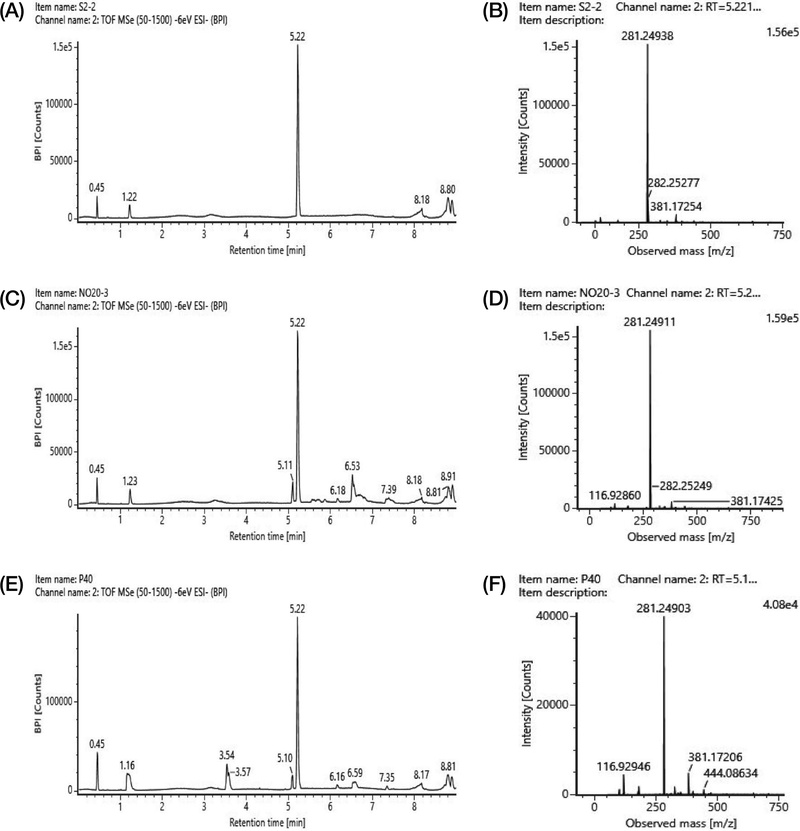
To evaluate the selective and accurate detection capability of the LC-MS/MS method for drone pupae oil and powder samples and the oleic acid standard, chromatographic separation and retention times (RT) were compared (Fig. 1). A single peak was detected at the same retention time (5.22 min) in all samples, which was separated from the other components (Fig. 1A, 1C, and 1E). In addition, the precursor ions of the peaks detected at 5.22 min in the drone pupae oil, powder samples, and oleic acid standard were compared. The measured m/z values were identical to three decimal places, confirming the formation of a single peak at the same position (Fig. 1B, 1D, and 1F).
2. Linearity, LOD and LOQ
Standard solutions of oleic acid were prepared at concentrations of 0.889, 1.778, 4.445, 8.890, and 17.780 μg/mL. The peak areas for each concentration were measured and a calibration curve was constructed using linear regression analysis (Table 2). The resulting calibration equation was y=113862x+54258, with an excellent coefficient of determination (R2) of 0.9965, indicating high linearity. The LOD was calculated by dividing 3.3 times the standard deviation by the mean slope of the calibration curve and the LOQ was calculated by dividing 10 times the standard deviation by the mean slope of the calibration curve. The calibration curve was measured in triplicate, yielding a mean slope of 120,077 and standard deviation of the y-intercept of 9,022.683. Using these values, the LOD and LOQ were determined to be 0.25 μg/mL and 0.75 μg/mL, respectively.
3. Accuracy and recovery rates
To evaluate the accuracy of the oleic acid standard solution, five replicate measurements were performed at concentrations of 2, 5, and 10 μg/mL (Table 3). The % RSD ranged from 4.30 to 7.80%, which was below 10%. This meets the acceptance criteria of ≤20% established by the MFDS guidelines for bioanalytical method validation and the SANTE guidelines (SANTE/11312/2021), confirming the validity of the analytical method (Díaz et al., 2023; MFDS, 2023). Additionally, oleic acid was spiked into drone pupae oil and powder samples at 2, 5, and 10 μL or 2.5, 5, and 10 μg, respectively, and analyzed in triplicate. The recovery rates were 91.49-111.18% for the oil and 73.96-117.67% for the powder, satisfying the 70-120% recovery criteria recommended in the residue analysis method guidelines (Schoenau, 2019).
4. Precision
Precision was evaluated in terms of repeatability and reproducibility, which correspond to sample-dependent repeatability and intermediate precision, respectively. To assess repeatability, drone pupae oil (10, 20, and 40 μL) and powder(20, 40, and 80 mg) samples were measured five times. The % RSD ranged from 4.01 to 7.07% for the oil and from 1.93 to 4.87% for the powder (Table 4), satisfying the ≤15% criterion reported by Zhou et al. (2011) for queen larva and drone pupae samples. Reproducibility was assessed by measuring the same samples using the same instrument on different days five times each by different analysts. The % RSD values were 9.04% for the oil and 7.68% for the powder (Table 5), meeting the ≤20% acceptance criteria of the MFDS bioanalytical method validation guidelines and SANTE guidelines (SANTE/11312/2021) (Díaz et al., 2023; MFDS, 2023).

Repeatability (intra-day precision) of the liquid chromatography-tandem mass spectrometry (LC-MS/MS) method for the analysis of drone pupae oil and powder
DISCUSSION
This study was conducted to establish an analytical method to quantify oleic acid in drone pupae (Apis mellifera L.). To enable a rapid and convenient quantification, an LC-MS/MS-based method was developed and validated. The method was validated for specificity, linearity, LOD, LOQ, accuracy, and precision. As a result, the method exhibited high specificity at RT 5.22 min and m/z 281.249, and good linearity with R2≥0.99 within a concentration range of 0.889-17.780 μg/mL. In addition, all validation parameters, including recovery (73.96-117.67%) and precision (RSD≤10%), met the acceptance criteria for residue analysis, bioanalytical method validation, SANTE guidelines, and previous validation criteria applied to queen larva and drone pupae samples, confirming the reliability of the developed method.
The optimized pretreatment procedure and LC-MS/MS quantification method developed in this study satisfied international validation standards, demonstrating that this oleic acid analytical method is suitable and applicable to various apicultural products. Previous studies have shown that analytical methods developed for food quality control have contributed to the management of nutritional components in food products (Shin et al., 2016; Hwang et al., 2019; Choi et al., 2022). Accordingly, oil and powder processed from drone pupae are expected to serve as valuable functional food materials, contributing to income enhancement for beekeeping farms. However, this study was limited to samples collected from a single apiary and drone pupae aged 17-23 days, and therefore may not fully reflect regional variations or developmental-stage characteristics. According to Gonçalves et al. (2025), drone pupae powder exhibits high protein and lipid content, indicating its nutritional superiority and potential as a food ingredient. Similarly, Ghosh et al. (2020) reported that as insect development progressed, the protein content increased, whereas the total fatty acid content decreased. Furthermore, the proportion of saturated fatty acids in drone pupae was higher than that of monounsaturated fatty acids, whereas the opposite trend was observed in adult drones. Overall, the current studies are limited in terms of storage stability, sensory properties, and consumer acceptability. Further studies are warranted to elucidate the effects of regional and developmental variations in the fatty acid composition of drone pupae.
Acknowledgments
This study was supported by the Cooperative Research Program for Agriculture, Science and Technology Development, Rural Development Administration, Republic of Korea (Project No. RS-2024-00399430). This study was supported by a 2025 Fellowship Program for Agricultural Science Research (National Institute of Agricultural Sciences, Rural Development Administration, Republic of Korea).
References
- Choi, H. M., S. G. Kim, H. Y. Kim, S. O. Woo and S. M. Han. 2022. Validation of UPLC analysis method for putrescine in lyophilized royal jelly. Korean J. Appl. Entomol. 614: 659-664.
-
Choi, H. M., S. M. Han, H. Y. Kim, S. O. Woo, S. G. Kim, K. W. Bang and H. J. Moon. 2019. Investigation of heavy metals from honeybee drone pupa (Apis mellifera L.) as an ingredient for novel foods. J. Apic. 34: 273-277.
[https://doi.org/10.17519/apiculture.2019.09.34.3.273]

-
Díaz, M. A., D. P. O’Connell, S. Jordan, C. O’Connor, P. Martin, J. C. Jones and J. Garvey. 2023. Analysis of pesticide levels in honey and pollen from Irish honey bee colonies using a modified, Dutch mini-Luke method with gas and liquid chromatography-tandem mass spectrometry detection. J. Agric. Food Chem. 71: 12657-12667.
[https://doi.org/10.1021/acs.jafc.3c02250]

-
Ghosh, S., H. Y. Sohn, S. J. Pyo, A. B. Jensen, V. B. Meyer-Rochow and C. Jung. 2020. Nutritional composition of Apis mellifera drones from Korea and Denmark as a potential sustainable alternative food source: Comparison between developmental stages. Foods. 9: 389.
[https://doi.org/10.3390/foods9040389]

-
Gonçalves, J. C., B. Vouga, C. A. Costa, F. Gonçalves, C. Coelho, R. P. F. Guiné and P. M. R. Correia. 2025. Production and characterization of powder from drone brood of honeybees (Apis mellifera). Appl. Food Res. 5: 100718.
[https://doi.org/10.1016/j.afres.2025.100718]

-
Han, J. H. 2015. A study on management of apiary and main factors for developing the beekeeping industry in Korea. J. Apic. 30: 127-133.
[https://doi.org/10.17519/apiculture.2015.06.30.2.127]

-
Hwang, K. M., J. W. Bae, S. J. Hu and K. S. Oh. 2019. Determination of β-carotene in infant formulas by high-performance liquid chromatography. J. Food Hyg. Saf. 34: 334-339.
[https://doi.org/10.13103/JFHS.2019.34.4.334]

-
Kim, H. Y., S. G. Kim, S. M. Kim, H. M. Choi, H. J. Moon and S. M. Han. 2021. Analysis on the optimal production seasonal of drone pupae (Apis mellifera L.) for the using food materials. J. Apic. 36: 23-29.
[https://doi.org/10.17519/apiculture.2021.04.36.1.23]

-
Kim, H. Y., S. O. Woo, H. M. Choi and S. M. Kim. 2024a. Analysis of fatty acid composition and phospholipid content of drone pupae (Apis mellifera L.) using different pretreatment methods. J. Apic. 39: 391-394.
[https://doi.org/10.17519/apiculture.2024.11.39.4.391]

-
Kim, H. Y., S. O. Woo, S. G. Kim, H. M. Choi, H. J. Moon and S. M. Han. 2020a. Antioxidant and antihyperglycemic effects of honeybee drone pupae (Apis mellifera L.) extracts. J. Apic. 35: 33-39.
[https://doi.org/10.17519/apiculture.2020.04.35.1.33]

-
Kim, H. Y., S. O. Woo, S. G. Kim, H. M. Choi, S. K. Kim, S. M. Kim, H. Y. Lee and S. M. Han. 2023a. Nutritional compositions and quality characteristics of beverages with added drone pupae powder in Korea. J. Apic. 38: 69-76.
[https://doi.org/10.17519/apiculture.2023.04.38.1.69]

-
Kim, H. Y., S. O. Woo, S. G. Kim, K. W. Bang, H. M. Choi, H. J. Moon and S. M. Han. 2019. Analysis of oxidative stability in drone pupae (Apis mellifera L.). J. Apic. 34: 63-66.
[https://doi.org/10.17519/apiculture.2019.04.34.1.63]

-
Kim, J. E., D. I. Kim, H. J. Kim, S. Y. Kim, Y. B. Lee, J. H. Moon, H. G. Park and Y. S. Choi. 2020b. Characteristics of hydrolysis of protein in drone pupa (Apis mellifera L.). J. Apic. 35: 169-177.
[https://doi.org/10.17519/apiculture.2020.09.35.3.169]

- Kim, J. E., D. I. Kim, H. Y. Koo, H. J. Kim, S. Y. Kim, Y. B. Lee, J. S. Kim, H. H. Kim, J. H. Moon and Y. S. Choi. 2020c. Analysis of nutritional compounds and antioxidant effect of freeze-dried powder of the honey bee (Apis mellifera L.) drone (pupal stage). Korean J. Appl. Entomol. 59: 265-275.
-
Kim, S. M., H. Y. Kim, S. O. Woo, S. G. Kim, H. M. Choi, H. J. Moon and S. M. Han. 2024b. Nutritional and sensory evaluation of bread incorporating Korean drone pupa. J. Apic. 39: 83-89.
[https://doi.org/10.17519/apiculture.2024.04.39.1.83]

-
Kim, S. M., S. G. Kim, S. O. Woo, H. Y. Kim, H. M. Choi, S. K. Kim, H. J. Lee, H. J. Moon, Y. S. Lee, S. Ryu and S. M. Han. 2023b. Cytokine profile in human skin keratinocytes exposed to drone (Apis mellifera L.) fat extract against inflammation. J. Apic. 38: 59-67.
[https://doi.org/10.17519/apiculture.2023.04.38.1.59]

- Lee, J. M., Y. L. Kim, C. H. Kim and S. H. Woo. 2019. Crisis and implications of the apiculture industry. Korea Rural Economic Institute. https://www.krei.re.kr/krei/page/15?cmd=view&bibliold=519237&pageIndex=1
- Lee, M. Y. 2024. Future directions for the Korean beekeeping industry. Chuksan News.
- Lee, Y. G., W. J. Song and K. J. Kim. 2025. Challenges for the livestock industry response to changes in the alternative food market. Korea Rural Economic Institute. https://www.krei.re.kr/krei/page/53?cmd=view&bibliold=542366&pageIndex=1
- MFDS (Ministry of Food and Drug Safety). 2023. https://www.mfds.go.kr/brd/m_1060/view.do?seq=15709
-
Oonincx, D. G. A. B., J. Van Itterbeeck, M. J. W. Heetkamp, H. Van Den Brand, J. J. A. Van Loon and A. Van Huis. 2010. An exploration on greenhouse gas and ammonia production by insect species suitable for animal or human consumption. PLoS One. 5: e14445.
[https://doi.org/10.1371/journal.pone.0014445]

- Park, H. R. and S. J. Kim. 2024. Current status and revitalization strategies of the beekeeping industry. Agricultural, Fisheries and Livestock News.
- RDA (Rural Development Administration). 2020. Study for new food raw material registration of honeybee pupa. Rural Development Administration.
- RDA (Rural Development Administration). 2022. Establishment of food standards for domestic bee products and development of high value-added food materials. Rural Development Administration.
-
Schoenau, E. A. 2019. Elements of method design. In current challenges and advancements in residue analytical methods. 3-16. American Chemical Society.
[https://doi.org/10.1021/bk-2019-1300.ch001]

-
Shin, Y. W., H. J. Lee, H. S. Ham, S. C. Shin, Y. J. Kang, K. M. Hwang, Y. K. Kwon, I. W. Seo, J. M. Oh and Y. E. Koo. 2016. Establishment of biotin analysis by LC-MS/MS method in infant milk formulas. J. Food Hyg. Saf. 31: 327-334.
[https://doi.org/10.13103/JFHS.2016.31.5.327]

- U.S. FDA (U.S. Food and Drug Administration). 2018. FDA Completes Review of Qualified Health Claim Petition for Oleic Acid and the Risk of Coronary Heart Disease. Accessed November 3, 2025.
-
Zhou, J., Y. Qi, Y. Hou, J. Zhao, Y. Li, X. Xue, L. Wu, J. Zhang and F. Chen. 2011. Quantitative determination of juvenile hormone III and 20-hydroxyecdysone in queen larvae and drone pupae of Apis mellifera by ultrasonic-assisted extraction and liquid chromatography with electrospray ionization tandem mass spectrometry. J. Chromatogr. B. 879: 2533-2541.
[https://doi.org/10.1016/j.jchromb.2011.07.006]